The 18 tips for enhancing the safety and efficacy of complex cataract surgery presented herein also apply to standard procedures. For this reason, it makes sense for you to learn these skills and techniques and to have the equipment available for all cases.
No. 1. UNDERSTAND PHACO SETTINGS AND DYNAMICS
Phacoemulsification requires hand-eye-foot-ear coordination. Understand your machine’s settings. Do not “set it and forget it.”
Inexperienced? Practice using the foot pedal without patients. Listen to the pump’s crescendo as you pinch the tubing to simulate partial and total occlusion. Practice foot position 1 without unintentionally lightening up to foot position 0 so that you avoid loss of chamber and obviate the need for continuous irrigation. Switch from foot position 3 to 2 without losing vacuum, thereby facilitating chop.
Case gone awry? Think about your machine’s settings. Reduce the flow rate to slow things down? Decrease the vacuum, or raise the irrigation bottle? Is there chatter? Consider lowering phaco power or switching to noncontinuous mode. Establish flow in foot position 2 before engaging ultrasound in foot position 3, and recognize the occlusion bell in order to avoid wound burn. When is burst or pulse preferable? When is linearity advantageous or counterproductive? Understand rise time, duty cycle, and the behavioral differences of Venturi and peristaltic pumps. This essential knowledge can save the day in both complex and standard cases.
No. 2. MAINTAIN A NORMOTENSIVE EYE AND NORMAL PHYSIOLOGIC TISSUE RELATIONSHIPS WHEN POSSIBLE
The eye is most vulnerable when you are removing instruments through an incision that is not truly closed, leaving low pressure in the chamber with resultant collapse. In every case, support the anterior chamber (AC) by irrigating balanced salt solution (BSS; Alcon) through the sideport incision before removing the phaco and I/A tips. When ocular anatomy is abnormal, you may have to instill an ophthalmic viscosurgical device (OVD) through the paracentesis prior to removing instruments.
Not only is allowing a chamber to shallow undesirable, but so is permitting sudden deepening. In susceptible patients, such as those with high myopia or status post vitrectomy, prevent iris retropulsion (reverse pupil block). In an OVD-filled chamber, enter the AC with the phaco tip in foot position 0. Lift the iris off the anterior capsule with the instrument in your nondominant hand prior to initiating foot position 1 so as to equalize fluid between the AC and posterior chamber (PC).
Maintaining a stable chamber and a normotensive eye throughout surgery is the most critical secret to success in complex and complicated cases in particular.
No. 3. MAXIMIZE VISUALIZATION
High-magnification, dynamic microscope focus and capsular dyes are essential tools for cataract surgeons. Placing trypan dye without an air bubble can lead to a loss of chamber and is mildly toxic to the endothelium. For eyes with white cataracts, irrigate to fill the AC with dye that is then progressively displaced by an OVD. In eyes with subluxation or an errant continuous curvilinear capsulorhexis (CCC), first compartmentalize the AC from the PC with an OVD. Then, paint dye strategically to stain capsular structures under the OVD. Staining is always helpful when you are planning to use endocapsular devices.
No. 4. MAINTAIN AND FOSTER PATIENTS’ COOPERATION
Respond promptly and kindly to patients’ discomfort. Use appropriate anesthesia. Choose akinesia via peribulbar injections when the surgical plan includes sutures to the sclera or iris. Administer minimal doses of short-acting intravenous anxietolytics and analgesics for patients’ comfort and cooperation, not for hypnosis or amnesia. Use “vocal local” liberally.
Maintain positive pressure during a patient’s Valsalva maneuver. Many surgeons withdraw instruments when patients cough. This response facilitates chamber collapse, can cause incisional prolapse of the iris, and invites retrodirected fluid later in the case, leading to posterior pressure. To maintain positive pressure, lock your hands onto the patient’s face, train the scrub nurse to lay a hand on the patient’s forehead to hold his or her head on the table, and stay in foot position 1 or 0 (never 2 or 3). Preserving physiologic relationships within the eye prevents complications related to a Valsalva maneuver.
