As cataract surgery has evolved, so have patients’ expectations. Customizing a surgical plan for patients with concurrent Fuchs endothelial dystrophy and a visually significant cataract is crucial to achieving successful outcomes.
PREOPERATIVE ASSESSMENT
The first step is to assess the severity of the endothelial disease and the likelihood of corneal decompensation after phacoemulsification. Surgeons must ask patients about diurnal fluctuations in vision. Eyelid closure results in reduced tear film evaporation and endothelial dysfunction from anoxic stress, leading to decreased vision after sleeping; recovery is often slower on humid days. Patients with fluctuating vision will benefit from combined endothelial keratoplasty and cataract surgery. Any history of bullae is also indicative of an imminent keratoplasty. The patients’ age, visual goals, and other medical problems must be taken into account. Because of the progressive nature of the Fuchs endothelial dystrophy, a 50-year-old patient has a greater lifetime risk of needing a keratoplasty than an older patient. Accordingly, the surgeon should have a lower threshold for considering a combined procedure for the former. In contrast, a nursing home patient with a short life expectancy or a patient with a history of poor adherence to prescribed therapy may benefit from phacoemulsification alone because of the faster and simpler postoperative recovery.
AT A GLANCE
• The first step is to assess the severity of the endothelial disease and the likelihood of corneal decompensation after phacoemulsification.
• If proceeding with a combined procedure, DMEK surgery achieves superior outcomes compared with DSAEK. Because of the known hyperopic shift after endothelial keratoplasty, cataract surgeons should target a myopic result when selecting the IOL power.
• Phaco chop is a better choice than divide and conquer in this population. Another alternative is prechopping and softening the nucleus with a femtosecond laser.
On examination, patients with significant Fuchs endothelial dystrophy often have an endothelial reflection that is brighter than the epithelial reflection, confluent corneal guttae, edema, haze, and/or a Descemet membrane that has a beaten-metal appearance. If a re-examination in the morning reveals subtle microcystic edema or if a trial of hypertonic saline solution improves vision, a combined procedure is indicated.
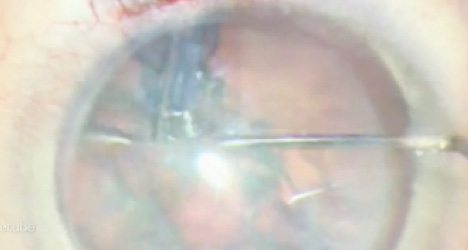
Figure. Chopping techniques rely more on vacuum than ultrasound power, minimizing the damage to the endothelial cells.
It is important to consider if the patient’s level of vision correlates to the extent of the cataract or if the endothelial disease is contributing to visual disturbances. Because Fuchs endothelial dystrophy reduces contrast sensitivity and causes glare, patients who complain of extreme glare that is out of proportion to their level of cataract when driving at night or in bright sunlight will generally be best served by a combined procedure. Ophthalmologists should also consider the risk of corneal decompensation from cataract removal. A patient with a mature brunescent cataract will require more ultrasound energy during surgery, increasing the risk of pseudophakic bullous keratopathy. Because this condition can cause permanent corneal scarring and haze, preempting its occurrence is essential.
Pachymetry values were long the primary factor in considering cataract surgery alone for these patients, with the upper limit’s being 640 µm.1 Because pachymetry values vary in the normal population, they have limited value unless historical data are available. Interestingly, in patients with subtle corneal edema, optical pachymetry using the Orbscan (Bausch + Lomb) overestimates corneal thickness compared with ultrasound pachymetry.2 This discrepancy may sway the surgeon to proceed with a combined procedure. Specular microscopy also has limited value in surgical decision making, because the endothelial count in patients with significant Fuchs is often immeasurable. In current practice, pachymetry and specular microscopy are used as screening tools for high-risk patients but should not be used alone to evaluate them for clinically significant Fuchs endothelial dystrophy.
PLANNING FOR AN ENDOTHELIAL KERATOPLASTY
Descemet membrane endothelial keratoplasty (DMEK) transplants only donor Descemet membrane and endothelium. If proceeding with a combined procedure, DMEK surgery achieves superior outcomes compared with Descemet stripping automated endothelial keratoplasty (DSAEK). In a study comparing outcomes among patients who had DMEK in one eye and DSAEK in the other, investigators reported a BCVA of 20/24 in the DMEK eye versus 20/32 in the DSAEK eye, and 85% of patients reported a better quality of vision in their DMEK eye.3
With DSAEK, the thicker lenticule causes significant light scatter from the graft-host stromal interface, which degrades the image quality and limits vision. The rate of rejection is also lower in DMEK (0.7%) than DSAEK (9%) or penetrating keratoplasty (17%).4
Because of the known hyperopic shift after endothelial keratoplasty, cataract surgeons should target a myopic result when selecting the IOL power. With DSAEK, the shape of the donor tissue induces a minus lens effect, creating approximately a +1.00 D hyperopic shift. In DMEK, an increase of +0.30 D in the spherical equivalent has been reported secondary to the decreased swelling in the posterior stroma and an associated steepening of the posterior corneal curvature.5
IMPROVING OUTCOMES WITH CATARACT SURGERY ALONE
Careful surgical planning, counseling, and excellent cataract surgery skills are required. Patients with Fuchs endothelial dystrophy should expect a slower recovery and be aware of their risk of future surgical intervention. Multifocal IOLs are contraindicated in this population because the lenses may exacerbate the visual disruption from the diseased endothelium. If during cataract surgery a capsular tear occurs, a sclera-sutured or sulcus-fixated posterior chamber IOL is preferable to an anterior chamber IOL in case endothelial keratoplasty is required in the future. Nd:YAG capsulotomy should be delayed in patients with Fuchs, because an open capsule can result in posterior dislocation of the IOL during future transplantation.
