Most patients are extremely happy with their vision after cataract surgery. Postoperatively, they take drops to help prevent infection, alleviate pain, and quell inflammation. Because many patients become frustrated by the duration of postoperative drug therapy and the frequency of instillation, different strategies have been used to help reduce the need for drops.
Pharmaceutical companies have worked toward improving drugs’ ability to penetrate the cornea. Just a few years ago, it was fairly common for patients to instill postoperative drops four times a day. Today, similar drops are only used twice or, sometimes, only once a day. For instance, the makers of the nonsteroidal anti-inflammatory drops bromfenac (Bromday, Prolensa; Bausch + Lomb) and nepafenac (Ilevro, Nevanac; Alcon) have lowered the agents’ pH to increase their lipophilicity, thereby facilitating corneal penetration. Compared with suspension formulas, difluprednate (Durezol; Alcon) is the only steroid emulsion formulation that has better bioavailability in the cornea.
A new dexamethasone-loaded punctal plug from Ocular Therapeutix safely and effectively delivers a 4-week tapered release of the corticosteroid to the ocular surface.1 Made of hydrogel and containing 400 µg of dexamethasone, the plug is inserted noninvasively through the punctum and resides within the canaliculus (Figures 1 and 2). The plug is ideal for use after cataract surgery when patients’ compliance with their postoperative drug regimen is a concern.
RESULTS OF THE PHASE 2 STUDY
In a recent phase 2 study, the dexamethasone punctal plug provided sustained reductions of both inflammation and pain for up to 1 month after cataract surgery.1 This prospective, multicenter, randomized, double-masked study evaluated 59 patients and was conducted at four private practice sites in the United States. Patients were randomized 1:1 to receive either the dexamethasone punctal plug or a placebo vehicle punctal plug, which was inserted into the inferior distal canaliculus of the operated eye. The plugs were placed during cataract surgery.
In this study, 20.7% of patients who received the dexamethasone plug had an absence of anterior chamber cells at day 8 compared with 10.0% of patients who received the placebo plug.1 Additionally, a significantly larger proportion of patients who received the dexamethasone plug compared with those who received the placebo plug reported no ocular pain at all time points throughout the study. For all time points through day 30 except on day 1, a larger proportion of patients who received the dexamethasone plug had an absence of anterior chamber cells than those who received the placebo plug. As expected, on day 1, neither group showed an absence of anterior chamber cells.
The plug is easy to insert and stays in place. In my experience, placement only takes about 30 seconds intraoperatively, and the plug contains a visualization aid for retention monitoring throughout the treatment period (Figure 3).
In the phase 2 study, the punctal plugs were visualized in all patients in both groups on the day of insertion.1 By day 30, 96.6% of the plugs containing dexamethasone and 93.1% of the placebo plugs were successfully visualized. The physicians participating in the study judged the plugs to be “easy” or “very easy” to insert in 93.3% of patients receiving the dexamethasone plugs and in 100% of the patients receiving the placebo plugs.1
SAFETY
In the study, patients reported that they could not feel the plug during insertion or postoperatively. After therapy is complete, the hydrogel resorbs and exits the nasolacrimal system without the need for removal by the physician.
PUNCTAL PLUG DELIVERY OF SUSTAINED-RELEASE DEXAMETHASONE
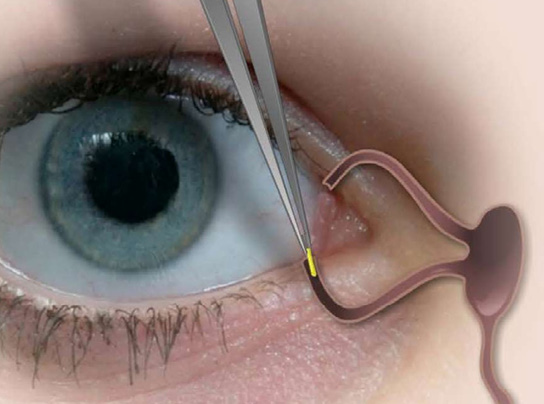
Figure 1. The punctal plug can be inserted into either the upper or lower punctum.
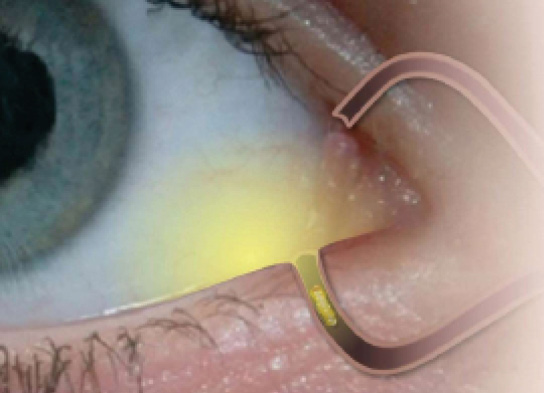
Figure 2. Over time, the drug will be delivered into the eye, and the plug will degrade and be absorbed.
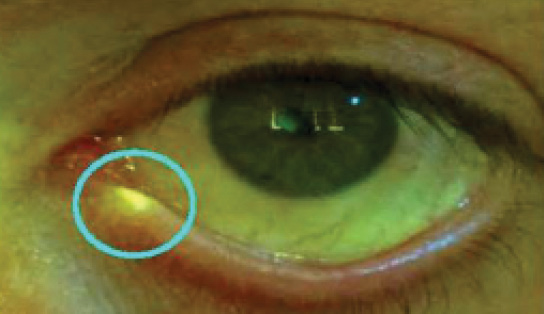
Figure 3. Fluorescent plug observed under blue light.
Safety is another benefit. More than three times as many patients in the placebo group compared with the dexamethasone group experienced an adverse ocular event in the study eye (43.3% compared with 13.8%).1 Only two treatment-related adverse ocular events were reported, and both were in the placebo group. Importantly, fewer patients in the dexamethasone group required additional anti-inflammatory (rescue) medications at every time point compared with those in the placebo group.1
The plug has entered phase 3 clinical studies to further evaluate its safety and efficacy. The prospective, multicenter, randomized, double-masked trial has already completed enrollment totaling 486 patients treated at 32 sites throughout the United States.
CONCLUSION
Patients could benefit in two important ways from surgeons’ incorporating the dexamethasone punctal plug into their therapeutic regimens. The plug could either eliminate the need for an additional anti-inflammatory drop, or it could be used concomitantly with a nonsteroidal anti-inflammatory drop to provide a dual mechanism of action for reducing inflammation and pain. A sustained-delivery steroid punctal plug may help to eliminate problems with patients’ compliance/adherence to a postoperative steroid regimen, and it should ensure that patients receive the appropriate dose for an adequate length of time to minimize postoperative inflammation and pain.n
1. Walters TR. Evaluation of sustained-release dexamethasone for safety and efficacy after cataract surgery in a multicenter study. Paper presented at: American Academy of Ophthalmology (AAO) 2014; October 19, 2014; Chicago, IL.
Shamik Bafna, MD
• practices at the Cleveland Eye Clinic, Brecksville, Ohio
• (216) 287-2060; drbafna@clevelandeyeclinic • consultant to and investigator for Alcon and Ocular Therapeutix