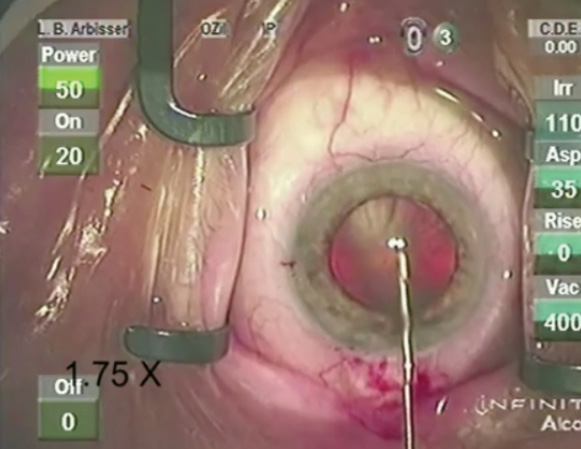
Figure. This eye presents typical signs of loose zonules such as a pincushion effect upon puncture of the capsule.
No. 5. CONSTRUCT THE INCISION TO CONTROL FLUIDICS
Surgeons typically size the main incision for a tight fit, but according to Richard Mackool, MD, traditional 1-mm paracenteses lose 22 mL/min of BSS when a chopper is in place (written communication, April 2011). Consider a small-profile Rosen phaco splitter (Katena Eye Instruments) for chopping to reduce paracenteses to less than 0.5 mm. This minimizes flow and turbulence and prevents you from having to chase particles to the incision.
No. 6. MANIPULATE THE PUPIL CAREFULLY
Malyugin Rings (MicroSurgical Technology [MST]) have dramatically changed cataract surgery on patients with intraoperative floppy iris syndrome. Becoming adept at using this device will benefit you and your patients. In my experience, iris hooks are a better choice only when an extra foreign body in the eye is problematic or for viewing a specific area of lens. The Fry stretching maneuver can still be useful for normal or stiff irides. This technique involves gentle, two- or three-point widening of the pupillary aperture, which stretches but preferably does not tear the sphincter. Always be careful not to overstretch or tear iris tissue, which can cause postoperative anomalies.
No. 7. ANTICIPATE ZONULAR LAXITY
The history and examination may provide preoperative warning signs like pseudoexfoliation, trauma, previous surgery, and asymmetric or incongruous AC depth. Recognize zonular laxity intraoperatively by the pincushion effect during the capsule’s puncture (ie, a dimpled appearance with striae radiating peripherally; Figure). Regrasp the edge of the capsulorhexis to direct vector forces. Stabilize even loose lenses for the CCC by completely filling the AC with an OVD. Seek the midpoint of the subluxated lens in order to center the CCC on the lens and not on the pupil. Recognizing extreme laxity or lysis early facilitates better intervention (capsule-expansion hooks or segments secured with iris hooks to stabilize the bag) before hydrodissection or when hydrodissection fails to achieve a freely rotating nucleus. (For a video demonstrating this technique, go to eyetube.net/?v=ugomo.) Placing a capsular tension ring (CTR) at this stage can further stress zonules and often traps cortex; CTRs distribute forces to stronger zonules, but they do not avoid or resolve dislocation of the bag.
No. 8. AVOID CONVEXITY OF THE LENS DOME
When eyes exhibit poor scleral rigidity or lenticular pressure (pediatric cataract, intumescent lenses, and nanophthalmic eyes or just when the AC is crowded), administer mannitol (0.25 g/kg of intravenous push) 15 minutes preoperatively to “deturgesce” the eye and create more space. This tactic often eliminates the need for a dry vitreous tap in eyes with a shallow chamber. Use a more viscous-cohesive OVD such as Healon GV (Abbott Medical Optics) or DisCoVisc (Alcon) or a viscoadaptive agent such as Healon5 (Abbott Medical Optics). These OVDs help prevent the capsulorhexis from running downhill, because they maintain space in the absence of shearing (flow).
