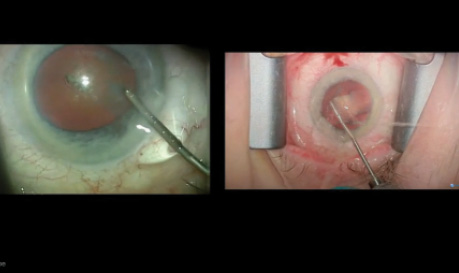
Pharmaceutical approaches.
By David A. Goldman, MD

In our pursuit of perfect cataract surgery, we ophthalmologists have recognized the need for good intraoperative mydriasis. Small pupils prolong surgery and increase the risk of complications, as noted in a recent American Society of Cataract and Refractive Surgery survey of patients with intraoperative floppy iris syndrome.1 This article reviews the pharmaceutical approaches to managing a small pupil for cataract surgery.
AT A GLANCE
• Small pupils are associated with more complex cataract surgery and a higher risk of complications.
• Omidria, an intraoperative mydriatic agent, is a new method of maintaining pupillary dilation perioperatively.
• When pharmacological methods and viscomydriasis do not provide sufficient pupillary expansion, devices can offer greater safety than surgical maneuvers for enlarging a miotic pupil.
• Dr. Stephenson finds the APX 200 easy to insert and remove, and its use has not required her to alter the location of incisions for other instrumentation.
PHARMACEUTICAL STRATEGIES
What began as preoperative cocktails of phenylephrine, tropicamide, and other dilating agents soon saw the addition of flurbiprofen and other nonsteroidal anti-inflammatory drugs (NSAIDs). Shown to disrupt the arachidonic acid cascade, specifically prostaglandin formation, topical NSAIDs became widely used in the preoperative regimen to minimize intraoperative miosis.
For patients with tamsulosin-induced intraoperative floppy iris syndrome, some physicians recommended preoperative dosing with atropine eye drops several days to a week before surgery. What likely became the most commonplace strategy in these patients was the use of epi-Shugarcaine, named after its discoverer, the late Joel Shugar, MD. He combined 9 mL of BSS Plus (Alcon), 3 mL of 4% preservative-free lidocaine, and 4 mL 1:1,000 of preservative-free and bisulfite-free epinephrine. Administered at the beginning of surgery, this solution not only improves iris tone, but in many cases, it also prevents the classic floppy iris scenario from occurring.
RECENT ADDITION
Since the development of epi-Shugarcaine, there has not been much innovation in intraoperative pharmaceuticals for cataract surgery with the exception of Omidria (Omeros). Recently FDA approved, Omidria is a combination of 1% phenylephrine and 0.3% ketorolac for addition to the irrigation solution prior to surgery. In this manner, the patient’s anterior chamber is constantly exposed to a dilating agent and an NSAID. In addition, phenylephrine has a-adrenergic effects alone. These may affect the cardiovascular system less than epinephrine, a nonselective adrenergic agonist, which has traditionally been added to the bottle.
Watch it Now
Robert J. Weinstock, MD, provides a side-by-side comparison of cataract surgery with and without Omidria (Omeros).
In a full-factorial phase 2b clinical trial in which all patients received standard preoperative topical mydriatic and anesthetic agents, only 6% of subjects receiving Omidria had an intraoperative pupillary diameter of less than 6 mm compared with 46% of those receiving vehicle control, 35% of those receiving ketorolac alone, and 22% receiving phenylephrine (data on file with Omeros). Phase 3 trials confirmed the beneficial effects of Omidria on pupillary diameter and on reducing postoperative ocular discomfort.2 In addition, data from postlaunch case-controlled studies to be reported at the upcoming meeting of the American Society of Cataract and Refractive Surgery show that Omidria statistically significantly reduced complication rates, decreased the use of pupil-expanding devices, shortened age-adjusted surgical times, improved visual acuity on the first postoperative day, and prevented miosis during the use of a femtosecond laser.3-5
CONCLUSION
Multiple options are available for the pharmaceutical dilation of a pupil for cataract surgery. The newest, Omidria, also represents a safe and effective method of maximizing surgical success and patients’ satisfaction.
