Watch the latest episodes of the Innovation Journal Club series on Eyetube.

The pace of innovation in the retina space over the past decade has been remarkable, bringing forth new drugs, novel treatment concepts, improved diagnostics, and even evolutions in how delicate operations in the posterior segment are viewed, illuminated, and performed. In episode 8 of Innovation Journal Club, renowned retinal surgeon Steve Charles, MD, FACS, FICS, FASRS, joined Dr. Singh to provide an update on two recent innovations.
A Surgical Option for Neovascular AMD
Although intravitreal anti-VEGF injections have undoubtedly improved outcomes in the treatment of neovascular age-related macular degeneration (AMD), their use comes at a cost to patients and their families, as they must be re-injected on a frequent and recurring basis. Patients are monitored with serial imaging to detect changes to the retina structure secondary to AMD, which proves additive in the overall treatment burden.
The real-world impact on patients’ lives associated with anti-VEGF therapy is a motivating factor in ongoing research into options for improving the durability of treatment options. One potential solution, Susvimo ranibizumab injection for ocular implant (formerly Port Delivery System [PDS]; Genentech), a surgically implanted drug reservoir that can be refilled with a special formulation of the anti-VEGF agent ranibizumab (100 mg/mL, Genentech), recently gained FDA approval and is starting to become more widely used in retina practice. In the interest of updating ophthalmologists about an emerging trend in retina, Dr. Charles joined Dr. Singh in this episode to review the data from the Susvimo phase 3 clinical trial.1
- In terms of efficacy, Susvimo, which was refilled at week 24 per protocol, was noninferior and equivalent to monthly ranibizumab injections. As well, 98.4% of patients in the device group required no supplemental treatment (ie, rescue therapy) in the first 24-week interval.
- The safety analysis revealed some novel signals in the Susvimo arm, including necrotizing retinitis (secondary to endophthalmitis) and implant dislocation.
According to Dr. Charles, some of the safety issues have already been addressed by the device’s sponsor; beyond that, how meticulously the surgeon attends to some of the details of the surgery, such as closure of the Tenon layer and conjunctiva, may affect the device’s safety and performance postoperatively. Indeed, Dr. Charles said he prefers to refer consultations for Susvimo to his surgical associate, despite having been trained on the device. The safety issues are not overly concerning relative to the benefits, but it may be that surgeons willing to dedicate themselves to using the device correctly are more likely to achieve the intended result.
“My associate did the clinical trials work on the PDS,” Dr. Charles said. “I trained him, and I said, ‘You’re great at this. You should do it. Let’s keep at the highest level. I’ll send my cases to you.’ I’m not afraid to do the case. I just want to see his expertise built.”
ART
Complex macular hole (MH) has historically been a challenging clinical problem. Although vitrectomy, with or without peeling of the internal limiting membrane, is effective for most cases of MH in terms of achieving anatomical closure, surgical options have remained limited for myopic MHs, refractory MHs, large to giant primary MHs, and combined MH-rhegmatogenous retinal detachments.
Autologous retinal transfer (ART), which involves harvesting a donor graft from one area of the retina and using it to close the MH, was introduced by Tamer Mahmoud, MD, PhD, from Associated Retinal Consultants in Royal Oak, Michigan, in 2016 and has since been performed by multiple surgeons around the world. Dr. Charles has worked closely with Dr. Mahmoud in developing the surgical technique. Despite initial pushback on the concept, ART has proven successful in achieving meaningful results in cases in which there was no other viable option. Data from a recent review of real-world global outcomes confirm that notion (Table).2
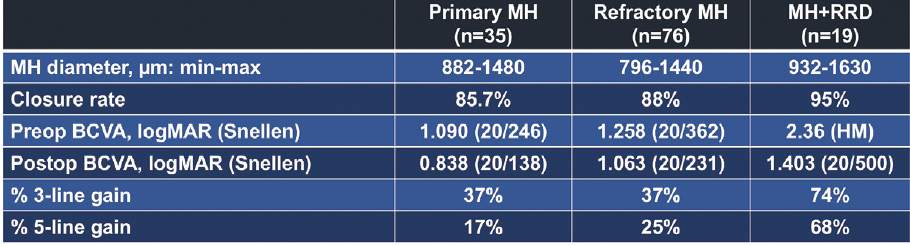
Table. Outcomes with ART from a global consortium of real-world cases.
To date, Dr. Charles has performed more than 80 cases and says he achieves closure in around 80% of cases, with about three quarters also achieving what he called “reasonable vision outcomes.” In talking with Dr. Singh, he discussed some of the finer points of ART, including his criteria for patient selection, why perfluorocarbon may be preferable as a tamponade, the procedure’s suggested mechanism of action (Figure 1), and the evidence supporting functional outcomes after ART (Figure 2).
