Working in a tertiary referral center, I am used to operating on patients I have not seen before surgery, as their presurgical workup is routinely done by another doctor on our team. In retrospect, then, it should have raised a red flag in my mind when one of my partners warned me, “Today you are going to have a challenging cataract surgery because of a cloudy cornea.” Instead, I naïvely thought, “How cloudy can a cornea get?”
When it was this patient’s turn in the OR, I confidently greeted him on the operating table and then glanced at his electronic health record. By the time I realized that this 82-year-old gentleman had no light perception in his contralateral eye after a severe stroke, he was already under general anesthesia because of several comorbidities.
Despite years of practice, doing surgery on a patient’s only seeing eye still feels different to me. As a sidenote, optical biometry had not been possible on the patient’s operative eye, so we had to rely on acoustic axial length measurement and manual keratometry. Retinal detachment was ruled out by B-scan ultrasound.
ON TO SURGERY
After surgical draping and insertion of the speculum, I looked through the microscope. My heart sank when I saw dense corneal opacities caused by previous penetrating keratoplasty with subepithelial scarring (Figure 1). I could not positively identify the pupil, nor the lens.
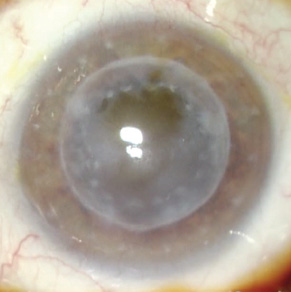
Figure 1. Surgical view of the cloudy cornea under standard diffuse microscope light.
There was no way I could operate under these conditions. I took a deep breath and was already thinking of how I was going to explain to the patient what happened (nothing at all) when inspiration hit me: What if I changed the microscope illumination? Indeed, after adjusting to a horizontal slit instead of the standard diffuse illumination, there was some red reflex (Figure 2). I was saved.
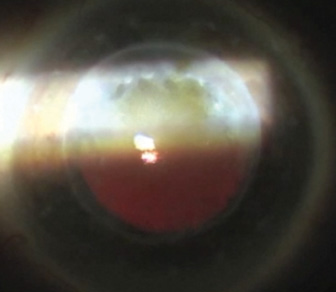
Figure 2. Slit-illumination on the surgical microscope reveals some red reflex, allowing cataract surgery to proceed.
I carefully constructed a paracentesis and a 2.5-mm sclerocorneal tunnel incision. Deliberate rotation of the globe throughout the procedure would facilitate obtaining and maintaining the red reflex. I injected some OVD as usual. Because the cataract was brunescent, I painted the anterior lens capsule with trypan blue dye by injecting it underneath the OVD. This is an excellent alternative to trypan blue injection under air followed by OVD exchange, as used by many surgeons. A video of the procedure can be watched below.
After refilling the anterior chamber with OVD, I used Utrata forceps to make a continuous curvilinear capsulorhexis. After that, I performed careful hydrodissection in multiple quadrants and made sure the lens was rotating freely.
I inserted the phaco handpiece and debulked the central lens using ultrasound energy. With the aid of a chopper, I then cracked the nucleus and removed the first quadrant of the rock-hard lens. Next, I elevated the lens material into the anterior chamber to avoid damaging the posterior capsule (as it was still only barely visible). Of course this put more stress on the corneal endothelium, but I wanted to avoid at all cost a capsular defect, as that could potentially lead to lens material dropping into the vitreous cavity. In this case, cataract surgery alone was challenge enough for me.
As soon as all hard nuclear and cortical material was removed, and after replenishing the OVD, I inserted the IOL. I then continued to remove soft outer cortex from in front of and behind the IOL using the I/A handpiece. Finally, I removed the OVD and injected cefuroxime intracamerally.
A SLOW RECOVERY
The case described here is very recent. Postoperative recovery for this patient was much slower than normal, as would be expected, and plenty of osmotic agents and dexamethasone eye drops were prescribed. The patient is still recovering, but visual acuity will remain poor. Repeat keratoplasty in this patient’s only seeing eye may yet become unavoidable in the future.