1. Chang DF, Braga-Mele R, Mamalis N, et al, for the ASCRS Cataract Clinical Committee. Clinical experience with intraoperative floppy-iris syndrome: results of the 2008 ASCRS member survey. J Cataract Refract Surg. 2008;34:1201-1209.
2. Hovanesian JA, Sheppard JD, Trattler WB, et al. Intracameral phenylephrine and ketorolac during cataract surgery to maintain intraoperative mydriasis and reduce postoperative ocular pain: integrated results from 3 pivotal phase 3 studies. J Cataract Refract Surg. 2015;41(10):2060-2068.
3. Walters T, Schaaf D, Buznego C. Intracameral phenylephrine/ketorolac to maintain pupil diameter and reduce postoperative pain regardless of cataract surgery duration. Paper presented at: ASCRS/ASOA Symposium & Congress; May 9, 2016; New Orleans, LA.
4. Visco D. Use of iris fixation ring with and without intracameral phenylephrine/ketorolac in patients with poor pupil dilation. Paper presented at: ASCRS/ASOA Symposium & Congress; May 8, 2016; New Orleans, LA.
5. Rosenberg E, Nattis A, Alevi D, Donnenfeld E. Initial experience, visual outcomes, and efficacy of intracameral phenylephrine/ketorolac (1.0%/0.3%) during cataract surgery. Paper presented at: ASCRS/ASOA Symposium & Congress; May 9, 2016; New Orleans, LA.
David A. Goldman, MD
• private practice, Goldman Eye, Palm Beach Gardens, Florida
• (561) 630-7120; david@goldmaneye.com
• financial interest: none acknowledged
One surgeon’s experience with a new expansion device.

There is no one cause for small pupils, and thus no single device will be effective in all cases. Small pupils can be the result of medication use (eg, pilocarpine, tamsulosin), pseudoexfoliation, fibrosis due to other conditions, uveitis, posterior synechiae, or trauma. Sometimes, a pupil will not dilate well even when the iris shows no sign of an unusual condition. Pupils may also come down after laser cataract surgery.
SOME DEVICES FOR EXPANDING THE PUPIL
• Morcher Pupil Dilator (Morcher; distributed in the United States by FCI Ophthalmics)
• Malyugin Ring (MicroSurgical Technology)
• Graether 2000 Pupil Expander (Eagle Vision)
• Perfect Pupil Injectable (Milvella)
• Siepser Iris Protector Ring
• Oasis Iris Expander (Oasis Medical)
• APX 200 (APX Ophthalmology)
Miosis can hamper visualization, decreasing surgical safety and increasing the risk of damage to intraocular structures. A 6-mm pupil that shrinks to 3.5 mm during surgery reduces the operative view by 66%. Most cataract surgeons address the problem by stretching the pupil via pharmacological means or viscomydriasis. Because these techniques usually only enlarge the pupil by a few millimeters, surgical maneuvers are often required.
Most of the surgical techniques for enlarging a pupil and preventing its intraoperative constriction are not as safe as ophthalmologists could wish. The maneuvers can increase the risk of a torn iris sphincter, bleeding, iris damage, posterior capsular tears, and loss of the vitreous body. Potential postoperative complications include a poor cosmetic result (ie, atonic pupil of irregular shape) and resultant photophobia. The use of pupil expansion rings rather than surgical enlargement can decrease the risk of these complications. Several such devices are currently on the market; they differ in terms of the ease of intraoperative handling and the learning curve (see Some Devices for Expanding the Pupil).
This article shares my experience with the APX 200 (APX Ophthalmology).
TECHNIQUE
The APX 200 comes in a small cassette with its own forceps that fold the expander and lock it closed until the time of insertion (Figure 1). I make the main and sideport incisions under viscoelastic as usual. I create astigmatism-neutral self-sealing incisions (1-1.2 mm), either with a diamond knife or a microvitreoretinal blade. Because I position the main incision at 12 o’clock, I make the side incisions at 3 and 9 o’clock (Figure 2).
