Many surgeons are interested in streamlining the delivery of medications and the postoperative care associated with cataract surgery. In 2005, pioneers such as James Gills, MD, and Paul Koch, MD, explored and assembled combinations of steroids and antibiotics and also tested various delivery methods. Their experiences guided subsequent formulations and methodologies useful for developing a dropless cataract surgery product, now known as Tri-Moxi-Vanc (compounded triamcinolone, moxifloxacin, and vancomycin; Imprimis Pharmaceuticals), for concurrent placement at the time of intraocular surgery.
PROBLEM IN NEED OF A SOLUTION
My rationale for developing compounded triamcinolone, moxifloxacin, and vancomycin, administered with each procedure, was based on a multitude of clinical considerations associated with the intraoperative and postsurgical treatment of my ocular surgery patients.
Cataract procedures represent a high percentage of the surgeries I perform, and most of my patients undergoing cataract surgery are elderly and have comorbidities. After surgery, controlling ocular inflammation and administering topical antibiotics to prevent infection, historically done by prescribing various eye drops, are critically important. Many patients, however, report difficulty in self-administering many different antibiotic and steroid eye drops, multiple times per day and on different schedules, often for 4 or more weeks after surgery. Old and young patients have difficulty complying with the eye drop regimen, and family members are often (inconveniently) recruited to assist patients.
Moreover, I found that fourth-generation fluoroquinolone antibiotics were harder for the patients to obtain because of increasingly restrictive insurance formularies. Due to these frequent formulary issues and because the high cost of drugs affected access to optimal antibiotics, surgeons in my office and many others were compelled to use older-generation products, despite the fact that pathogens were becoming increasingly resistant to these more commonly used and less expensive antibiotics. I was concerned that this would increase the risk of catastrophic infection for my patients, especially as methicillin-resistant Staphylococcus aureus became prevalent in our community.
On a separate front, in ambulatory surgery centers (ASCs), the Centers for Medicare & Medicaid Services announced a complete ban on any multiuse products in the OR, limiting the use of antibiotics in the ocular irrigation solutions and for intracameral injections.
DEVELOPING AN INTRAOCULAR STEROID-ANTIBIOTIC COMBINATION
I realized it would be possible to overcome these therapeutic noncompliance and access issues by recruiting accredited compounding pharmacists to develop a single, sterile injectable product that would ultimately achieve all of my clinical goals. Working with clinical pharmacists familiar with intraocular compounded drugs, we studied the chemical compatibility and solubility of certain antibiotics and steroids in a properly balanced pH and osmolar formulation. We took a cue from retina specialists and their preference for the treatment of endophthalmitis and cystoid macular edema. We also noted bacterial resistance profiles in the United States. After extensive efforts, the combination of these two effective broad-spectrum, gram-positive antibiotics, vancomycin and moxifloxacin, were blended with long-acting triamcinolone.
We considered various drug-delivery techniques, based on past published experiences, including subconjunctival placement, pars plana injection (under direct visualization), and transzonular cannular injection after IOL insertion. Drs. Stewart Galloway and Kevin Scripture shared their encouraging experience with the transzonular method, which was explored and documented as safe and effective using endoscopic videography. The particulate nature of the Tri-Moxi-Vanc was refined to permit easy delivery through a 30-gauge cannula.
Injecting antibiotics inside the eye at the time of the surgical event, when the potential contamination with the causative pathogen most likely occurs, treats the eye directly and concurrently with inoculation. Additionally, because endophthalmitis occurs in the nutrient-rich vitreous, delivery of the drugs into the vitreous cavity has fundamentally greater traction to prevent postoperative infection compared to anterior chamber placement.
Combining these antibiotics with a medium-strength corticosteroid in a single formulation, subsequently bound into the protein matrix of the vitreous, simultaneously controls for surgically induced inflammation over several postoperative weeks. In most cases (93%-97%), use of this sterile preservative-free injectable compounded product eliminates the need for postsurgical antibiotics and steroid eye drops. Postoperative cystoid macular edema rates are lower and superior to topical therapies, and IOPs are not elevated at this delivery dose.1-4 Additionally, patients' satisfaction is very high, with short-term floaters and immediate vision issues dissipating quickly. Specifically, 80% of patients noticed floaters on the first postoperative day, and only 20% noticed floaters on the first postoperative week visit. Patients' satisfaction was determined by interview (not quantitative) and represents the general experience of myself and others.
DROPLESS CATARACT SURGERY
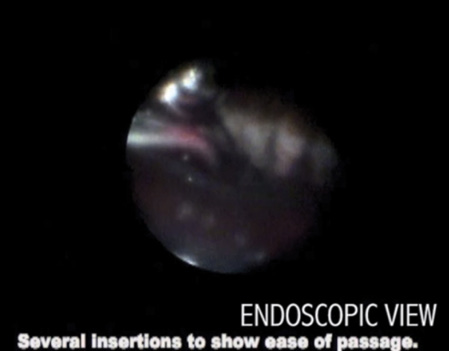
An endoscopic view of the cannula's tip traversing the zonules during Tri-Moxi-Vanc injection with a 27-gauge Knolle cannula (2-mm distal segment) after IOL placement and under viscoelastic.
Injection of Tri-Moxi-Vanc showing the plume of visible triamcinolone suspension (with two antibiotics) within the vitreous. Note that viscoelastic is beginning to exit the wound as the IOL moves more anteriorly during expansion of the posterior segment, a helpful confirmatory sign. This intravitreal plume of medication is seen from the microscope's view in 90% of the injection events; 10% of the the medicine is properly placed but not visualized centrally. To view the entire surgical video, go to eyetube.net/?v=uflim.
PATENTED PRODUCT AND PHARMACEUTICAL MANUFACTURING
The manufacturing process was carefully documented, using only the highest-quality American-made drugs, with the detailed compounding steps carefully monitored and videoed. Manufacturing follows national and FDA standards defined as Good Manufacturing Process. Initially, I documented (and filmed) how the pharmacists were assembling the medications and what primary drugs were being used in the process. This graduated to full-on quality control measures to reflect the FDA's stringent mandates.
Once the compounded product had several years of demonstrated performance and the number of eyes exceeded 10,000, the drug combination and methodology (with other similarly produced drug combinations explored and tested) were submitted for a US patent, which was then transferred to Imprimis Pharmaceuticals for national and international distribution under its Dropless Cataract Surgery campaign.
More recently, the FDA has elevated the manufacturing responsibilities of compounded pharmaceuticals, bringing them to a quality level on par with that of traditional FDA-approved manufactured drugs.
CONCLUSION
Dropless cataract surgery using Tri-Moxi or Tri-Moxi-Vanc has become an important part of modern ophthalmology for an increasing number of eye surgeons in the United States—more than 200—and in many other countries. The opportunity to reduce infection risk, control postoperative inflammation, simplify therapeutic compliance, and reduce costs for patients with a single intraoperative injection is a powerful intervention that adds value to an already outstanding and life-changing procedure.n
Jeffrey T. Liegner, MD
• practices at Eye Care Northwest in Sparta, New Jersey
• liegner@eyecarenw.com
• consultant to and investor in Imprimis Pharmaceuticals