Preoperative pulsed therapy with difluprednate (Durezol; Alcon) has been shown to reduce inflammation, the loss of endothelial cells, and macular thickening as well as to restore postoperative vision more effectively than prednisolone acetate.6 Because the endothelial cell layer is derived from the neuroectoderm, when the inflammatory system is downregulated, the effects of surgical trauma are minimized.
Watch it Now
Divya M. Varu, MD, performs cataract surgery on a patient with Fuchs endothelial dystrophy.
Some ophthalmologists recommend creating a scleral tunnel incision so as to enter the eye more posteriorly and minimize corneal striae during phacoemulsification. It is also worth considering using BSS Plus (Alcon), which includes glutathione, sodium bicarbonate, and glucose, in order to create a more physiologic anterior chamber.
To protect the endothelium, many surgeons rely on a dispersive ophthalmic viscosurgical device (OVD) such as Viscoat (Alcon) or Endocoat (Abbott Medical Optics), which can be reapplied throughout the cataract procedure. Others use the techniques described by Steve Arshinoff, MD, in which the surgeon first coats the corneal endothelium with a dispersive OVD and then injects a cohesive (soft-shell technique) or viscoadaptive (ultimate soft-shell technique) OVD centrally to deepen the anterior chamber and force the dispersive OVD towards the cornea.7 In the ultimate soft-shell technique, a layer of balanced salt solution is injected below the viscoadaptive OVD to prevent it from being aspirated during phacoemulsification. At the end of the case, complete removal of the cohesive or viscoadaptive OVD is necessary, but surgeons can consider leaving a thin layer of the dispersive OVD in the eye to coat the endothelium. If concerned about a postoperative IOP spike, ophthalmologists may leave the eye slightly soft and/or preemptively treat the patient with an ocular hypotensive drop or oral acetazolamide.
During phacoemulsification, thermal and ultrasound energy damage the endothelial cells. In already susceptible eyes, this destruction can lead to permanent edema and haze. Techniques to minimize damage include lowering the bottle height, raising the vacuum setting, decreasing the phaco power, and disassembling the nucleus in the capsular bag. Compared with divide and conquer, chopping techniques reduce ultrasound energy and are preferable in these cases (Figure).8 In a 2010 survey, however, far fewer respondents were performing phaco chop compared to divide and conquer (32% vs 62%), likely because of the technical challenges associated with learning chopping techniques.9 Laser cataract surgery is thus an option to consider; the procedure reduces ultrasound time by prechopping and softening the nucleus. With advances in technology and the ability now to section the cataract without ultrasound energy, surgeons may be more inclined to perform cataract surgery alone and delay a transplant in patients with significant Fuchs endothelial dystrophy.
CONCLUSION
Cataract surgery can achieve successful outcomes in patients with Fuchs endothelial dystrophy. Key strategies include identifying high-risk individuals who may benefit from a combined procedure, carefully counseling patients preoperatively, and employing cataract surgery techniques that minimize endothelial damage and allow for successful future endothelial keratoplasty if needed.
1. Seitzman GD, Gottsch JD, Stark WJ. Cataract surgery in patients with Fuchs’ corneal dystrophy: expanding recommendations for cataract surgery without simultaneous keratoplasty. Ophthalmology. 2005;112(3):441-446.
2. Menees A, Thompson VM, Berdahl JP. Orbscan and ultrasound pachymetry measurement comparison in Fuchs endothelial dystrophy. Paper presented at: ASCRS/ASOA Congress & Symposium; April 24, 2012; Chicago, IL.
3. Guerra FP, Anshu A, Price MO, Price FW. Endothelial keratoplasty: fellow eyes comparison of Descemet stripping automated endothelial keratoplasty and Descemet membrane endothelial keratoplasty. Cornea. 2011;30(12):1382-1386.
4. Anshu A, Price MO, Price FW Jr. Risk of corneal transplant rejection significantly reduced with Descemet’s membrane endothelial keratoplasty. Ophthalmology. 2012;119(3):536-540.
5. Ham L, Dapena I, Moutsouris K, et al. Refractive change and stability after Descemet membrane endothelial keratoplasty. Effect of corneal dehydration-induced hyperopic shift on intraocular lens power calculation. J Cataract Refract Surg. 2011;37(8):1455-1464.
6. Donnenfeld ED, Holland EJ, Solomon KD, et al. A multicenter randomized controlled fellow eye trial of pulse-dosed difluprednate 0.05% versus prednisolone acetate 1% in cataract surgery. Am J Ophthalmol. 2011;152(4):609-617.
7. Arshinoff SA, Norman R. Tri-soft shell technique. J Cataract Refract Surg. 2013;39:1196-1203.
8. DeBry P, Olson RJ, Crandall AS. Comparison of energy required for phaco-chop and divide and conquer phacoemulsification. J Cataract Refract Surg. 1998;24:689-692.
9. 2010 Survey of US ASCRS Members. http://www.analeyz.com/AnaleyzASCRS2010.htm. Accessed March 25, 2016.
Divya M. Varu, MD
• private practice, Dell Laser Consultants, Austin, Texas
• dvaru@dellvision.com
• financial interest: none acknowledged