We have so many choices in cataract surgery today, and every surgeon has preferences. For me, that means a mix of niche and workhorse IOLs. My workhorse is the AcrySof IQ platform. The science and design behind these monofocal, toric, and multifocal lenses help deliver exceptional outcomes for my patients every day, and they allow me to perform smooth, elegant, and highly predictable procedures.
Aspheric Monofocal Lens
The AcrySof IQ aspheric monofocal lens (Alcon) showed improved functional vision versus a spherical monofocal IOL,1 in large part due to its unique BioMaterial, BioMechanics and BioOptics. Its asphericity is designed to deliver the same total spherical aberration as found in the youthful eye, which helps give patients good contrast sensitivity.2 The AcrySof IQ design and the blue light chromophore has also been reported to help reduce glare during night-time driving.3-5
The crystalline lens increases in sphericity as the eye ages. The spherical aberration of the AcrySof IQ lens combined with the aberration of the cornea returns the eye to its youthful aberration state. The goal for optimal vision is for the total spherical aberrations to be slightly positive at 0.1 μm.2,6
The AcrySof IQ IOL has a haptic design that enables conformation to the shape of the capsular bag with consistent compression force. The results are precise centration, predictable refraction, and stable positioning over the long term.
I consider the AcrySof IQ monofocal the top-of-the-line option in its category, and it is my standard lens of choice. I want a lens that is consistently straightforward to implant in the capsular bag, centers well, and delivers the quality of vision that patients desire. That is exactly what the AcrySof IQ lens does. It makes my patients happy. I’ve used the lens on photographers, painters, and designers, all of whom require a high degree of color appreciation, precision, and sharpness, and I have received positive reviews from these patients.
Toric and ReSTOR Multifocal Lenses
With the same BioMaterial, BioMechanics, and BioOptics benefits as the Acrysof IQ monofocal lens, two other excellent lenses in this line are the AcrySof IQ toric IOL (Alcon) and AcrySof IQ ReSTOR multifocals (Alcon).
For patients with astigmatism who want to rely less on glasses for their distance vision, I choose the AcrySof IQ toric lens above other torics because of its stability. Like the AcrySof IQ monofocal, the toric lens does not rotate. It remains in its optimal position long term (Figure 1). The AcrySof BioMaterial, a hydrophobic acrylic polymer that is highly biocompatible, offers greater fibronectin bioadhesion than other monofocal lens materials in vitro (Figure 2).7 Fibronectin bioadhesion, combined with other key design features, contributes to the excellent adhesion of this IOL to the capsular bag.
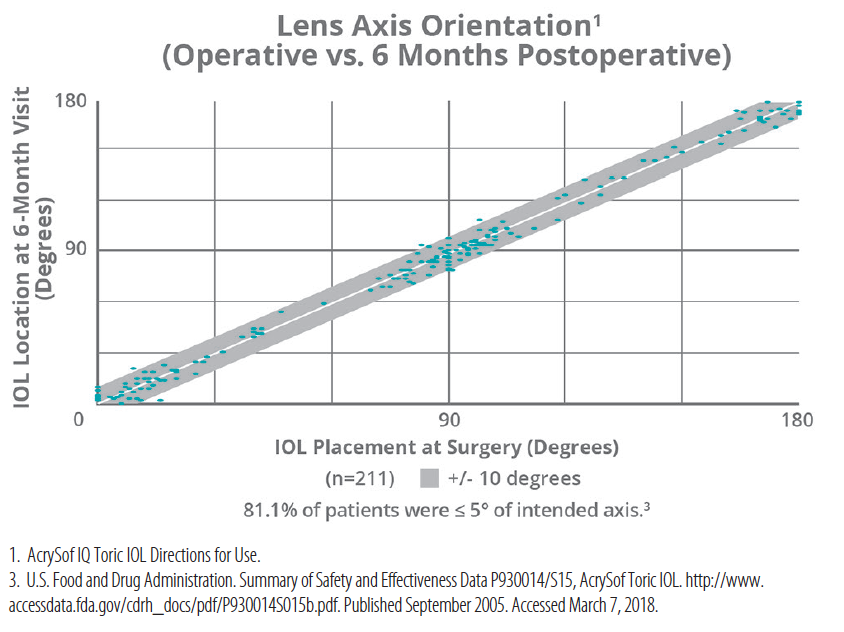
Figure 1. Mean lens axis orientation from operative visit to 120 to 180 days postoperatively.
