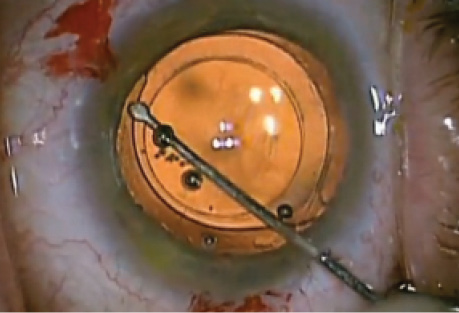
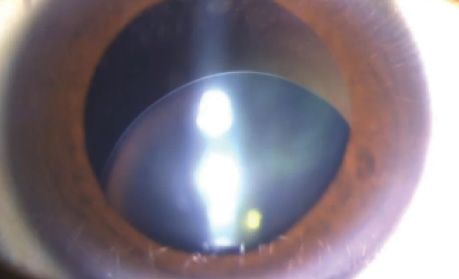
Five circumstances in which this technology is particularly beneficial.
By Samuel Masket, MD

I avoid the emotional confrontations regarding the general use of laser cataract surgery for routine case management. My sense is that good technology will always win in the end, despite the difficulty associated with the business model in this instance.
With respect to the literature, the following comes from a recent and extensive review of the literature on the subject:
“Randomized controlled trials (RCTs) were searched in PubMed, Embase and the Cochrane Central Register of Controlled Trials. Nine qualified studies with a total of 989 eyes were included. Compared with traditional phacoemulsification, FLACS [femtosecond laser-assisted cataract surgery] significantly reduced mean phaco energy and effective phacoemulsification time (EPT) required in the surgery. Central corneal thickness (CCT) was significantly lower in FLACS at 1 day of follow-up, but CCT and corneal endothelial cells count was [sic] comparable at 1 week of follow-up or longer. FLACS achieved a better visual outcome at postoperative 1 week and 6 months, but the difference was not significant at postoperative 1-3 months. Regarding surgical complications, the incidences of intraoperative anterior capsule tear, postoperative macular edema and elevated intraocular pressure were similar. In conclusion, femtosecond laser pretreatment can reduce phaco energy and EPT, which may reduce the heat damage to ocular tissues by ultrasound. This novel technique might be beneficial for patients with dense cataract and/or low preoperative endothelial cell values.”1
Based on personal experience and the evidence-based literature, I have come to recognize five general conditions in which laser cataract surgery is particularly beneficial for my patients:
• shallow anterior chamber
• low endothelial cell count
• dense nuclear cataract
• zonulopathy
• capsule considerations
This article explores these conditions and offers clinical examples, as necessary, for consideration. Any or all of the conditions may coexist.
THE FIRST THREE CONDITIONS
Shallow Anterior Chamber
Watch it Now
Samuel Masket, MD, uses a laser for an IOL exchange in a case of marked contraction of the anterior capsulorhexis.
Eyes with a shallow anterior chamber or crowded anterior segment often have “positive pressure” from behind, making the anterior capsulorhexis challenging, because the capsular tear tends to extend to the periphery. Alternatively, the surgeon may intentionally reduce the size of the capsulotomy, which can lead the anterior capsule to tear out during phacoemulsification. It is also more difficult to ensure that the IOL is fully implanted within the capsular bag if the anterior capsulotomy is small. Moreover, with a shallow chamber, phacoemulsification takes place closer to the endothelium, risking increased endothelial cell loss. With laser prechopping of the nucleus in any format, less ultrasound energy is required to achieve nuclear removal, reducing collateral damage to the endothelium.
Low Endothelial Cell Count
Laser cataract surgery has enabled me to approach patients with Fuchs dystrophy in a different manner than previously. Individuals with definite stromal or epithelial edema clearly require endothelial keratoplasty in advance of or concomitant with cataract surgery. In my experience, however, patients with eyes that have reduced cell counts, of any number, and that are free of edema may undergo laser cataract surgery with careful observation of the cornea postoperatively.
