CASE PRESENTATION
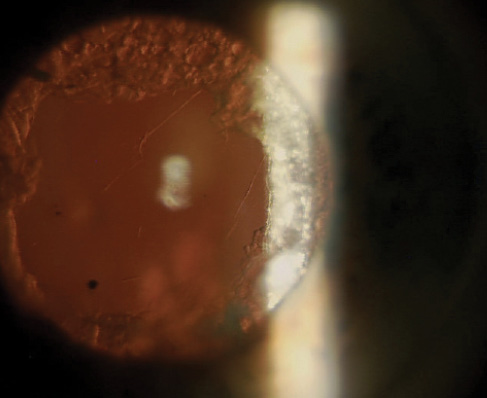
Figure 1. The eye showed marked phimosis 6 weeks after cataract surgery.

ROBERT J. CIONNI, MD
The situation represented by this case is not uncommon. Unfortunately, a CTR does not necessarily prevent any chance of phimosis,1 which speaks to the importance of intense polishing under the residual anterior capsular rim at the time of the initial surgery in cases of PXF. I use a modified Singer Sweep (Epsilon) to aggressively clean the rim for 360º.
In this case, the surgeon was correct to recheck for phimosis shortly after the procedure, but the complication will likely progress, which will result in further zonular compromise and IOL decentration if simply observed. Performing a four-cut anterior capsule YAG laser capsulotomy will likely prevent further phimosis and enlarge the central opening. It would be my first step in managing this case.
If significant pseudophacodonesis is present and/or significant IOL decentration occurs, I would perform suture fixation to the scleral wall. A double-armed Gore-Tex suture (off-label use; W.L. Gore & Associates) or a 9–0 Prolene suture (Ethicon) could be passed through the periphery of the capsular bag to capture the CTR and suture it to the scleral wall via a Hoffman pocket.2 A single fixation superiorly is likely all that would be needed, but if the IOL decentered superiorly during this maneuver, I would also fixate the CTR in a similar fashion inferiorly. As evidenced by the phimosis, there should be sufficient peripheral fibrosis to ensure that the capsular bag will not rupture during or after this procedure.

ALAN S. CRANDALL, MD
This presentation is unusual only in the timing of the phimosis of the capsule. My colleagues and I have reported late in-the-bag IOL subluxation,3,4 which occurs about 8.5 years postoperatively on average. The increase in the subluxation appears to relate to the use of the CCC and is seen frequently in eyes with PXF syndrome. It appears to be associated with zonular weakness or stress during surgery and especially capsular fibrosis.1 The use of a CTR helps to center the bag complex but does not prevent subluxation or capsular phimosis.1
In this case, I would not be comfortable waiting for the subluxation. I would enlarge the capsular opening. One could use a YAG laser to cut the anterior capsule (usually four openings), releasing the zonular stress. Samuel Masket, MD, has told me via email that a femtosecond laser can be used to increase the capsulotomy’s diameter. In an eye with pseudophacodonesis, however, both procedures would be very tricky, and because the bag is already moving, I would take the patient to the OR and manually open the capsule; I would use scissors to initiate a tear and bimanually enlarge the capsulotomy’s diameter. I would use an ab externo approach to scleral fixate the complex.
In the patient’s second eye, I would use a zonule-friendly technique just as in the first eye, which would include a CTR. I would preemptively select a Malyugin Ring (MicroSurgical Technology) or a Cionni Ring for Scleral Fixation or an Ahmed Capsular Tension Segment (both from Morcher and distributed in the United States by FCI Ophthalmics).5-8

RICHARD MACKOOL Jr, MD
For this patient with severe capsular contracture, it would be necessary to stabilize the operated eye before proceeding to the fellow eye. An anterior YAG capsulotomy (and posterior capsulotomy if required) should be sufficient to clear the visual axis and relieve zonular tension. When performing an anterior capsulotomy, surgeons should monitor the IOP after laser treatment, because transient IOP spikes can occur. It is also advisable for the patient to use a topical steroid for the first postoperative week.
For the second eye, I would perform phacoemulsification with removal of the lens epithelium. Polishing the undersurface of the anterior capsule for 360º with a 90º I/A tip and polishing the posterior capsule with a cannula while injecting viscoelastic would minimize the residual cellular material responsible for capsular phimosis. A CTR and hydrophobic acrylic IOL could then be inserted. A postoperative steroid for 6 weeks is my usual regimen. If the second eye developed phimosis, I would laser the capsule.

