CASE PRESENTATION
A 70-year-old man was scheduled for laser cataract surgery and implantation of a toric monofocal IOL to correct a low amount of astigmatism. The patient had a history of adult-onset diabetes that was controlled with oral medication, controlled hypertension, and hypercholesterolemia.
The course of cataract surgery was uneventful until the posterior capsule ruptured during polishing (Figure 1). How would you proceed, and which IOL would you implant?
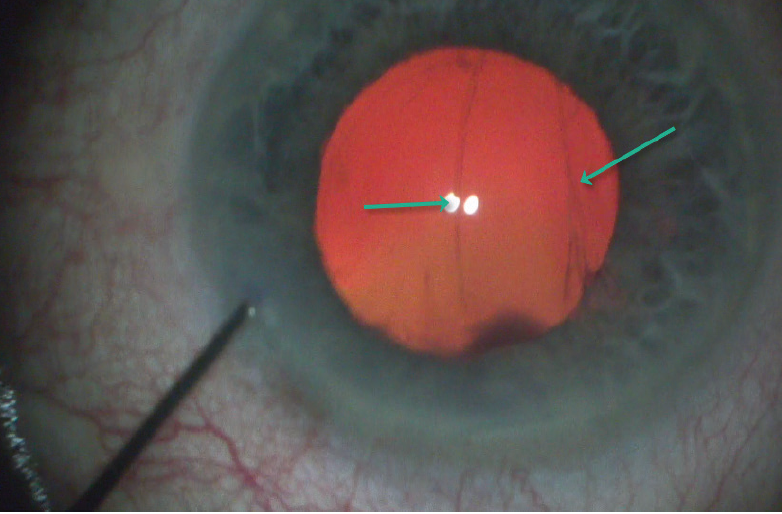
Figure 1. The posterior capsule ruptures during polishing.
—Case prepared by P. Dee G. Stephenson, MD, FACS

KENDALL E. DONALDSON, MD, MS
We cataract surgeons spend a lot of time counseling patients on lens options and postoperative expectations. It is frustrating and disappointing when we feel that we may not be able to deliver the very best outcome and meet the patient’s expectations. The current situation sounds unpredictable. Sometimes, however, complications can be anticipated, and they should be discussed in advance of surgery in high-risk cases such as a patient with a traumatic cataract and loose zonules or a patient with pseudoexfoliation syndrome and small pupils.
Diabetes increases this patient’s risk of experiencing cystoid macular edema (CME) after vitreous loss, so it is worth considering optic capture of a three-piece IOL. When a small tear occurs in the posterior or anterior capsule, I may still implant a toric IOL in the capsular bag. In this case, however, the capsular tear is large, and there is a significant risk of vitreous prolapse and subsequent CME. I would carefully inspect the hyaloid face to determine if it is intact. If I had reason to suspect vitreous prolapse, I would perform either an anterior vitrectomy or a limited pars plana vitrectomy and use triamcinolone acetonide to identify residual vitreous.
The opening in the posterior capsule could expand as an IOL is placed within the capsular bag. I would therefore instead implant a three-piece monofocal spherical IOL in the ciliary sulcus with optic capture. The lens will tamponade potential vitreous prolapse.
Postoperatively, I would explain to the patient that a weakness found within the capsular bag rendered placement of a toric IOL inadvisable, and the money he paid for astigmatism correction would be refunded. I would consider performing laser vision correction if the residual astigmatism is significant but only after observing the patient for 3 months for the development of CME. If macular OCT reveals irregularities during this time, I would have him continue administering a topical NSAID for an extended postoperative period.

SOOSAN JACOB, MS, FRCS, DNB
I would stabilize the anterior chamber by injecting a dispersive OVD before withdrawing the cataract instrument in order to prevent an extension of the posterior capsular tear and vitreous prolapse. Because cortical aspiration is almost complete, I would check for the presence of prolapsed vitreous by instilling preservative-free triamcinolone acetonide. A pars plana vitrectomy can be performed to remove prolapsed vitreous. It is simpler to provide infusion via the anterior chamber through an irrigating cannula held through the paracentesis or, if the surgeon needs both hands free, with a trocar anterior chamber maintainer.
Figure 1 does not clearly show the upper border of the tear. Attempting in-the-bag implantation carries an increased risk of tear extension, vitreous loss, an unstable IOL, or a dropped IOL. This risk is greater for a toric IOL because surgical manipulation of the lens is required to align it to the correct axis after implantation. Additionally, with a toric IOL, all OVDs must be thoroughly removed at the conclusion of surgery, including from under the IOL. I would therefore implant a three-piece monofocal IOL in the sulcus and capture the optic behind the well-centered laser capsulotomy.
Because this patient is 70 years of age and the amount of astigmatism is low, I would correct his residual astigmatism with a laser corneal procedure after the refraction stabilized. Other options include limbal relaxing incisions and the placement of a Sulcoflex toric IOL (Rayner, not available in the United States).