No. 9. BURP THE BAG TO PREVENT TAMPONADE OF THE CCC
During hydrodissection, nuclear ballottement within the capsular bag and slight pressure on the posterior lip of the incision as the fluid wave progresses prevent the adherence of the capsule’s edge to the anterior cortex. (Pressure building in the bag has the potential to blow out the posterior capsule.) Avoid creating a vigorous fluid wave in a crowded chamber. Trapped femtosecond laser gas can raise pressure, which benefits from the ballottement maneuver.
No. 10. MOBILIZE THE NUCLEUS, BUT BEWARE FIBROSIS
Hydrodelineate without hydrodissection for central posterior capsular cortical adhesions (posterior polar cataracts), or you risk tearing the taut and possibly thin posterior capsule within the pancake plaque. Fluctuations in chamber depth risk early rupture. Significant peripheral cortical-capsular fibrosis requires thorough, gentle hydrodissection. Monitor surface white cortical changes, because they change clock hours with the lens’ rotation. If fibrotic cortex bounces back when you try to turn the lens, zonules are stretching, and the lens is not free of capsular bag attachments. Multidirectional hydrodissection may resolve the adhesion. Ideally, start phacoemulsification with the nucleus free, particularly if it is dense.
No. 11. KEEP THE PHACO TIP IN THE “SAFE ZONE”
Vertical phaco chop keeps instruments inside the anterior capsulorhexis. Commence ultrasound only after visualizing the tip to reduce the likelihood of engaging peripheral capsule.
No. 12. KNOW THE LOCATION OF THE CCC’s EDGE, AND PROTECT THE VECTOR
When performing the CCC, constantly watch the location of its edge. Control the tear by regrasping to obtain appropriate vectors so that movement by the patient results in tears toward the center rather than the periphery. If the vector shifts in a direction other than intended, early recognition—within a millimeter—will help you to keep the capsulorhexis from tearing to the equator, where continuity is difficult to recover.
No. 13. SHIELD THE POSTERIOR CAPSULE FOR FINAL FRAGMENT REMOVAL
Protect the posterior capsule during the application of ultrasound energy by holding the instrument in your nondominant hand behind the phaco tip. Avoid sharp-tipped choppers, which are unnecessary even for vertically chopping brunescent lenses. Because phaco tip surge can snag the posterior capsule, reduce the fluidic settings when retrieving the final fragments, or remove the chopper entirely at those moments to deepen the chamber by eliminating leaks at the paracentesis. Complex cases often involve floppy bags that need to be kept on stretch with the equator expanded, whether with a CTR, capsular hooks (such as those from MST), or a dispersive OVD. (For a video showing circumferential disassembly, go to eyetube.net/?v=ifefa.)
No. 14. MAINTAIN THE CAPSULAR CUL-DE-SAC DURING I/A
A silicone (vs a metal) sleeve conforms to the incision and promotes a closed chamber during I/A by expanding the cul-de-sac. I find that adding a 45º bent tip will improve access to the cortex and reduce accidental grabs of the capsule, thanks to improved subincisional ergonomics. Remove subincisional material while the bag is expanded by the bulky cortex. Consider polishing rather than vacuuming the posterior capsule, especially when zonular behavior is abnormal. Use bimanual I/A or a manual “dry” technique (maintaining a normotensive cohesive OVD environment) with a syringe and cannula whenever turbulence is unacceptable, as in the setting of an open capsule or broken zonules.
No. 15. INSERT THE IOL WITH CARE
Ensure that the volume of viscoelastic suffices to keep the capsule concave or the sulcus expanded during the IOL’s insertion. Do not displace the subincisional zonules by snagging the capsule with the leading edge of the implant. Reserve single-piece lenses for placement in the bag. Position the haptics for reliable coverage by the anterior capsule. Convert any rent in the posterior capsule to a truly continuous tear to reliably secure a lens in the bag.
For sulcus placement, an IOL with a haptic diameter of 13.5 mm is ideal. At the very least, the lens should have a round anterior edge.1 Whenever possible, attempt membrane capture of the optic. This may involve capture from the sulcus through the anterior rhexis to the bag, capture from the bag through a posterior rhexis into Berger space, or a reverse capture from the bag through the anterior rhexis into the sulcus. Confirm the lens’ stability with an OVD-free tap test, or secure the IOL to the iris or sclera. Unsecured, standard bag/sulcus lenses often eventually find their way through any zonular tear. Use toric lenses if a stable bag can be obtained and centration assured.
