CASE PRESENTATION
Case No. 1
A 69-year-old woman presents with a highly myopic left eye 10 years after pars plana vitrectomy (PPV) and a scleral buckle procedure on her right eye. The patient has light perception vision with full projection. She has not been able to recognize faces for about 2 years with this eye but had fairly good vision for some time after her retinal detachment (RD) surgery. She reports that she was told not to touch the eye because “the risk is too high.” She has been depending on her left eye for vision, but it has developed a cataract, causing her to lose her driver’s license. Her right eye has a Morgagnian cataract without a relative afferent pupillary defect (APD).
The patient requests surgery on her seeing eye but is fearful, because she has been told she will be at increased risk for an RD.
Case No. 2
A 70-year-old woman presents with a highly hyperopic right eye 2 years after noticing she could not see well with this eye. Her vision has been getting progressively worse over time, and now, she cannot see at all with her right eye. The patient seeks help, because her left eye has developed a cataract and she has lost her driver’s license. She has never seen an eye doctor before, and she wants her right eye fixed.
The patient has light perception vision with temporal projection and a 1.2-log unit relative APD with a Morgagnian cataract in her right eye. The IOP measures 19 mm Hg OD and 15 mm Hg OS. The angles are not closable, but she does not tolerate gonioscopy well. No other pathology is identified.
How would you proceed with these two patients?
—Cases prepared by Lisa Brothers Arbisser, MD.

SANDY T. FELDMAN, MD
These cases represent two different considerations of Morgagnian cataract. Prior to surgery, I would warn both patients that additional surgery might be required and that their visual outcome is unsure, due to prior retinal status in the first case and optic nerve damage in the second.
Case No. 1
This patient is rightfully fearful of cataract surgery on her left eye, because the procedure increases the risk of an RD. Someone who has had an RD in one eye is at increased risk of suffering an RD in the other eye, whether or not cataract surgery has been performed. This patient is also highly myopic, which further increases the chance of RD. Moreover, her history of PPV could increase the risk of intraoperative complications.
That said, the patient has few choices. The cataract is affecting her daily activities. Surgery obviously must be performed.
The presence of a Morgagnian cataract without elevated IOP or an APD suggests that the proteins have not yet significantly leaked out of the capsular bag and that cataract surgery should be performed before this happens. Depending on the tenseness of the capsular bag, I would favor a laser capsulotomy over a manual capsulorhexis, because the former will take less time and exert more uniform forces on the eye. I would not presoften the lens with the laser, however, out of concern over air bubbles.
Case No. 2
Lens proteins have leaked, increasing the IOP and damaging the optic nerve. The treatment is cataract surgery, and I would reduce inflammation and the IOP until the procedure was performed. I would order a B-scan and optical coherence tomography of the anterior segment. The high hyperopia and Morgagnian cataract with likely glaucoma increase the risk of a suprachoroidal hemorrhage.
The surgery itself is more complicated. I would prescribe mannitol preoperatively and administer a block because of the patient’s poor vision. I would paint the capsule with trypan blue and puncture it prior to creating the capsulorhexis to release liquid and help prevent capsular tears.

ERIC FRY, MD
Case No. 1
I would offer to perform surgery on the patient’s left eye initially after a long discussion with her regarding the risk of an RD, given her highly myopic status and history of RD in the right eye.
If the patient chose to proceed, I would approach the cataract in my normal fashion, using topical anesthesia and a temporal clear corneal incision. After entering the eye with the phaco tip, I would be especially watchful for reverse pupillary block, which is common in highly myopic eyes. If this problem occurred, I would gently lift the iris off the anterior capsule to equalize the pressure around the capsular bag and allow the capsular bag and iris complex to return to a normal configuration. In my experience, if reverse pupillary block develops, then it will recur each time the eye is re-entered. In these cases, I will occasionally place an iris hook to prevent recurrence.
Because it would be important to maintain the anterior chamber throughout the case, I would inject an ophthalmic viscosurgical device (OVD) through the sideport before removing the phaco or I/A tip. Chamber collapse could put traction on the vitreous, leading to a retinal tear.