Dense Nuclear Cataract
As noted earlier, laser cataract surgery reduces dispensed phaco energy and effective phaco time. This translates to less corneal damage for a routine case.2 As one might well imagine, the benefit is particularly evident with dense cataracts.3 Other than those rare cataracts that are too dense for penetration by the laser’s imaging system, the technology can facilitate nuclear fragmentation, markedly reducing ultrasound energy.
Case Report
An example that illustrates these three conditions is that of a 78-year-old vascular surgeon with high hyperopia and prior angle-closure glaucoma necessitating a laser iridotomy. The patient was referred to me with a 5+ brunescent cataract, an axial length of 21.43 mm, an aqueous depth of 1.55 mm, and a lens thickness of 4.91 mm; corneal white to white measured 11.3 mm. Central corneal thickness measured 580 µm, and the endothelial cell count was 1,485 cells/mm2.
I used the laser to chop the cataract into sextants and instilled copious amounts of a dispersive ophthalmic viscosurgical device during surgery. The procedure was uncomplicated, with mild central corneal edema noted on day 1. The cornea was clear and had returned to nearly preoperative thickness at the 2-week postoperative visit.
ZONULOPATHY/SUBLUXATED CATARACT
Overview
The capsulorhexis is recognized as the key step in cataract surgery. In general terms, if you can do the capsulorhexis, you can do the case. For eyes with significant zonular weakness, with or without subluxation of the cataract, however, it is often difficult to create an adequately sized and appropriately placed anterior capsulotomy, because the weakened zonular purchase limits propagation of the tear. Moreover, inadvertent additional damage to the zonule can occur. I routinely use laser anterior capsulotomy in eyes with pseudoexfoliation.

Figure 1. The laser platform’s optical coherence tomography system reveals subluxation and modest tilt of the cataract.
When dealing with cataracts in eyes that have weak or absent zonular fibers, it is necessary to produce a capsular opening that allows the use of capsular hooks and that permits the placement of capsular tension rings of all designs. In this setting, I find that an automated capsulotomy saves the day.
Case Report
A 70-year-old man had sustained a tennis ball injury to his left eye some 20 years earlier. The patient had tolerated a progressive diminution of vision until recently. He was referred to me with a dense nuclear cataract and 4 to 5 clock hours of zonulysis on the nasal side. Traditional cataract surgery would have been challenging with respect to the capsulotomy and nuclear removal. Laser cataract surgery (for the capsulotomy and nuclear disassembly) combined with capsular support hooks and a sutured Cionni Ring for Scleral Fixation (Morcher; distributed in the United States by FCI Ophthalmics) allowed for uncomplicated surgery with a routine outcome, an in-the-bag toric IOL, and a postoperative distance UCVA of 20/25 (Figures 1-3). Although some surgeons are concerned that a laser-generated anterior capsulotomy may not be strong enough to support capsule hooks, my experience shows otherwise.
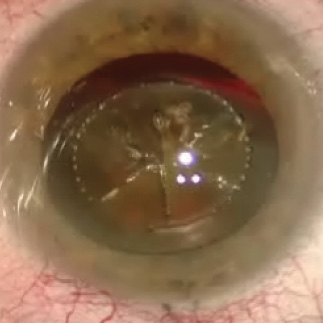
Figure 2. Operative view at the outset of surgery after laser anterior capsulotomy and sextant nuclear division. Note 4 to 5 clock hours of nasal zonulopathy and the dense cataract.
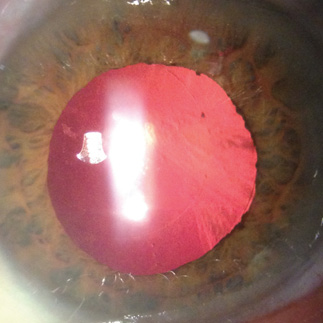
Figure 3. One postoperative examination at the slit lamp demonstrates a perfectly centered capsular bag-capsular tension ring-IOL complex.
