CASE PRESENTATION
A 65-year-old man suffered a contusion injury to his right eye when he was 10 years old. He reports no surgical intervention and progressive vision loss since the injury to the point of functional blindness. The patient states that everyone has told him not to touch this eye. Now, however, a cataract has developed in his fellow eye that is making it difficult for him to drive at night, and he is seeking help.
On examination, there is barely a 0.3 log unit relative afferent pupillary defect (APD) with good light projection in the right eye. The cornea is unremarkable but for a cell count of 800. The pupil shows a small sphincter defect, only dilates to 4 or 5 mm, and is white with scarring of the anterior capsule without phacodonesis (Figure). The IOP is within normal limits. B-scan shows no retinal detachment or mass. The patient's left eye is normal but for 3+ nuclear sclerosis and a mild posterior subcapsular cataract resulting in 20/50 acuity.
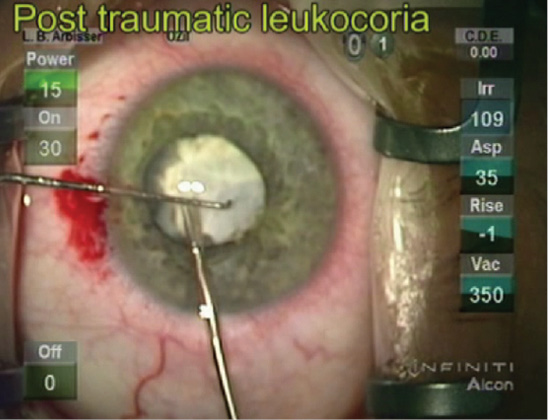
Figure. Traumatic leukocoria.
What intervention would you offer to this patient? What would your surgical plan be? What drugs or devices would you have handy in the OR?
—Case prepared by Lisa Brothers Arbisser, MD.
D. MICHAEL COLVARD, MD
It is difficult to know the visual potential of the patient's right eye, but I think it is worth trying to restore the best level possible. I would start by performing gonioscopy to see whether or not there is an angle recession. If there is, the underlying zonules are likely to be the weakest, so I would try to place the incision opposite the area of suspected fragility. This orientation would help me to avoid maneuvers that aggravate zonular weakness. I would make a scleral incision in case extracapsular cataract extraction became necessary.
The anterior capsule is fibrotic. Actually, the entire lens may be nothing more than a very thick, hard plate of fibrotic tissue. High-frequency ultrasound biomicroscopy (Paradigm Medical Industries) or anterior segment optical coherence tomography (Carl Zeiss Meditec) would noninvasively provide high-resolution in vivo imaging of the anterior segment. Either modality would help the ophthalmologist to visualize a cross-section of the lens and plan the surgical approach, but I do not think using the technology is essential in this case. The surgeon just has to be prepared for the worst.
An ordinary continuous-tear capsulorhexis will not work here, and attempting one will likely place unwanted stress on the zonules. A laser capsulotomy is also not likely to work well, because the thick, bone white anterior capsule will bisect and diffuse the laser energy. This will tend to prevent optical breakdown. The only dependable way to open this anterior capsule is to find a relatively thin area in it, to create a hole large enough for the introduction of microsurgical scissors (MicroSurgical Technology [MST]), and to cut an “anterior capsulotomy.” Again, I recommend being prepared to cut through a fibrotic plate that incorporates what is left of the entire lens. In that situation, I would inject a dispersive ophthalmic viscosurgical device (OVD) under the plate to prevent forward movement of the vitreous.
I would implant a three-piece IOL in the sulcus. Because of the density of the crystalline lens and the relative unreliability of standard ultrasound measurements for biometry, intraoperative aberrometry would be quite helpful for IOL power selection. I like the tables prepared by Warren Hill, MD, for converting bag-placement calculations to sulcus placement, because the reduction of IOL power varies as a function of IOL power
(http://www.doctor-hill.com/iol-main/iol_main.htm).
I would plan to use pupillary expanders. I would also have at the ready capsular tension rings (CTRs), capsular hooks, and Cionni Rings for Scleral Fixation (Morcher, distributed in the United States by FCI Ophthalmics), although these might not be useful if the anterior and posterior capsules are fused. Finally, I would be prepared to suture the three-piece IOL in place if the capsule were unstable.
JASON JONES, MD
I would offer surgery on the patient's left eye first, because this is the presenting complaint. I anticipate a normal surgical course for this eye with routine recovery and no special considerations for surgery.
I would encourage the patient to undergo surgery on his right eye, given the small APD and light projection vision. I would explain that the risks of surgery are greater for this eye because of the history of trauma, that surgery and recovery will take longer than usual and pose a higher risk to the cornea (in both the short and long term), and that the final visual outcome is uncertain. Not performing surgery or deferring intervention remains a reasonable option.