Provided the cataract procedure went well, I would offer surgery on the patient’s fellow eye. I would expect surgery to produce reasonable vision, given her good visual acuity after the RD repair. I would approach the procedure on the second eye similarly to the first. That said, I would be more wary of loose zonules and have capsular tension rings and segments as well as capsular support hooks on hand. I would stain the capsule with trypan blue and puncture it with a cystotome. Lens milk is likely to spill forth. I would aspirate it with a 27-gauge cannula and replace the OVD as needed. I would disassemble the lens using a combination of vertical and horizontal chops and would frequently refill the eye with a dispersive OVD to protect the endothelium. I would place a single-piece hydrophobic acrylic IOL in the bag.
Case No. 2
Although the patient wants her right eye “fixed,” I would initially offer surgery on her left eye. With the 1.2-log unit APD and only temporal projection, it is unlikely that surgery on the right eye alone will improve her vision enough to allow her to drive. If she wished to proceed with surgery on the right eye first, however, I would have a long discussion with the patient on the guarded visual prognosis.
I would begin with peribulbar anesthesia and a temporal scleral tunnel incision in case extracapsular cataract extraction (ECCE) became necessary. I would expect the anterior chamber to be extremely shallow. After staining it with trypan blue and instilling a dispersive OVD, I would deepen the chamber and flatten the anterior capsule with a cohesive OVD placed centrally in a soft shell technique. I would puncture the capsule with a cystotome. I would aspirate the lens milk likely to spill from the puncture with a 27-gauge cannula and replace the OVD as necessary.
I would crack the nucleus into many small fragments using a combination of vertical and horizontal chops. I would refill the anterior chamber often with a cohesive OVD so as to protect the endothelial cells. Because it would be essential to avoid shallowing of the anterior chamber, which could lead to a choroidal hemorrhage in this small eye, I would instill an OVD through the sideport prior to exiting. I would place a single-piece hydrophobic acrylic lens in the bag.

CYNTHIA MATOSSIAN, MD
Case No. 1
Complex cases like this one require a clear preoperative discussion of the increased risks with the patient. Informed consent for a possible vitrectomy is recommended. Based on her high myopia and history of an RD and repair, I would refer the patient for a retina consultation prior to cataract surgery. Depending on the surgery center, I would try to schedule the case on a day when a retina surgeon would be available on standby.
Because of the patient’s high myopia, I would recommend using intraoperative aberrometry (ORA System; Alcon) or at least the Wang-Koch adjustment for the IOL power calculations to ensure the best refractive outcome. If I observed a taut capsule with liquefied cortex, I would puncture the capsule and be ready to aspirate the lens milk and liquefied cortex with the I/A tip. I would then make a small capsulotomy to try to avoid the Argentinian flag sign while ensuring that the anterior chamber was sufficiently filled with a viscoadaptive OVD.
I would first attempt traditional phacoemulsification. If the nucleus proved too dense, I would enlarge the main corneal wound, proceed with an ECCE, and close the eye with multiple interrupted sutures.
Case No. 2
Again, this case requires a careful preoperative discussion with the patient and appropriate informed consent.1 Prior to surgery, I would refer this patient for a glaucoma consultation for additional input on the optic nerve, given the APD. Because of the high hyperopia, I would consider prescribing mannitol preoperatively to decrease the vitreous volume. If I felt the anterior chamber space was still too tight, I might perform a PPV to create more working room in the anterior chamber. I would perform intraoperative aberrometry to ensure the best refractive outcome.
Uveal effusion is always a concern in highly hyperopic eyes.2 I would try to maintain consistent fluidics to avoid IOP fluctuations by constructing watertight corneal incisions and minimizing the insertion and removal of instruments. I would also lower the bottle height. I would try to keep the anterior chamber as stable as possible to avoid endothelial cell loss, so I would pause frequently to refill the anterior chamber with an OVD.
As in the first case, if I observed a taut capsule with liquefied cortex, I would make a puncture and be ready to aspirate the lens milk and liquefied cortex with the I/A tip. I would then create a small capsulotomy in an effort to keep the capsulorhexis from tearing out to the equator, while making sure that the anterior chamber was sufficiently filled with a viscoadaptive OVD at all times.