ANITA NEVYAS-WALLACE, MD
The progressive capsular phimosis of the first eye requires treatment by multiple short, radial, YAG laser incisions at the edge of the capsulorhexis.
Protecting the zonule during surgery on the second eye will be key. I would stabilize the bag intraoperatively using capsule retractors (MicroSurgical Technology) and size the capsulorhexis to permit capture of the lens implant’s optic.
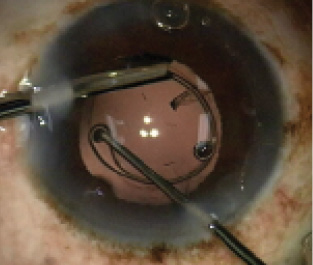
Figure 2. Guiding the leading haptic of a CTR prevents it from disinserting the zonule. (Reprinted with permission from Wang MX. Refractive Lens Exchange: a Surgical Treatment for Presbyopia. Thorofare, NJ: Slack; 2015.)
After hydrodissection, I would attempt slight rotation of the lens nucleus. Any “snap back” of the nucleus to its original position suggests that attempted rotation is stressing the zonule. Safe options for mobilizing the nucleus at that point include repeated hydrodissection and early cortical aspiration (by the bevel-down phaco tip). Removing cortex creates space for further hydrodissection and allows zonule-friendly mobilization of the nucleus for chopping.
After vertical chop and during removal of the nuclear segments, I would frequently re-inflate the bag with viscoelastic in order to keep a billowing posterior capsule from being aspirated into the phaco tip. If, after nuclear removal, the bag were only mildly mobile, then an anterior chamber maintainer might keep back the capsule and facilitate cortical removal by repeated hydrodissection. If the bag were more than mildly mobile, cortical removal by repeated viscodissection would be a safer choice. Directly peeling cortex from the capsule stresses the zonule unnecessarily and should be avoided in this setting.
Leaving the capsular bag as free of cortical material as possible may reduce the chance of capsular phimosis and irregular contracture. I find irrigating into the equator of the bag to be a helpful technique for finding and liberating hidden cortical material. Strands and sheets of cortex adherent to the capsule may also be dislodged by hydrodissection. This is a preferable strategy to vacuuming or polishing the capsule, both as far as the former’s avoidance of zonular traction and the reduced risk of tearing the capsule. In my experience, the irrigating stream from the 27-gauge sandblasted Nevyas-Wallace Cortex Liberation cannula (Katena) liberates cortex more safely than does pulling the material from the capsule.
Although the first eye has demonstrated that a CTR cannot be counted upon to prevent phimosis, placing such a device still might help to stabilize the bag. Should either eye’s bag-IOL complex someday dislocate, the CTR might facilitate suture fixation. To insert a CTR without stressing the zonule, I would use a second instrument to guide the ring’s leading haptic so that it does not contact the bag’s equator until most of the ring has been introduced (Figure 2). The CTR will most easily be flexed if its injector does not extend far into the anterior chamber. I would take care to avoid placing the CTR through the loop of a capsule retractor.
In my experience, placing a three-piece IOL in the ciliary sulcus and capturing the optic posteriorly through the capsulorhexis serve to protect the iris from chafing by the optic, help to prevent capsular phimosis, and improve the chances of long-term stability. I would use an aberration-neutral aspheric, three-piece, monofocal IOL to minimize sensitivity to decentration such as the LI61AO (Bausch + Lomb), AQ2010V (STAAR Surgical), or Sensar AR40 (Abbott Medical Optics).
After removing the capsule support hooks, I would implant the IOL in the ciliary sulcus and then capture the optic posteriorly into the capsular bag. After viscoelastic removal, I would instill triamcinolone into the anterior chamber to verify that no vitreous was present.

ROBERT J. WEINSTOCK, MD
The operated eye shows marked anterior capsular phimosis relatively early in the postoperative period. If it is visually significant, I would consider intervening with either a YAG or a femtosecond laser. The least invasive method would be to use a YAG laser to treat the anterior capsule and attempt to enlarge the anterior capsular opening. Multiple shots with low energy settings usually do the job. Alternatively, the femtosecond laser could be used to make a secondary, larger capsulotomy outside the margin of the current capsular opening. Although I have yet to try this technique, it has been reported to be a successful management option but does require surgical intervention.9
For the fellow eye, I would make a 7- to 8-mm capsulotomy with a femtosecond laser, which would reduce the mechanical stress on the zonules that usually occurs with a manual capsulorhexis in PXF patients. I would perform thorough hydrodissection and hydrodelineation, followed by gentle nuclear and cortical removal. I would also polish the underside of the anterior capsule to remove all lens epithelial cells. I would place a CTR and a three-piece IOL such as the LI61AO. In my experience, this lens is very resistant to decentration and is not as prone to capsular fibrosis as single-piece hydrophobic acrylic lenses.