If the patient decided to proceed with cataract surgery on his right eye, I would approach the procedure in a largely routine manner with topical/intracameral anesthesia and intravenous sedation. The patient would be kept nil per os and his surgery scheduled for the end of the day. Through a temporal clear corneal incision and using the Arshinoff soft shell technique to inflate the anterior chamber,1 I would place iris hooks to expand the pupil and then paint the anterior capsular surface with trypan blue ophthalmic solution (VisionBlue; Dutch Ophthalmic USA). I would incise the capsule with a disposable 30-gauge needle and fashion a capsulorhexis with a Utrata forceps. A Fugo Blade (Medisurg) might be useful for cutting the adherent capsule, given its fibrotic appearance.
Although I would attempt hydrodissection, I anticipate poor success with this step, in which case I would proceed to “debulking” the nucleus using modulated nonlongitudinal phacoemulsification with low fluidic parameters. Once sculpted deeply, the nucleus could be cracked and a dispersive OVD used to inflate the capsule and viscodissect from it the cataract. The remaining nucleus could be approached with moderate fluidic parameters and the periodic re-application of a dispersive OVD to protect the endothelium. Any residual cortex should be aspirated with bimanual I/A. A posterior chamber IOL (PCIOL) could then be placed in the bag.
If the capsule became compromised with nucleus still present, I would convert to a temporal scleral tunnel and manual extraction of the cataract. I would stain the vitreous with triamcinolone acetonide (Triesence; Alcon) and perform a bimanual limbal anterior vitrectomy. IOL choices would include a sclera-fixated (glued or sutured) or iris suture-fixated PCIOL or an appropriately sized anterior chamber IOL. If possible, I would avoid using an anterior chamber IOL because of the compromised cornea. Regardless of the events of surgery, the retina requires inspection after surgery.
P. DEE G. STEPHENSON, MD
This interesting case requires a lot of thought about safety, what is best for the patient, and realistic outcomes. Cataracts caused by blunt trauma classically form stellate- or rosette-shaped posterior axial opacities that can be stable or progressive. Blunt trauma is responsible for coup and contrecoup ocular injury. The former is the mechanism of direct impact, whereas the latter is the distant injury caused by shockwaves traveling along the line of concussion. If the anterior surface of the eye is struck bluntly, rapid anterior-posterior shortening is accompanied by equatorial expansion. Such stretching can disrupt the lens capsule, zonules, or both. A combination of coup, contrecoup, and equatorial expansion is responsible for the formation of a traumatic cataract after blunt ocular injury.
The APD most likely associated with anterior ischemic optic neuropathy may limit this patient's postoperative vision. It would therefore be extremely important to make him aware of the guarded visual outcome and to ensure that he understands that the ophthalmologist has more than one surgical plan.
The patient's right eye might be well suited to laser capsulotomy and fragmentation because of the low cell count. The laser procedure can decrease the amount of phaco ultrasound needed and thereby reduce the loss of cells.
Manual phacoemulsification can be performed if the lens capsule is intact and sufficient zonular support remains. Staining of the capsule would be imperative, as would be the placement of pupil expanders and a CTR. I would also plan to use microcutters and micrograspers such as those manufactured by MST, because I am sure the capsule is very fibrotic and will be difficult to tear.
If there is a chance of a ruptured posterior capsular, posterior dislocation of the lens, or severe zonular instability, a pars plana lensectomy and vitrectomy would also be an option.
I would use a three-piece, fourth-generation, silicone PCIOL like the LI61AO (Bausch + Lomb) or a three-piece hydrophobic acrylic like the EC-3 (Aaren Scientific). Both lenses could be fixated to the capsule with adequate capsular and zonular support or to the sulcus.
WHAT I DID:
LISA BROTHERS ARBISSER, MD
The effectively counseled patient is given a guarded prognosis and is prepared for the possibility of more than one surgery to achieve the best outcome. I explained to this patient the challenge of the longstanding cataract and scarred capsule. I anticipated the possible need to implant the IOL in the sulcus. My “tough case bin” was at hand, which contains intraocular microinstrumentation, CTRs, and the like. Also available were devices for zonular abnormalities and a backup STAAR AQ IOL (STAAR Surgical).
Topical atropine along with tropicamide and phenylephrine as well as intracameral 1:4,000 preservative-free epinephrine and gentle synechiolysis dilate stiff, small pupils—often avoiding the need for pupil-expanding devices. I inspected the periphery by retracting the edge of the iris to evaluate zonular status. The edge of the nucleus can masquerade as the equator of the bag in such cases and give the appearance of missing zonules. Staining the capsule with trypan blue dye was essential. A scarred capsule of long standing can delaminate. Staining is confusing in capsular schisis, however, so I was prepared to paint extra trypan blue dye strategically under an OVD with an Osher cannula (opening not at the tip but underneath). I also employed a more viscous cohesive OVD (Healon GV; Abbott Medical Optics) under the usual soft shell of Viscoat (Alcon) to help stabilize the white cataract in case of intralenticular pressure, although the longstanding injury made it likely that any liquefied cortex had been partially absorbed.