I would first attempt traditional phacoemulsification. If the nucleus were too dense, I would enlarge the main corneal wound, proceed with an ECCE, and close with multiple interrupted sutures. Enlargement of the wound and the associated IOP fluctuations might increase the risk of uveal effusion.

WHAT I DID: LISA BROTHERS ARBISSER, MD
Case No. 1
The patient and I decided I would operate on her right eye first and aim for emmetropia (she had worn contact lenses for years) with the understanding that I would operate on her left eye after treatment of a superior hole in lattice and once her right eye stabilized. After full consent (including the risk of diplopia due to long-standing sensory deprivation), under a peribulbar block, the right eye was prepped with mannitol ¼ g per kg intravenous push. A supersoft shell of Viscoat (Alcon) and Healon5 (Abbott Medical Optics) stabilized the chamber, and I painted trypan blue dye sparingly underneath to stain the anterior capsule. Upon raising a small flap in the central anterior capsule with a cystotome, I controlled escaping lens milk with the instillation of additional Healon5.
Using a forceps, I created a small continuous curvilinear capsulorhexis (CCC; Figure 1). I lifted the iris off the anterior capsule with a Rosen splitter inserted through the paracentesis to prevent reverse pupillary block and deepening of the anterior chamber in this large eye with a floppy iris, while automated I/A removed the lens milk from the anterior chamber. During this step, I instilled more dispersive OVD to keep the floating brunescent nucleus in the bag. I measured and enlarged the CCC to prevent rupture in case the nucleus prolapsed but kept the CCC smaller than the optic. Using an endocapsular cross-action chop circumferential disassembly technique with burst mode on and a panel setting of 90% longitudinal ultrasound, I finished removing the nucleus and repeated instillation of an OVD as needed to sandwich the lens material within the bag and prevent endothelial stress. There was no cortex. I vacuumed the anterior capsule, polished the posterior capsule with a Terry squeegee, and spiraled the CCC again to overlap the optic by a millimeter once in place.
Postoperatively, the patient has transient diplopia with a UCVA of 20/20. She has a high and dry buckle on dilated examination. When her right eye stabilizes, her fellow eye will undergo cataract surgery with careful retinal follow-up.
Case No. 2
Informed consent included the very poor prognosis for useful vision in the patient’s right eye and the distinct possibility that more surgery or laser treatment would be needed to achieve the best outcome or even to avoid a total loss of vision. I further explained to the patient that she might even lose the eye. After hearing of laser cataract surgery, she begged for the option so that she would know everything possible had been done. My practice received funding on a humanitarian basis for the laser surgery, and I decided to attempt a laser capsulotomy. First, I consulted with other experienced surgeons, who recommended new parameters. As a result, I increased the energy and manually adjusted the automated program to recognize the posterior surface of the lens and perform a 4.8-mm capsulotomy. Under a peribulbar block and with an experienced laser technician at my side, I watched the Catalys Precision Laser System (Abbott Medical Optics) complete the capsulotomy in 2 seconds and saw small bubbles for 360º.
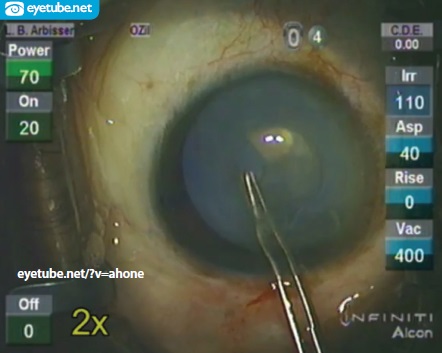
Figure 1. Dr. Arbisser clears the lens milk by adding an OVD to control the chamber in order to see the capsular opening she created.