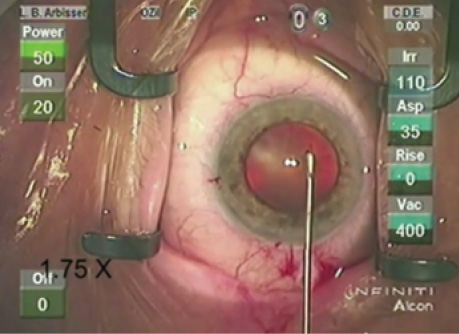
WHAT I DID: LISA BROTHERS ARBISSER, MD
The patient’s first eye required YAG laser anterior capsular relaxing incisions. I used a contact goniolens and low power to make small cuts 180º apart in six pie-shaped wedges. The edges were under significant tension and were conservatively placed so as not to extend too far peripherally (Figure 3A). I prescribed an extra topical steroid and nonsteroidal anti-inflammatory drug.
Watch it Now
Surgery on the patient’s second eye was uncomplicated, but Lisa Brothers Arbisser, MD, later recognized that one modification to her technique was warranted.
Two weeks later, I was surprised to see there was already evidence of fibrotic healing of the YAG incisions, which I therefore further enlarged with a second sitting at the laser. (Figure 3B).
Two months postoperatively, the eye was quiet and stable with a visual acuity of 20/20 and mild pseudophacodonesis. The patient understood the low risk of future bag-lens dislocation, and we agreed to proceed with cataract surgery on the fellow eye.
In my experience, one thing bag-lens dislocations have in common is an intact CCC and usually phimosis. I therefore decided I needed a stent to stabilize the fellow eye. In addition to placing a CTR, I planned to implant a three-piece IOL with the haptics in the sulcus and to capture the optic through the CCC into the bag.
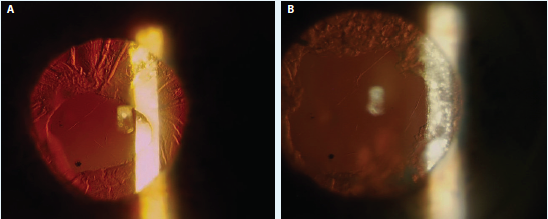
Figure 3. Dr. Arbisser performed YAG laser anterior capsular relaxing incisions (A) and then enlarged them at the laser 2 weeks later to address fibrotic healing (B).
The zonules in this eye proved to be at least as lax as in the first eye. The case was uncomplicated. With a hovering caliper, I carefully kept the diameter of the CCC just under 5 mm to facilitate capture. Surgery went as planned, but I subsequently realized one modification to my technique was needed. One should remove the ophthalmic viscosurgical device (OVD) from the bag after placing the CTR but before implanting the lens so as not to entrap much OVD in the optic-sealed bag. The patient in this case did not experience a capsule expansion syndrome, but this could be a risk if too much OVD were left behind the optic. The OVD can imbibe fluid, necessitating release via an early posterior YAG capsulotomy to restore normal lens position and refraction. This potential complication is therefore best avoided.
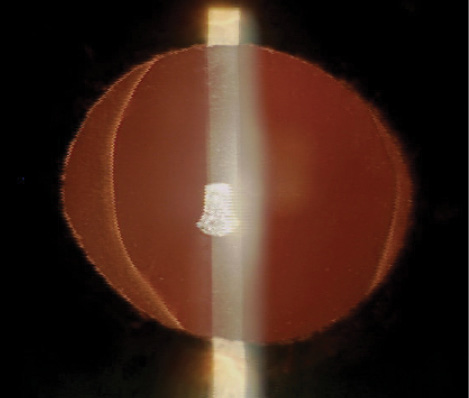
Figure 4. Dr. Arbisser performed cataract surgery on the patient’s second eye with optic capture of the IOL.
On day 1 and at all subsequent visits, the patient’s visual acuity was 20/20, the IOP was normal, and the lens was stable with no pseudophacodonesis and no phimosis (Figure 4).