It is rarely possible to achieve a truly continuous tear with fibrosis and schisis. Under high magnification, I nevertheless managed to complete at least the top layer of the capsulorhexis by frequent regrasping and the application of centripetal vector forces as needed. The white subcapsular plaque also stained as though there were still a layer of capsule adherent. Exploration with a Sinskey hook and MST grasper forceps revealed a dense, calcific plaque of anterior cortex. This was bluntly and sharply dissected from the underlying cortex and epinucleus. I took great care not to stress fragile zonules. Once free, the plaque required sectioning—as for an IOL exchange—in order to be extracted through the 2.4-mm clear corneal incision. The question then became how to free the nucleus from the bag, because it all appeared to be a solid block. (To view the surgical video, visit eyetube.net/?v=abepa.)
The uncertain integrity of the edge of the capsulorhexis led me to perform limited, multidirectional hydrodissection, which proved ineffective. Gently rocking the dense lens did not quite allow rotation. A CTR and capsule-expansion hooks were contraindicated, so I began cross-action vertical chop phacoemulsification. I stepped down aggressive fluidics to protect the fragile capsule. Longitudinal burst mode at a 50% ultrasound panel setting (appropriate to lens density and brunescence) facilitated chopping of the nucleus into hemispheres without zonular stress. As often happens, this approach allowed me to lift the calcific, dense nucleus away from the “nucleofied” epinucleus, which remained as a stent in the bag while I further chopped and evacuated the last nuclear fragments (circumferential disassembly). By then, the epinucleus was mobile and removed, with the chopper positioned to protect the floppy bag. One cannot afford to lose chamber at any point in a surgery without an intact capsulorhexis or capsule.
I injected an OVD through the sideport before removing the phaco tip. Roughing up thinned cortical remnants adherent to the posterior capsule with a Terry squeegee facilitated manual cortical removal while avoiding the turbulence and capsular risk associated with automated I/A. A clean bag fornix was obtained with inspection and targeted removal. In my opinion, residual cortex—no matter how scant—increases the chance of late IOL decentration and fibrosis. In a case with an intact anterior capsulorhexis and zonules, I would perform a posterior capsulorhexis to remove residual plaque but not in this case of uncertain anterior capsular integrity.
My choice of IOL was informed by careful inspection, which revealed adequate anterior capsule to reliably cover the haptics of a single-piece acrylic lens and showed what appeared to be a stable bag. After standard lens implantation, acetylcholine for miosis proved coverage of the IOL's edges despite a preoperative traumatic sphincter tear and obviated the need for iris sutures. I instilled Triesence in addition to my routine, off-label intracameral injection of moxifloxacin (Vigamox; Alcon) to ensure the absence of unseen vitreous prolapse and to provide inflammatory prophylaxis. The patient had 20/20 vision with scattered sotomata secondary to old commotio retinae. The other eye subsequently had a 20/20 result.n
1. Arshinoff SA. Dispersive-cohesive viscoelastic soft shell technique. J Cataract Refract Surg. 1999;25:167-173.
Section Editor Lisa Brothers Arbisser, MD
• emeritus position at Eye Surgeons Associates, the Iowa and Illinois Quad Cities
• adjunct associate professor, John A. Moran Eye Center, University of Utah, Salt Lake City
• (563) 343-8896; drlisa@arbisser.com
• acknowledged no relevant financial interest
Section Editor Tal Raviv, MD
• founder and director, Eye Center of New York
• clinical associate professor of ophthalmology, New York Eye and Ear Infirmary of Mount Sinai
Section Editor Audrey R. Talley Rostov, MD
• private practice with Northwest Eye Surgeons, Seattle
D. Michael Colvard, MD
• clinical professor of ophthalmology, Keck School of Medicine, University of Southern California, Los Angeles
• director, Colvard-Kandavel Eye Center, Encino, California
• (818) 906-2929; mike@mcolvard.com.
• acknowledged no relevant financial interest
Jason Jones, MD
• medical director, Jones Eye Clinic, Sioux City, Iowa
• (712) 239-3937; jasonjonesmd@mac.com
• acknowledged no relevant financial interest
P. Dee G. Stephenson, MD
• private practice, Stephenson Eye Associates, Venice, Florida
• associate professor of ophthalmology, University of South
Florida College of Medicine, Tampa
• (941) 468-6363; eyedrdee@aol.com
• acknowledged no relevant financial interest