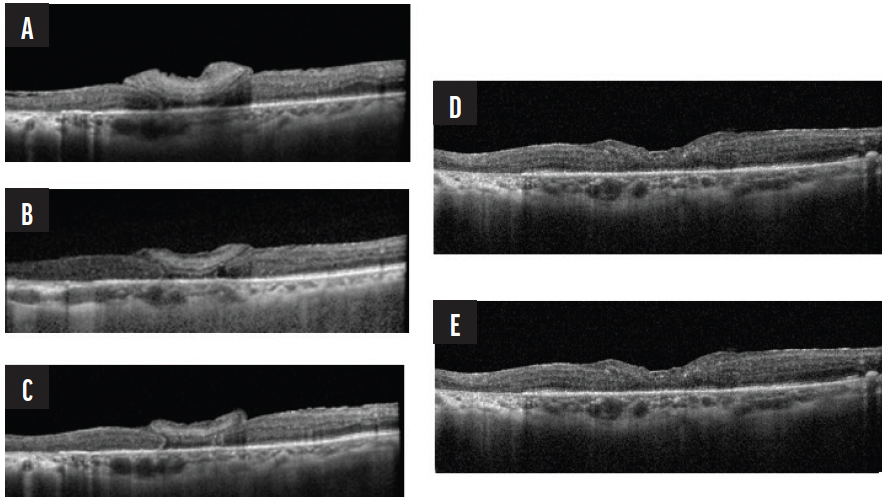
Figure 1. Although the exact mechanism of graph assimilation is unclear, Dr. Charles proposes that the graph tissue and retina demonstrate plasticity—retinal function returns as the intentionally oversized graph (seen most clearly in A) contracts to the space over time, connects with existing nerve signaling in the surrounding retina, and eventually becomes indiscernible from the rest of the retina (E).
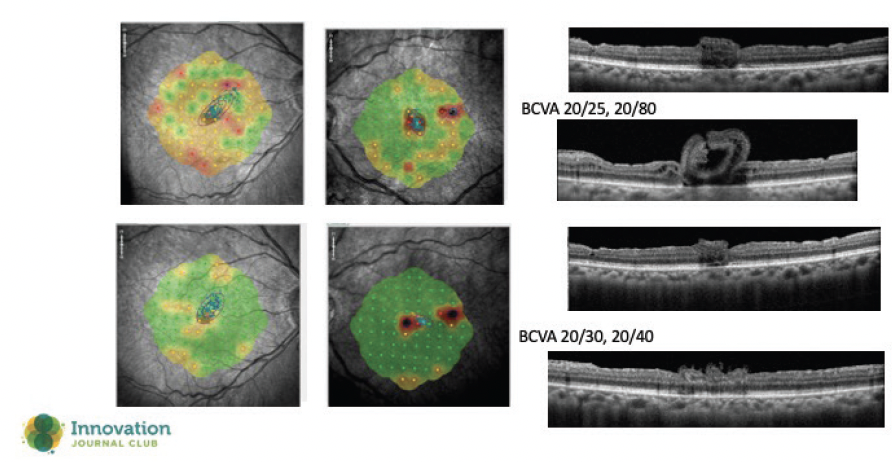
Figure 2. Microperimetry studies, performed 1 year apart, demonstrate improvement in macular function postoperatively. This finding also supports Dr. Charles’ hypothesis about retinal plasticity.
Throughout the interview, Dr. Charles highlighted the fascinating history of how ART was developed, and he suggested that it may impart some lessons about the nature of innovation itself. Despite significant skepticism and doubt from prominent members of the retina community when he first presented the concept, Dr. Mahmoud persisted because of his faith in its scientific principles. Over time, what was initially seen as unconventional has come to be accepted as a viable surgical option where none previously existed.
“The notion that the tissue is dead gone, and you can’t fix it, is simply wrong,” Dr. Charles said. “But Tamer was attacked with people saying that, and the lesson is, listen to people and let the science work and give these people a chance. That’s how things are advanced.”
“That’s how innovation occurs,” Dr. Singh added, “because they don’t give up on those ideas.”
1. Holekamp NM, Campochiaro PA, Chang MA, et al; all Archway Investigators. Archway randomized phase 3 trial of the Port Delivery System with ranibizumab for neovascular age-related macular degeneration. Ophthalmology. 2022;129(3):295-307.
2. Moysidis SN, Koulisis N, Adrean SD, et al. Autologous retinal transplantation for primary and refractory macular holes and macular hole retinal detachments: the global consortium. Ophthalmology. 2021;128(5):672-685.


