


Of the various techniques for nuclear disassembly, manual fragmentation provides surgeons with the distinct advantage of reducing the amount of ultrasound energy typically required to sculpt the nucleus with traditional divide and conquer or stop and chop methods. Decreasing the amount of ultrasound energy used during surgery can translate to less postoperative corneal edema and better visual outcomes for patients. The following case examples illustrate two of our preferred fragmentation techniques for different lens densities.
CASE NO. 1: USE OF an AKAHOSHI PRECHOPPER
Presentation. A 74-year-old man was referred for a cataract evaluation. He reported experiencing glare when driving at night and developing mildly blurry vision in both eyes over the past several years.
On presentation, the patient’s BCVA was 20/30 OD and 20/40 OS, which decreased by approximately 2 Snellen lines with the brightness acuity tester. A 2+ nuclear sclerotic cataract and trace cortical and posterior subcapsular changes in each eye were found during the slit-lamp examination. The eyes were otherwise healthy. The risks and benefits of cataract surgery and IOL options were discussed with the patient, and he was scheduled for cataract surgery on both eyes.
Technique. A soft to moderately dense lens nucleus can be easily divided with a universal Akahoshi prechopper (scan the QR code for a video of the prechopper). The paddle-shaped blades of this cross-action instrument are embedded in the nucleus and then splayed to vertically prechop and crack the nucleus (Figure 1). Several instrument models are available; their primary differences are blade thickness and sharpness, paddle size, and the extent (width) to which the jaws open. Depending on blade thickness, prechoppers can be used to divide dense nuclei, but the risk of damage to the posterior capsule increases with a sharp instrument if the lens is soft.
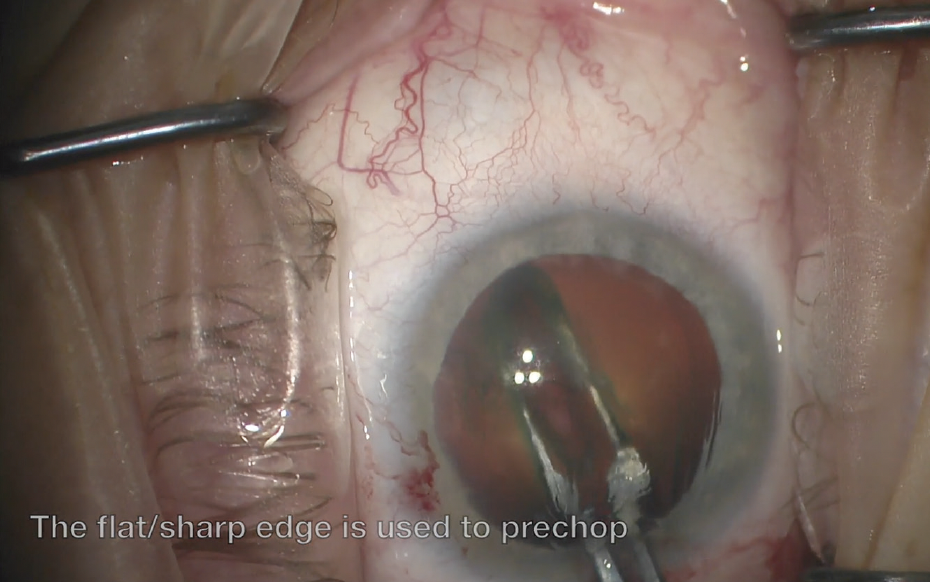
Figure 1. The paddle-shaped blades of an Akahoshi prechopper are embedded in the nucleus and then splayed to vertically prechop and crack the nucleus.
A continuous curvilinear capsulorhexis and hydrodissection are performed, followed by placement of a small amount of an OVD over the center of the lens to clear cortical material and improve visualization of the initial crack. The slow application of consistent pressure with the straight paddle edge is required to embed the prechopper in the nucleus and is essential while the instrument’s jaws are opened. Without continuous pressure or if the paddles are not embedded deeply enough, cortex and epinucleus can shave off the top of the nucleus, rendering the prechop unsuccessful. If an initial crack does not propagate the entire length or depth of the lens, the depth or position of the instrument can be adjusted to complete disassembly. During this step, the blunt curved edge of the paddles should point downward, and the surgeon’s hand should be flat to prevent damage to the capsular bag (scan the QR code for a video demonstration of this surgical step).