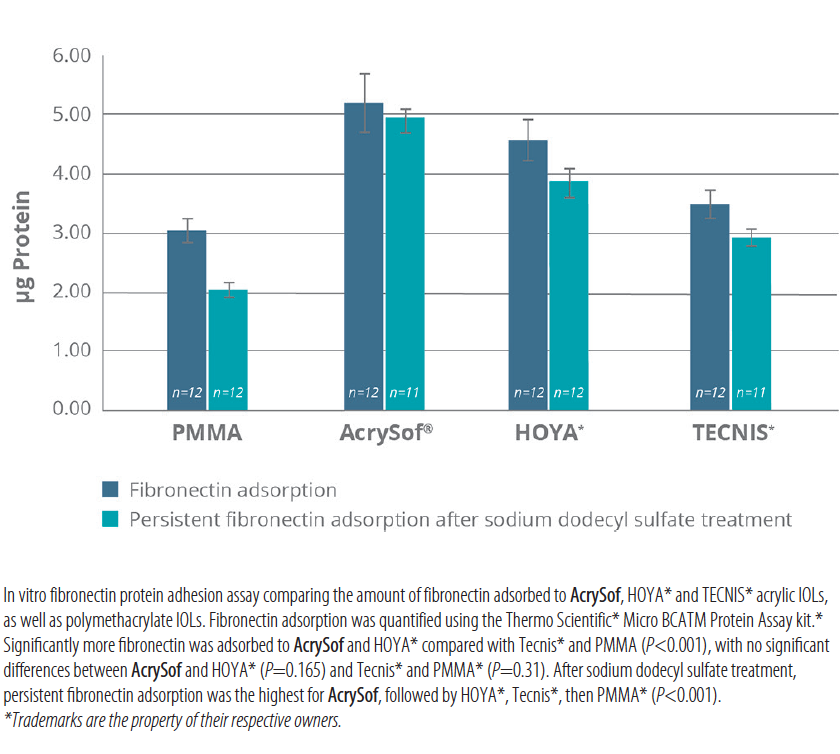
Figure 2. In vitro fibronectin absorption.
At 6 months, 62% of patients with the AcrySof IQ toric lens had ≤ 0.50 D residual refractive cylinder, while 88% had ≤ 1.00 D.8 In the same period, the lens was within 5° of the intended axis in 81.1% of patients.8 I am confident that the lens will not move. It stays where I place it, and it works. Because the AcrySof IQ toric IOL offers a range of spherical powers (+6.00 D to +34.00 D in half-diopter increments) and cylinder powers (seven, ranging from 0.75 D to 4.11 D), I can use the lens to achieve excellent outcomes for many of my astigmatic patients.
The AcrySof IQ ReSTOR +2.5 IOL with the ACTIVEFOCUS optical design is my lens of choice for patients who want presbyopia correction. The lens has a lot of options, which make it ideal for tailoring treatment to patients’ activities and visual goals. I like to combine the ReSTOR +2.5 with ACTIVEFOCUS design lens and the ReSTOR +3.0 lens for maximum patient benefit. I typically implant the ReSTOR +2.5 with ACTIVEFOCUS design lens in the dominant eye and the ReSTOR +3.0 lens in the nondominant eye. I believe that by using the ReSTOR +2.5 with ACTIVEFOCUS design lens, which is predominantly for distance, in the dominant eye that glare and halo are likely to be reduced due to the aspheric IOL design and excellent contrast sensitivity demonstrated with ReSTOR +2.5 with ACTIVEFOCUS design lens.
I appreciate the unique ACTIVEFOCUS design used in the ReSTOR +2.5 IOL. Like prior ReSTOR models, the lens has apodized segment heights that change the percentage of light focused on near and far across the lens, allowing more light to be concentrated on the distance image and near image depending on the pupil size. This is a proven platform. This is one of countless examples why Alcon's IOL platform remains my long-standing workhorse.