CAPSULE CONSIDERATIONS
Overview
Uncommon conditions of the anterior or posterior capsule may pose a threat at the time of cataract surgery. Anterior and posterior lenticonus may affect tensile strength of the capsule, and it is generally held that a posterior polar cataract is associated with approximately a 25% risk of posterior capsular rupture (Figure 4). With anterior lenticonus, I find that a laser anterior capsulotomy is more likely successful than routine mechanical methods. Regarding a posterior polar cataract, having a perfectly sized and placed anterior capsulotomy may be helpful if there is a defect in the posterior capsule that makes bag fixation untenable. Given a reliable laser anterior capsulotomy in that situation, I can opt for optic capture or reverse optic capture of the IOL to provide stable fixation long term. Although I strongly prefer to use the laser to create the anterior capsulotomy in eyes with a posterior polar cataract, I avoid laser nuclear division, because it risks damage to the posterior capsule due to increased pressure in the capsular bag from gas production.
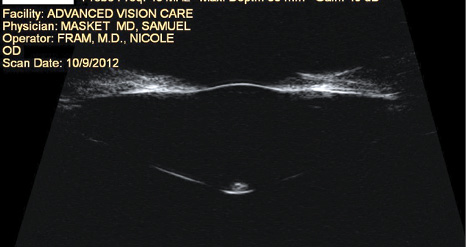
Figure 4. Ultrasound biomicroscopy demonstrates anterior and posterior lenticonus with a posterior polar cataract. Use of a laser resulted in uncomplicated cataract surgery.
Another application of the laser can be to facilitate IOL exchange in cases with late anterior capsular contraction through the creation of a secondary anterior capsulotomy.
Case Report
A 78-year-old-woman underwent bilateral cataract surgery with placement of multifocal IOLs 3 years ago. Unfortunately, ocular comorbidities, including age-related macular degeneration, epithelial basement membrane dystrophy, and astigmatism, resulted in a poor quality of vision. Marked contraction of the anterior capsulorhexis served as a significant deterrent to IOL exchange with preservation of the capsular bag (Figure 5). I used the laser to recut the anterior capsulotomy and successfully exchanged the IOL for a monofocal lens (Figure 6). The patient achieved a marked improvement in BCVA as well as a greater overall quality of vision.
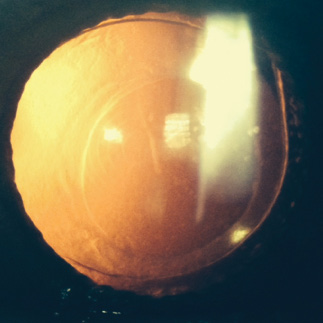
Figure 5. Anterior capsular contraction is evident.
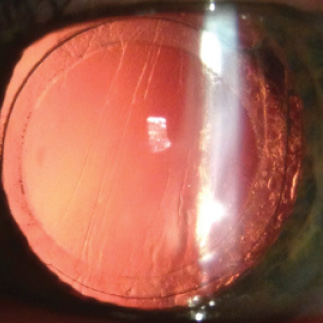
Figure 6. Six months after IOL exchange, the monofocal lens is well centered behind a laser-generated secondary anterior capsulotomy.
CONCLUSION
Although some surgeons question the need for and value of laser cataract surgery for the management of routine cases, my experience has demonstrated benefits of the technology in certain circumstances.
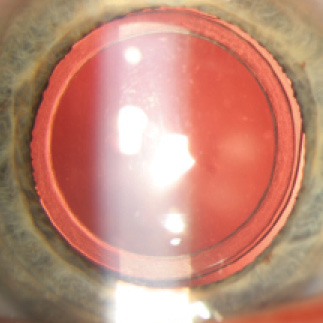
Figure 7. Postoperative view of the Morcher 90S IOL (Masket IOL). Note the peripheral groove in the optic to accept the anterior capsulotomy and the excellent centration of the lens.