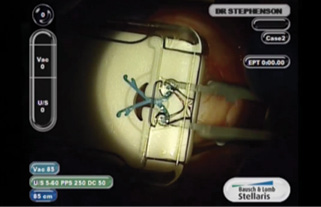
Figure 1. The APX 200 comes in a small cassette with its own forceps that fold the expander and lock it closed until the time of insertion.
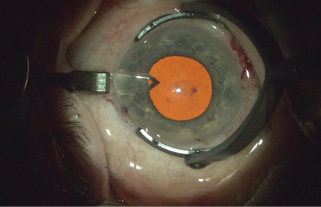
Figure 2. Dr. Stephenson makes the side incisions at 3 and 9 o’clock.
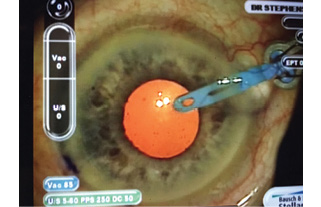
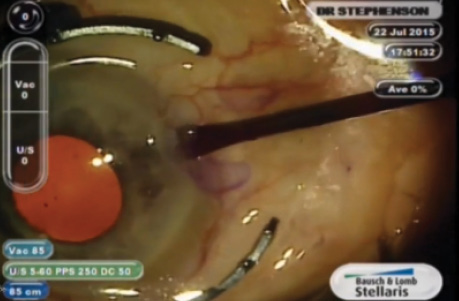
Figure 3. The surgeon inserts the device.
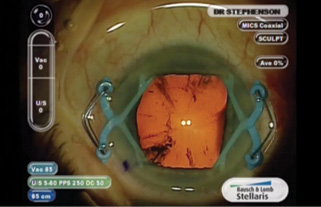
Figure 4. With the APX 200 in place, the pupil assumes a square configuration.
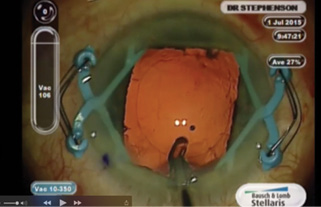
Figure 5. Dr. Stephenson performs phacoemulsification with the device in place.
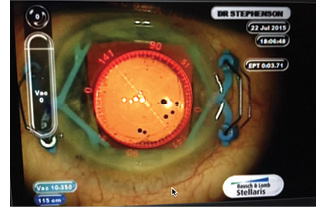
Figure 6. The author has not found the device to disrupt readings with the ORA System with Verifeye+.
After unlocking the forceps, I insert the APX 200 under viscoelastic through a self-sealing incision and gently expand and retract the iris. I repeat this maneuver on the other side (Figure 3). The pupil will now be in the shape of a large square (Figure 4). I can then proceed with the capsulorhexis, hydrodissection and hydrodelineation, and phacoemulsification without having to change where I place the incisions for other instrumentation (Figure 5).
I have not found the device to affect readings with the ORA System with Verifeye+ (Alcon; Figure 6). Nor have I had any problems with laser cataract surgery when the APX 200 has been in place prior to laser treatment.
TIPS
Based on my experience, I can offer three tips for success with the device:
1. When extracting the APX 200, always make sure that the chamber is filled with viscoelastic.
2. Remove the expander either before or after implanting the IOL.
3. Perform mild hydration of the incision to ensure a watertight seal after removal of the device.
CONCLUSION
I have been using the APX 200 for the past year with great success. I find this disposable pupil expander easy to insert and remove without damaging the iris. The learning curve is small.
This device has made cataract surgery safer for my patients with small pupils, and it has increased my sense of comfort and control in these cases.
P. Dee G. Stephenson, MD
• president, American College of Eye Surgeons
• CEO/founder, Stephenson Eye Associates, Venice, Florida • (941) 485-1121; eyedrdee@aol.com; www.stephensoneye.cpm
• financial interest: none acknowledged