Blue Light Filtration and Ultraviolet blocking
The AcrySof IQ platform is available in two separate options, a blue light-filtration lens and a ultraviolet blocking-only lens. The blue light-filtration lens features a natural chromophore that mimics the healthy, natural crystalline lens’ transmission of light.4 Complementing the lens’ spherical aberration, blue light filtration also enhances contrast, helps reduce glare, and assists the eye in recovering from photostress.4
A study comparing the effects of blue light-filtering lenses with those that do not filter blue light found that this feature significantly reduced glare disability.4 Contrast sensitivity was much better in the blue light-filtering eyes, and eyes recovered from photostress much faster.4,9 For patients or surgeons who prefer ultraviolet blocking, the AcrySof IQ is also available in an ultraviolet blocking-only lens.
Smooth, Efficient Implantation
I have used the lenses in the AcrySof IQ platform for years, and it is by far my favorite go-to lens platform. Outcomes factor most heavily in that choice, and the ease and efficiency of the surgical procedures are compelling as well. In my experience, most of the AcrySof IQ IOLs (monofocal, toric, and multifocal) can be inserted through a small 2.2-mm incision if desired.

Figure 3. UltraSert Pre-Loaded Delivery System with the AcrySof IQ monofocal IOL.
I have implanted the AcrySof IQ monofocal with the UltraSert Pre-loaded Delivery System (Alcon) for several years. The delivery system is intuitive, and my staff and I do not need to handle and load the lenses, so the IOL remains pristine and untouched (Figure 3). In my experience with UltraSert, the AcrySof IQ lenses are delivered into the eye beautifully. Occasionally, I will have to reposition a haptic during delivery when using UltraSert. This repositioning takes seconds and simply entails rotating the lens into the bag. Overall, surgery with the UltraSert Pre-loaded Delivery System mirrors the lenses themselves, with an elegant design and comforting predictability that ultimately provide excellent outcomes for my patients.
© 2019 Alcon Inc. 06/19 US-NIQ-19-E-1113
AcrySof and UltraSert are trademarks of Alcon. All other brand/product names are the trademarks of their respective owners.
1. AcrySof IQ IOL Directions for Use.
2. Wang L, Santaella RM, Booth M, Koch DD. Higher-order aberrations from the internal optics of the eye. J Cataract Refract Surg. 2005;31:1512-1519.
3. Alcon. Data on Fil.
4. Hammond BR Jr, Renzi LM, Sachak S, Brint SF. Contralateral comparison of blue-filtering and non-blue-filtering intraocular lenses: glare disability, heterochromatic contrast, and photostress recovery. Clin Ophthalmol. 2010;4:1465-1473.
5. Gray R, Perkins SA, Suryakumar R, et al. Reduced effect of glare disability on driving performance in patients with blue light-filtering intraocular lenses. J Cataract Refract Surg. 2011;37(1):38-44.
6. Levy Y, Segal O, Avni I, Zadok D. Ocular higher-order aberrations in eyes with supernormal vision. Am J Ophthalmol. 2005;139(2):225-228.
7. Ong M, Wang L, Karakelle M. Fibronectin adhesive properties of various intraocular lens materials. Alcon Laboratories, Fort Worth, TX, USA. ARVO 2013.
8. US Food and Drug Administration. Summary of Safety and Effectiveness Data P930014/S15, AcrySof® Toric IOL. http://www.accessdata.fda.gov/cdrh_docs/pdf/P930014S015b.pdf. Published September 2005. Accessed February 21, 2018.
9. Fisher BL, Hammond BR Jr, Venkiteshwar MS. Visual effects of additional short-wave filtering in patients with clear intraocular lenses. Poster presented at: ASCRS; April 25-29, 2014; Boston, Massachusetts.
AcrySof® Family of Single-Piece IOLs Important Product Information
(AcrySof® UV, AcrySof ®IQ, AcrySof® IQ Toric, AcrySof®IQ ReSTOR®, and AcrySof® IQ ReSTOR® Toric IOLs)
CAUTION: Federal law restricts these devices to sale by or on the order of a physician.