Interest is growing in IOLs that are captured by the anterior capsulotomy. Potential advantages include a reduction of negative dysphotopsia, excellent IOL centration, an elimination of lens tilt, and stable meridional placement of toric IOLs. When the laser can allow the capsulotomy to be centered on the patient’s visual axis, higher-order optical aberrations can be eliminated. To me, it is obvious that, in order to use the anterior capsule to capture the IOL, new lens designs are required. One such device is the Morcher 90S IOL (Masket IOL) designed to eliminate negative dysphotopsia (Figure 7).
1. Chen X, Xiao W, Ye S, et al. Efficacy and safety of femtosecond laser-assisted cataract surgery versus conventional phacoemulsification for cataract: a meta-analysis of randomized controlled trials [published online ahead of print August 13, 2015]. Sci Rep. doi:10.1038/srep13123.
2. Takács AI, Kovács I, Miháltz K, et al. Central corneal volume and endothelial cell count following femtosecond laser-assisted refractive cataract surgery compared to conventional phacoemulsification. J Refract Surg. 2012;28(6):387-391.
3. Hatch KM, Schultz T, Talamo JH, Dick HB. Femtosecond laser-assisted compared with standard cataract surgery for removal of advanced cataracts. J Cataract Refract Surg. 2015;41(9):1833-1838.
Samuel Masket, MD
• Advanced Vision Care, Los Angeles
• clinical professor of ophthalmology, Geffen School of Medicine, UCLA, Los Angeles
• financial disclosure: consultant to Alcon and holds a financial interest in the Morcher 90S (Masket) IOL
The technology creates more barriers than benefits in these cases.
By Richard Schulze J, MPhil (Oxon), MD, and Steven G. Safran, MD


Many possible anatomic variables can make cataract surgery more complex, and most of these require surgeons to deviate from their normal routine. The problems may relate to extraocular anatomic issues such as a very deep-set eye, a small palpebral aperture, a prominent brow, or a patient’s postural problems that render proper positioning or cooperation more difficult. Adding a femtosecond laser to the equation will further complicate most of these issues. Docking and treatment with the laser are more problematic in patients with these anatomic constraints, which may lead to suction breaks and incomplete treatment, not to mention the time and energy spent on steps that are unnecessary if the surgeon has the skills to perform these steps manually.
Other cases are complex because of intraocular anatomic variables that require special attention and care. They include nondilating or bound-down pupils, zonular instability, dislocated lenses, opaque cataracts that may have coexisting capsular compromise, and corneal opacity or scars. In these situations, the use of a laser may create more barriers than benefits.
CHALLENGING PUPILS
Cataract surgeons overcome the problem of a nondilating pupil relatively easily, not infrequently by using modern devices for pupillary expansion such as iris retractors or the Malyugin Ring (MicroSurgical Technology). In laser cataract surgery, however, not only does the use of these devices become more problematic, but the need for them may be more prevalent. The laser causes significant miosis on its own due to prostaglandin release.1 If miosis is already present, it must be addressed before laser treatment of the capsule or the lens if the surgeon wishes to make an optimally sized capsulotomy. This may require shuttling the patient back and forth between the laser suite and the OR so that a pupillary expansion ring can be placed prior to treatment.
In an eye with posterior synechiae and a bound-down pupil or in the presence of an iridodialysis, iris retractors may be a better choice than a Malyugin Ring, but it may be impossible to use the laser with these retractors sticking out of the paracenteses. The laser may thus compromise a surgeon’s ability to deal with the pathology at hand.
