
Modifications of cracking and fragmentation maneuvers permit phacoemulsification of even brunescent and mature cataracts. Using the miLoop (Carl Zeiss Meditec) can also be advantageous in these cases. As a mechanical method to divide the nucleus into two or four fragments, the device can reduce ultrasound time and cumulative dissipated energy in eyes with dense cataracts. The miLoop can divide a leathery posterior nuclear plate because the inwardly directed mechanical cutting force is applied only after the nitinol loop encircles the entire nucleus in the sagittal plane. Furthermore, the direction of the nuclear disassembly minimizes stress on the capsule and zonules, thereby reducing the risks of zonular dialysis and removal of the entire bag.
OUR APPROACH
Standard cases. The cataract is divided into two hemi-nuclei. Each segment is held with vacuum and divided again with a vertical chop technique.
Sometimes, it is difficult to visualize the posterior location of the nitinol loop through a dense nucleus. In this situation, alignment of the miLoop can be challenging. The nuclear segments produced may be of unequal size or one pole may rotate or tilt toward the cornea. Counterpressure with a second instrument through an additional paracentesis can help limit rotation.
Hypermature cataract with liquefied cortex. These eyes generally present with high intralenticular pressure, diffuse zonulopathy, a lax posterior capsule, and an absent epinuclear cushion. The comorbidities increase the risk of capsulorhexis complications, endothelial cell loss, zonular dialysis, and a ruptured posterior capsule. Moreover, once the liquefied cortex has been aspirated, the posterior capsule tends to trampoline toward the phaco tip, further increasing the risk of posterior capsular rupture.
Our preferred approach is to release the intralenticular pressure by nicking the capsule before creating a small, controlled capsulorhexis under the tamponade of a dispersive OVD. The capsular opening can be enlarged later with a relaxing cut and by then performing a double capsulorhexis. The liquefied cortex is flushed from the eye with injections of balanced salt solution, and any remaining thin, fibrous cortex is aspirated with the phaco probe. Next, a generous amount of an OVD is injected. Using a Sinskey hook placed through the paracentesis, the proximal nuclear pole is levitated and supported anteriorly. A foldable one-piece IOL is injected into the bag between the nucleus and the posterior capsule. The nucleus is repositioned and centered over the IOL optic scaffold, which prevents the phaco tip from aspirating the floppy posterior capsule and allows nuclear emulsification to be safely performed more posteriorly.
Either surgery proceeds in a traditional fashion, or a miLoop is employed to divide the nucleus into two or four pieces. In our experience, performing nucleofractis with a miLoop on top of an IOL scaffold significantly reduces capsular and zonular trauma. This is because the IOL provides a firmer stabilizing platform than the posterior capsule. The IOL scaffold also shields the posterior capsule from accidental rupture.
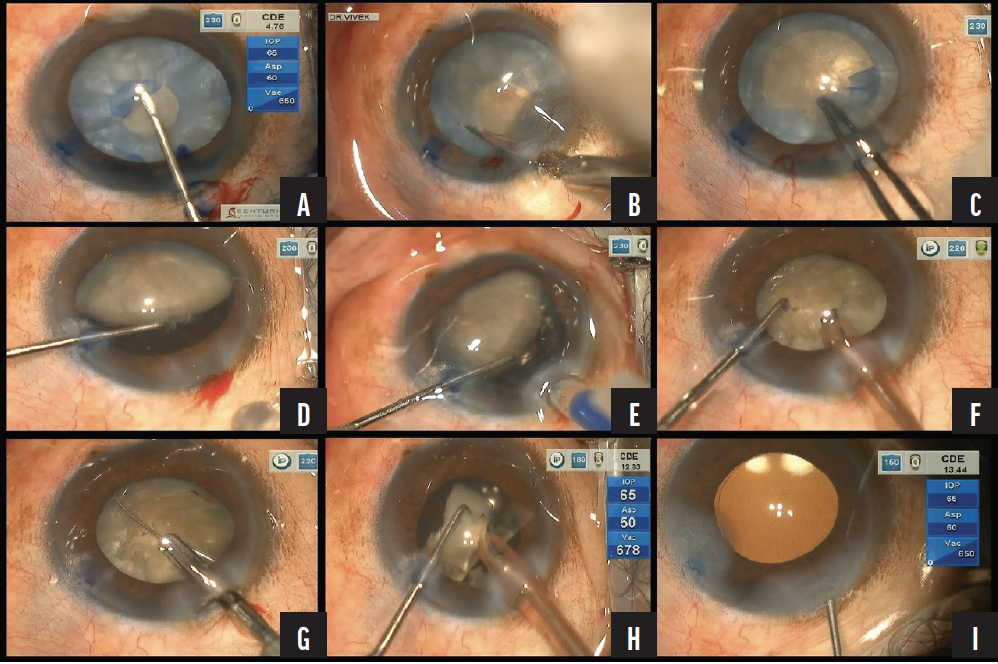
Figure. A small capsulorhexis is performed using a bent 26-gauge disposable needle mounted on a 2-mL syringe. Liquefied cortical matter is flushed from the eye with injections of balanced salt solution (A). An oblique cut is made with Vannas scissors to enlarge the capsulorhexis (B). The diameter of the capsulorhexis is widened with Utrata forceps (C). The proximal nuclear pole is lifted. A clear space is visible between the posterior surface of the nucleus and the capsular bag (D). A one-piece foldable IOL is implanted in the bag (E). Cortical and epinuclear material is emulsified (F). The miLoop divides the nucleus (G). With the IOL used as a scaffold, the fragments are emulsified (H). At the conclusion of the surgery, the IOL is well centered in the bag (I).
CASE EXAMPLE
A 63-year-old man with myopia presented with a hypermature cataract. The capsulorhexis was initiated with a 26-gauge bent cystotome, and the liquefied material was flushed from the eye with balanced salt solution. The capsulorhexis was completed using Utrata forceps and enlarged by making a relaxing cut with Vannas scissors. One pole of the nucleus was lifted, and a hydrophobic foldable IOL was placed in the bag.
The nucleus was bisected with the miLoop. Two subsequent passes of the device were required to create four quadrants, which were then emulsified with the Centurion Vision System (Alcon). The Figure shows several key maneuvers during the procedure.