The Akahoshi prechopper can be used to rotate the lens and further divide the nucleus into quadrants if desired. In our experience, use of the device can shorten the duration of cataract surgery and decrease the amount of phaco energy required. The instrument can be particularly useful for first-year residents who are not yet comfortable with bimanual divide and conquer disassembly.
CASE NO. 2: USE OF THE MILOOP
Presentation. A 64-year-old was referred for an evaluation of the cataract in his left eye. The patient was previously scheduled for surgery but was lost to follow-up during the COVID-19 pandemic. He stated that the vision in the left eye is much worse now than when he was last seen.
On presentation, his BCVA was count fingers at 6 feet OS, and his pinhole visual acuity in this eye was 20/400. A 4+ brunescent cataract was evident. There was no view to the retina, so B-scan ultrasonography was performed, which demonstrated a normal posterior segment. After discussion, the patient elected to proceed with surgery.
Technique. By mechanically cleaving the nucleus into segments, the miLoop (Carl Zeiss Meditec) eliminates the need for high levels of ultrasound energy that would otherwise be required to segment the lens for quadrant removal. (Scan the QR code for a video demonstration of surgery with the miLoop.) The memory-shaped nitinol loop is designed for full-thickness fragmentation regardless of nuclear grade. The loop uses centripetal rather than centrifugal forces, which place less overall stress on zonules than cracking.
It is important for the surgeon to be aware of the positions of the distal and proximal ends of the nitinol loop while the device is inside the eye. The first-generation device lacks a hub to prevent overinsertion into the main wound, and the fully expanded loop can put stress on the nasal zonules if the loop is not centered properly. The second-generation device uses a small, silicone, accordion-style hub to limit the depth of insertion into the eye. The surgeon, however, must not blindly rely on the hub to guide the depth of insertion. The nitinol loop can nasally subluxate a lens if the loop is pushed too far into the eye.
The loop should slide underneath the anterior lens capsule initially. OVD may be used to gently vault this edge if needed. When the loop is fully extended underneath the anterior capsule, the device is rotated in a righty-tighty fashion to orient the loop vertically (Figure 2). To ensure that the loop is well positioned, it can be overrotated slightly to allow the lag of the nitinol loop to catch up posteriorly and then be rotated back to center so that the lens can be evenly divided. A second instrument may be used to prevent prolapse of the lens segments as the loop is withdrawn.
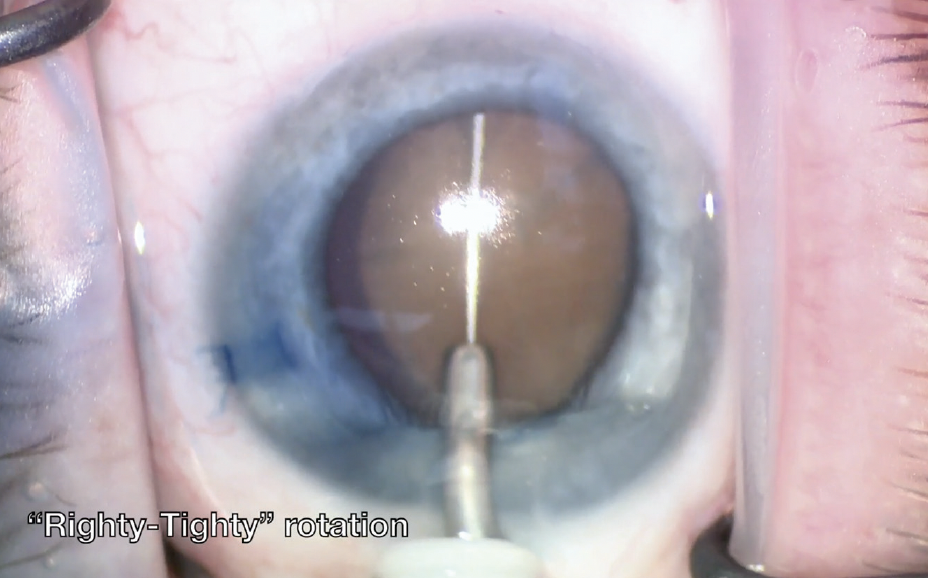
Figure 2. The miLoop is rotated in a righty-tighty fashion to orient the loop vertically.
CONCLUSION
The Akahoshi prechopper and miLoop are both useful tools for manual nuclear fragmentation. Perfecting these techniques can reduce the amount of ultrasound energy required for a variety of lens densities and elevate postoperative outcomes.