ZONULAR INSTABILITY
While manually creating a capsulorhexis, tactile feedback gives the surgeon insight into the status of the zonules and the capsular bag, which can help him or her better plan the rest of the procedure. Capsular support devices can be incorporated earlier, which can spare remaining zonules and help to avoid complications that may arise later in the case. Such feedback and insight are lost with a laser capsulotomy. Instead, the surgeon may first become aware of zonular or capsular compromise during phacoemulsification, when it may be too late to deal with the problem effectively in a preemptive manner. In cases of profound zonular loss such as an eye with microspherophakia (Figure 1), the lens may be too decentered to permit the creation of a properly centered laser capsulotomy. The surgeon has no choice in such a case but to create the capsulorhexis manually or to rely on a decentered or overly small laser capsulotomy.
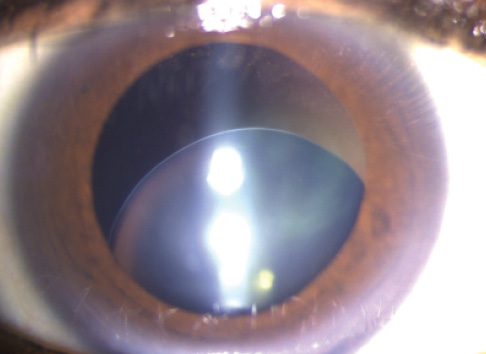
Figure 1. Microspherophakia.
Scanning electron microscopy has demonstrated the histologic imperfections of a laser capsulotomy compared with a manual capsulorhexis, and studies have shown that this jagged edge increases the risk of radial tears during surgical manipulation.2 In an eye with compromised zonules, capsule retractors and sutured capsular tension segments are more likely to be required, placing stress on the anterior capsule; the strongest possible capsulotomy for manipulation is thus the most desirable. A radial tear in this type of case could be disastrous, because it will render the use of a capsular tension ring and the placement of the IOL in the bag virtually impossible. For this reason, many surgeons who routinely operate on patients with ectopia lentis (Figure 2) and profound zonular instability prefer the elasticity and strength of a properly performed manual capsulorhexis. Obviously, reshaping or retearing the capsulorhexis to fit a unique situation at the end of the case can only be done by hand.
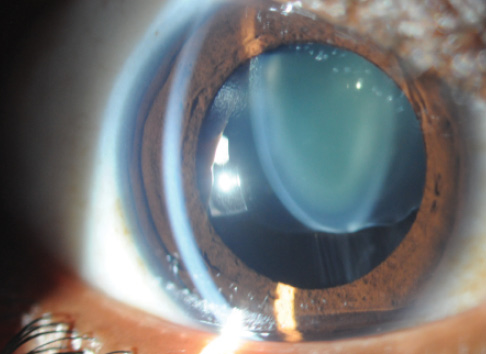
Figure 2. Ectopia lentis.
Laser prechopping of the nucleus into small pieces can also be problematic in eyes with zonular deficiency. Small cubes can easily migrate through zonular defects into the posterior segment or be inadvertently left behind, especially if the pupil is small and hides them. The concern is greater with the laser, which, again, can cause miosis.1,3
OPAQUE CATARACTS
The laser cannot cut through what it cannot see. Both the densest white and black cataracts are either impossible to fragment with laser energy, or they will be incompletely treated, leaving a dense posterior plate that may box in the surgeon. Thus, the cataracts that would benefit the most from presoftening are exactly those in which it cannot be reliably achieved.
ADDITIONAL CONCERNS
Problems using the laser in cases of polar cataract have been reported,4 with capsular blowout during phacofragmentation cited as a potential risk. Likewise, when there may be occult posterior capsular damage from plaques or trauma caused by vitreoretinal work, the surgeon has to be concerned about the potential for problems when relying on laser fragmentation. Increased pressure due to gas buildup within the capsular bag should be avoided for the same reasons surgeons do not perform hydrodissection in these cases, and there is no reason to add steps that could cause an uncontrolled increase in pressure within the capsular bag if it can be avoided.