No. 16. UNDERSTAND THE ROLES OF SUTURES AND ADJUNCT INSTRUMENTATION
I prefer nylon for corneal incisions and Vicryl (Ethicon) for sclerotomies and conjunctiva. Polypropylene degrades, especially when placed through an islet; 10–0 is best for suturing the iris to itself, to the sclera, or to haptics. Curved needles are most ergonomic for iris-to-iris suturing using Siepser knots. Straight needles are preferable for iridodialysis.
Many surgeons use 9–0 polypropylene for scleral fixation of the bag-haptic or haptics, because it is FDA approved for the eye, comes on a useful needle for ab interno techniques, and degrades more slowly. CV-8 (Gore-Tex; expanded polytetrafluoroethylene, on label for cardiovascular use only) provides superior long-term performance, especially through islets and in youthful patients, and its use is therefore becoming more mainstream. An ab externo technique with a hollow 26-gauge needle requires unbending the curve of the Gore-Tex needle with a sturdy needle holder for effective docking. Alternatively, a snare to retrieve suture ends without needles through a scleral stab may be preferable.
Intraocular instruments such as iris/capsule/suture scissors and forceps, IOL cutting scissors, and capsulorhexis forceps with a small profile are invaluable. Devices such as CTRs, capsular tension segments, and expansion hooks are often useful. Complication contingency equipment and pharmaceutical essentials include vitrector sets, chamber maintainers, triamcinolone for particulate identification of vitreous, acetylcholine and preservative-free epinephrine for iris sphincter modulation, a nonirrigating vectis, and nuclear spears.
No. 17. AVOID COLLATERAL DAMAGE TO OCULAR TISSUES
Consider fashioning a Hoffman pocket to conserve conjunctiva if using scleral fixation. Protect the endothelium at all times with a dispersive OVD. Gently stretch the iris and dissect synechiae; always preserve sphincter function and tissue. Prevent or immediately relieve iris prolapse by decompressing the chamber through the paracentesis and sweeping rather than stuffing tissue. Protect damaged fibers with an OVD. Use tangential cuts, and spiral the capsulorhexis to enlarge the CCC rather than radial relaxing incisions. Preserve capsule whenever possible. Respect the remaining zonules—which can unzip with minimal force once fellow zonules are lysed—by using chopping techniques rather than divide and conquer.
Preserve the anterior hyaloid face whenever possible. Remove displaced vitreous as well as vitreous that could adhere to anterior structures. Do not perform maneuvers through vitreous, because traction can lead to retinal tears and detachments. Reduce late scleral melts by judiciously cauterizing minimally vascularized sclera. Bury suture knots or leave the ends long and posterior to avoid erosion. Suture Tenon capsule and conjunctiva to avoid uncovered sclerotomies.
No. 18. USE PROPHYLAXIS PRUDENTLY
Prophylactically prevent infection and suppress inflammation. Although off label, intracameral antibiotics and cystoid macular edema prophylaxis combining a steroid with a nonsteroidal anti-inflammatory drug are essential for complex cases and, arguably, for routine ones as well. Hypertensive prophylaxis and prompt treatment are essential, especially if an OVD may be judiciously left behind to avoid turbulence and chamber fluctuation. Always perform a timely peripheral retinal examination in complicated cases, and/or refer the patient for subspecialty observation and treatment.
CONCLUSION
With thoughtful planning and surgery, in complex cases, you can often preserve and restore patients’ vision to the levels expected for individuals with routine cataracts.n
1. Guttman Krader C. Round anterior optic edged-IOL more superior. Ophthalmology Times. http://bit.ly/1vZXxST. Published October 9, 2013. Accessed January 6, 2015.
Lisa Brothers Arbisser, MD
• emeritus position at Eye Surgeons Associates, Iowa and Illinois Quad Cities
• adjunct associate professor at the John A. Moran Eye Center, University of Utah, Salt Lake City
• (563) 343-8896; drlisa@arbisser.com
• acknowledged no relevant financial interest