INDICATION: The family of AcrySof® single-piece intraocular lenses (IOLs) includes AcrySof® UV-absorbing IOLs (“AcrySof® UV”), AcrySof ®IQ, AcrySof ®IQ Toric® and AcrySof IQ ReSTOR® and AcrySof® IQ ReSTOR® Toric IOLs. Each of these IOLs is indicated for visual correction of aphakia in adult patients following cataract surgery. In addition, the AcrySof Toric IOLs are indicated to correct pre-existing corneal astigmatism at the time of cataract surgery. The AcrySof IQ ReSTOR IOLs are for cataract patients with or without presbyopia, who desire increased spectacle independence with a multifocal vision. All of these IOLs are intended for placement in the capsular bag.
WARNINGS/PRECAUTIONS:
General cautions for all AcrySof® and AcrySof® UV IOLs: Careful preoperative evaluation and sound clinical judgment should be used by the surgeon to decide the risk/benefit ratio before implanting any IOL in a patient with any of the conditions described in the Directions for Use that accompany each IOL. Caution should be used prior to lens encapsulation to avoid lens decentration or dislocation. Viscoelastic should be removed from the eye at the close of surgery.
Additional Cautions associated with AcrySof® IQ ReSTOR® IOLs: Some patients may experience visual disturbances and/or discomfort due to multifocality, especially under dim light conditions. A reduction in contrast sensitivity may occur in low light conditions. Visual symptoms may be significant enough that the patient will request explant of the multifocal IOL. Spectacle independence rates vary with all multifocal IOLs; as such, some patients may need glasses when reading small print or looking at small objects. Clinical studies indicate that posterior capsule opacification (PCO), when present, may develop earlier into clinically significant PCO with multifocal IOLs.
Additional Cautions associated with AcrySof® IQ Toric, AcrySof® UV Toric and ReSTOR® Toric IOLs: Optical theory suggests that, high astigmatic patients (i.e. > 2.5 D) may experience spatial distortions. Possible toric IOL related factors may include residual cylindrical error or axis misalignments. Toric IOLs should not be implanted if the posterior capsule is ruptured, if the zonules are damaged, or if a primary posterior capsulotomy is planned. Rotation can reduce astigmatic correction; if necessary lens repositioning should occur as early as possible prior to lens encapsulation.
Prior to surgery, physicians should provide prospective patients with a copy of the appropriate Patient Information Brochure available from Alcon informing them of possible risks and benefits associated with the AcrySof® IQ Toric, AcrySof® IQ ReSTOR® and AcrySof® IQ ReSTOR® Toric IOLs.
Do not resterilize. Do not store at temperatures over 45° C. Use only sterile irrigating solutions to rinse or soak IOLs.
ATTENTION: Refer to the Directions for Use labeling for the specific IOL for a complete list of indications, warnings and precautions.
IMPORTANT PRODUCT INFORMATION
UltraSert® Pre-loaded IOL Delivery System with the AcrySof® IQ aspheric IOL
CAUTION: Federal (USA) law restricts this device to the sale by or on the order of a physician.
INDICATIONS: The AcrySof IQ aspheric intraocular lens (“AcrySof IQ”) is intended for the replacement of the human lens to achieve visual correction of aphakia in adult patients following cataract surgery. This lens is intended for placement in the capsular bag.
WARNING/PRECAUTION:
Use the UltraSert™ Pre-loaded Delivery System (“UltraSert”) at temperatures between 18° C (64° F) and 23°C (73° F). Use only Alcon viscoelastics qualified for this device. Do not use the UltraSert if the nozzle appears damaged or deformed. Follow the Directions for Use for correct order and sequence of steps to avoid damage to the IOL or the UltraSert.
Careful preoperative evaluation and sound clinical judgement should be used by the surgeon to decide the risk/benefit ratio before implanting a lens in a patient with any of the conditions described in the Directions for Use. Caution should be used prior to lens encapsulation to avoid lens decentrations or dislocations.
Studies have shown that color vision discrimination is not adversely affected in the individuals with the AcrySof® Natural IOL and normal color vision. The effect on vision of the AcrySof® Natural IOL in subjects with hereditary color vision defects and acquired color vision defects secondary to ocular disease (e.g., glaucoma, diabetic retinopathy, chronic uveitis, and other retinal or optic nerve diseases) has not been studied. Do not resterilize: do not store over 45° C.
ATTENTION: Reference the Directions for Use for Model AU00T0 for a complete listing of indications, warnings and precautions.