Watch it Now
Steven G. Safran, MD, performs microspherophakia surgery with two sutured Ahmed Capsular Tension Segments for support and placement of a toric IOL.
In patients who have a history of silicone oil placement—even after its removal—the potential blockage of laser energy may lead to an incomplete capsulotomy.5 Likewise, because the laser cannot cut what it cannot see, the laser capsulotomy may be incomplete in eyes with corneal opacities and scarring. This may be of particular concern for patients who have a history of radial keratotomy or other corneal refractive surgery in the zone where the laser will be used.
Complex cataract cases often require surgeons to make additional small paracentesis incisions on the fly for iris or capsule retractors or for the passage of microinstrumentation. A blade is much more flexible and efficient for this purpose than having to plan and execute these steps with a laser. According to the FLACS ESCRS Study,6 more than two-thirds of surgeons performing laser cataract surgery do not use the laser to create incisions, even in routine cases. The technology clearly becomes a burden as the case’s complexity and the need for flexibility increase.
Finally, the use of the laser causes the release of prostaglandins,7 cytokines, chemokines, and other inflammatory mediators8 that may increase the amount of postoperative fibrosis and inflammation—exactly what a surgeon does not want in a complex case.
CONCLUSION
Complex cataract procedures require surgeons to deviate from their normal routine and demand a great deal of planning. The last thing the ophthalmologist needs is to be saddled with unnecessary steps that must be worked around unless they are essential to providing better and safer outcomes. Adding a laser is like taking extra luggage that will not be used on a trip. Why not leave it behind and have less to worry about?
1. Schultz T, Joachim SC, Stellbogen M, Dick HB. Prostaglandin release during femtosecond laser-assisted cataract surgery: main inducer. J Refract Surg. 2015;31(2):78-81.
2. Abell RG, Davies PE, Phelan D, et al. Anterior capsulotomy integrity after femtosecond laser-assisted cataract surgery. Ophthalmology. 2014;121(1):17-24.
3. Diakonis VF, Yesilirmak N, Sayed-Ahmed IO, et al. Effects of femtosecond laser-assisted cataract pretreatment on pupil diameter: a comparison between three laser platforms. J Refract Surg. 2016;32(2):84-88.
4. Alder BD, Donaldson KE. Comparison of 2 techniques for managing posterior polar cataracts: traditional phacoemulsification versus femtosecond laser-assisted cataract surgery. J Cataract Refract Surg. 2014;40(12):2148-2151.
5. Grewal DS, Singh Grewal SP, Basti S. Incomplete femtosecond laser-assisted capsulotomy and lens fragmentation due to emulsified silicone oil in the anterior chamber. J Cataract Refract Surg. 2014;40(12):2143-2147.
6. Barry P. FLACS ESCRS Study. Paper presented at: XXXIII Congress of the ESCRS; September 7, 2015; Barcelona, Spain.
7. Schultz T, Joachim SC, Kuehn M, Dick HB. Changes in prostaglandin levels in patients undergoing femtosecond laser-assisted cataract surgery. J Refract Surg. 2013;29(11):742-747.
8. Wang L, Zhang Z, Koch DD, et al. Anterior chamber interleukin 1B, interleukin 6 and prostaglandin E2 in patients undergoing femtosecond laser-assisted cataract surgery [published online ahead of print December 11, 2015]. Br J Ophthalmol. doi:10.1136/bjophthalmol-2015-307586.
Steven G. Safran, MD
• private practice in Lawrenceville, New Jersey
• (609) 896-3931; safran12@comcast.net
• financial disclosure: consultant to Bausch + Lomb and Topcon
Richard Schulze Jr, M Phil (Oxon), MD
• private practice, Schulze Eye & Surgery Center, Savannah, Georgia
• (912) 352-3120; richardschulze@comcast.net; Twitter @rschulzejr
• financial interest: none acknowledged