On the table, there was no view within the chamber, which was totally filled with lens milk. The eye was entered with an OVD to control the chamber. I attempted to identify the edges of the capsulotomy through painting with trypan blue. Next, I executed a dimple-down maneuver to release the capsule flap.3 With the forceps, I could detect no open edges. Using a gentle tapping technique (like a blind person with a cane), I finally found one open area, which was already headed for the periphery. An unseen vessel started to bleed from the iris root, remote from any of my manipulations and for no apparent reason (Figure 2). While trying to complete the capsular opening, a strangely clear area became evident at 7 o’clock.
Deciding to convert to an ECCE, I enlarged the incision and used a nonirrigating lens loop to remove the nucleus in a dispersive OVD sandwich. The incision was partially sutured, and the bleeding was controlled with an OVD and time (2 minutes by the clock). Because neither the zonules nor the capsule was intact, I removed the bag remnants and performed an anterior vitrectomy through the sutured-down clear corneal incision to make a tight fit with irrigation through the original paracentesis. My usual vitrectomy technique is through a pars plana sclerotomy; owing to the unknown potential posterior pathology and the now one-chambered eye, however, I opted for the anterior approach.
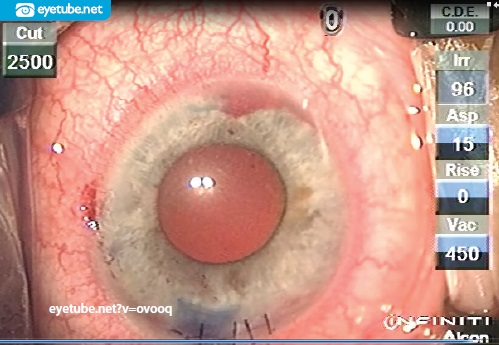
Figure 2. Appearance of the aphakic eye immediately after laser cataract n f inancial interest: none acknowledged surgery and unplanned vitrectomy with iris root clot (not iridodialysis) on an eye with a Morgagnian cataract.
A sterile indirect examination while the patient was on the table revealed a central retinal vein occlusion. Because of the poor visual prognosis, the likelihood of neovascularization and ischemia, and the white-to-white of 10.25 mm, I decide to leave the eye aphakic.
Postoperatively, there was a small hyphema, mild corneal edema, and vitreous haze. The pupil was round, and the IOP was normal. The patient had light perception vision with temporal projection with and without an aphakic refraction. I referred her to a retina specialist for observation of the central retinal vein occlusion and early neovascularization. Ultimately, the eye quieted, and it remains aphakic. The patient is content that all that can be done has been done. She opted for manual surgery for the fellow eye’s cataract.
1. Utman SA. Small eyes big problems: is cataract surgery the best option for the nanophthalmic eyes? J Coll Physicians Surg Pak. 2013;23(9):653-656.
2. Elagouz M, Stanescu-Segall D, Jackson TL. Uveal effusion syndrome. Surv Ophthalmol. 2010;55(2):134-145.
3. Arbisser LB, Schultz T, Dick HB. Central dimple-down maneuver for consistent continuous femtosecond laser capsulotomy. J Cataract Refract Surg. 2013;39(12):1796-1797.
Section Editor Lisa Brothers Arbisser, MD
• emeritus position at Eye Surgeons Associates, the Iowa and Illinois Quad Cities
• adjunct associate professor, John A. Moran Eye Center, University of Utah, Salt Lake City
• (563) 343-8896; drlisa@arbisser.com
• financial interest: none acknowledged
Section Editor Audrey R. Talley Rostov, MD
• private practice with Northwest Eye Surgeons, Seattle medical advisory board, SightLife, Seattle
Sandy T. Feldman, MD
• medical director, Clearview Eye & Laser Medical Center, San Diego, California
• sfeldman@clearvieweyes.com
Eric Fry, MD
• partner, Fry Eye Associates, Garden City, Kansas
• associate professor of ophthalmology, University of Kansas Medical School, Kansas City, Kansas
• efry@fryeye.com
Cynthia Matossian, MD
• founder and CEO of Matossian Eye Associates, with offices in Pennsylvania and New Jersey
• clinical instructor/adjunct faculty member, Temple University School of Medicine in Philadelphia and Robert Wood Johnson Medical School in New Brunswick, New Jersey
• cmatossian@matossianeye.com
• financial interest: none acknowledged