My hope is that this implantation method will prevent late dislocation, because the bag supports the lens, the lens haptics in the sulcus support the bag, the CTR expands the bag, and most importantly, the optic stents the CCC, thus preventing progressive phimosis. The stability of the complex in this case convinced me to use optic capture for all patients with PXF who demonstrate marked zonular laxity at surgery. I have only 7 years of follow-up (most dislocations happen 8-10 years postoperatively3), however, and do not have the capacity to collect enough data to know if this approach could solve the growing problem.
1. Werner L, Zaugg B, Neuhann T, et al. In-the-bag capsular tension ring and intraocular lens subluxation or dislocation: a series of 23 cases. Ophthalmology. 2012;119(2):266-271.
2. Hoffman RS, Fine IH, Packer M. Scleral fixation without conjunctival dissection. J Cataract Refract Surg. 2006;32(11):1907-1912.
3. Davis D, Brubaker J, Espandar L, et al. Late in-the-bag spontaneous intraocular lens dislocation; evaluation of 86 consecutive cases. Ophthalmology. 2009;116:664-670.
4. Jehan FS, Mamalis N, Crandall AS. Spontaneous late dislocation of intraocular lens within the capsular bag in pseudoexfoliation patients. Ophthalmology. 2001;108:1727-1731.
5. Slade DS, Hater MA, Cionni RJ, Crandall AS. Ab externo scleral fixation of intraocular lens. J Cataract Refract Surg. 2012;38(8):1316-1321.
6. Ahmed II, Crandall AS. Ab externo scleral fixation of the Cionni modified capsular tension ring. J Cataract Refract Surg. 2001;27(7):977-981.
7. Shingleton BJ, Crandall AS, Ahmed II. Pseudoexfoliation and the cataract surgeon: preoperative, intraoperative, and postoperative issues related to intraocular pressure, cataract, and intraocular lenses. J Cataract Refract Surg. 2009;35(6):1101-1120.
8. Kirk TQ, Condon GP. Simplified ab externo scleral fixation for late in-the-bag intraocular lens dislocation. J Cataract Refract Surg. 2012;38(10):1711-1715. Erratum in: J Cataract Refract Surg. 2013;39(3):489.
9. Wiley WF, . Optical coherence tomography-guided capsule bag-centered femtosecond laser capsule Paper presented at: ASCRS/ASOA Congress & Symposium; April 9-14, 2013; San Francisco, CA.
Section Editor Lisa Brothers Arbisser, MD
• emeritus position at Eye Surgeons Associates, the Iowa and Illinois Quad Cities
• adjunct associate professor, John A. Moran Eye Center, University of Utah, Salt Lake City
• (563) 343-8896; drlisa@arbisser.com
• financial interest: none acknowledged
Section Editor Brandon D. Ayres, MD
• surgeon in the Cornea Service, Wills Eye Hospital, Philadelphia
Section Editor Audrey R. Talley Rostov, MD
• private practice with Northwest Eye Surgeons, Seattle
• medical advisory board, SightLife, Seattle
Robert J. Cionni, MD
• medical director, The Eye Institute of Utah, Salt Lake City
• adjunct clinical professor, John A. Moran Eye Center, University of Utah, Salt Lake City
• (801) 266-2283
• financial disclosure: receives product royalty from Morcher for the Cionni Ring for Scleral Fixation
Alan S. Crandall, MD
• John A. Moran presidential professor; Val A. and Edith D. Green presidential endowed chair; senior vice chair; director of glaucoma and cataract; and codirector, Moran Outreach Division, John A. Moran Eye Center, University of Utah, Salt Lake City
• (801) 585-3071; alan.crandall@hsc.utah.edu
• financial interest: none acknowledged
Richard Mackool Jr, MD
• assistant director, The Mackool Eye Institute and Laser Center, Astoria, New York
• (718) 728-3400
Anita Nevyas-Wallace, MD
• private practice, Nevyas Eye Associates, Bala Cynwyd, Pennsylvania
• clinical assistant, University of Pennsylvania Department of Ophthalmology, Philadelphia
• (610) 668-2777; anw@nevyas.com
• financial interest: none acknowledged
Robert J. Weinstock, MD
• director of cataract and refractive surgery, Eye Institute of West Florida, Largo, Florida
• rjweinstock@yahoo.com; Twitter @EyeInstituteWFl
• financial disclosure: consultant to Bausch + Lomb