MICHAEL E. SNYDER, MD
The lens material has been fully evacuated, and a large posterior capsular tear has occurred. My first step would be to instill an OVD in the anterior chamber and pressurize the anterior chamber before removing the polishing (I/A) handpiece. Doing so will prevent the vitreous face from coming forward. A posterior capsular tear and vitreous prolapse are two distinct events; the latter occurs only when the anterior chamber pressure becomes lower than the vitreous pressure. When pressurizing the anterior chamber, I prefer to instill an OVD that has both retentive and cohesive qualities such as DisCoVisc (Alcon). Timing, however, is of the utmost importance in this setting, so the best OVD to instill is the one that is already on the sterile field. This is not the time to have the circulating nurse leave the room and go down the hall to search for a specific OVD in the storage refrigerator, only to reappear after the clinical situation has changed.
In Figure 1, the ends of the tear seem to extend beyond the 5-mm capsulotomy, so converting this tear into a posterior capsulorhexis probably is not realistic. Nonetheless, the anterior capsulotomy remains intact. I would select a three-piece posterior chamber IOL that has an optic larger than the capsulotomy opening and adjust the IOL power if there is a difference from the A-constant of the originally intended implant. I would inject the IOL into the ciliary sulcus and then capture the optic through the anterior capsulotomy, trapping the IOL into position. Because the optic will be in the plane of the capsular bag or Berger space, there is no need to adjust the IOL power for expected issues with effective lens position, unlike with passive sulcus placement. The barrier between the anterior and posterior chambers will be reestablished.
If attention is paid to positive anterior chamber pressure, vitreous should not become an issue. If a brief lapse in pressure control occurs and vitreous is encountered, then a limited anterior vitrectomy via a pars plana approach would be an efficient way to remove the offending vitreous gel.1
With the globe still pressurized, I would use a guarded blade with 550-µm depth to create a limbal relaxing incision just inside the conjunctival insertion on (what I hope is) the still identifiable steep axis. If axial landmarks are not reliable, then I would defer this step and address astigmatism later with laser vision correction, if the patient so desires.
The OVD can be aspirated in the usual manner, and the incisions sealed with hydration. I prefer to perform an intracameral injection of carbachol (Miostat, Alcon) at the end of a case such as this one in which OVD is invariably retained behind the IOL. The carbachol will ensure good IOP control during the early postoperative period. Acetylcholine (Miochol-E, Bausch + Lomb), in contrast, has a short half-life and provides little benefit in terms of IOP.

WHAT I DID: P. DEE G. STEPHENSON,MD, FACS
The anterior hyaloid face had not been violated, and no vitreous presented. After filling the bag with an OVD to control the setting, I used a Kuglen hook to push back the iris so as to see the extent of the posterior tear. It did not extend to the equator. After placing a capsular tension ring to maintain the integrity of the bag, I used the ORA System (Alcon) to obtain an aphakic reading and the axis and magnitude of astigmatism.
Because the bag seemed to be intact except for the posterior tear, I decided to place a one-piece enVista toric IOL (MX60ET, Bausch + Lomb) in the bag. I could have implanted a three-piece IOL with optic capture or sulcus fixation, but I could see the end of the tear. Because of the aspheric design of the MX60ET lens, the power remains equal from the center to the edge of the optic, and a small amount of tilt and decentration will not affect quality of vision.2 I also find that the lens opens in a slow and controlled fashion. The IOL was placed without sequalae, and the lens lined up nicely with the steep axis and appeared to be stable despite the posterior capsular tear (Figure 2).
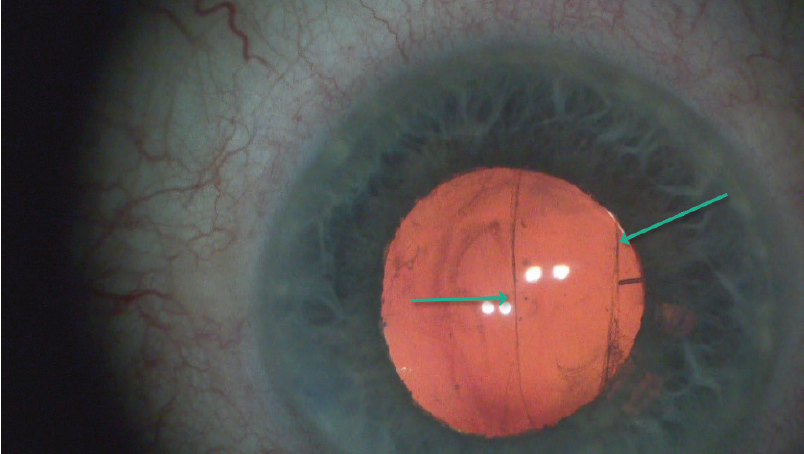
Figure 2. A one-piece enVista toric IOL in the bag.
Postoperatively, UCVA was 20/25, and BCVA was -0.25 +0.50 x 24º = 20/20+2. Eight months after surgery, the patient presented with a nasally displaced IOL. UCVA remained 20/25. On examination, no vitreous was present, and there was no iridodonesis. Because the patient was asymptomatic and had an aspheric IOL that induces no aberrations when tilted or decentered, I chose not to intervene further (Figure 3). The patient continues to be asymptomatic and to enjoy freedom from spectacles for distance visual acuity.
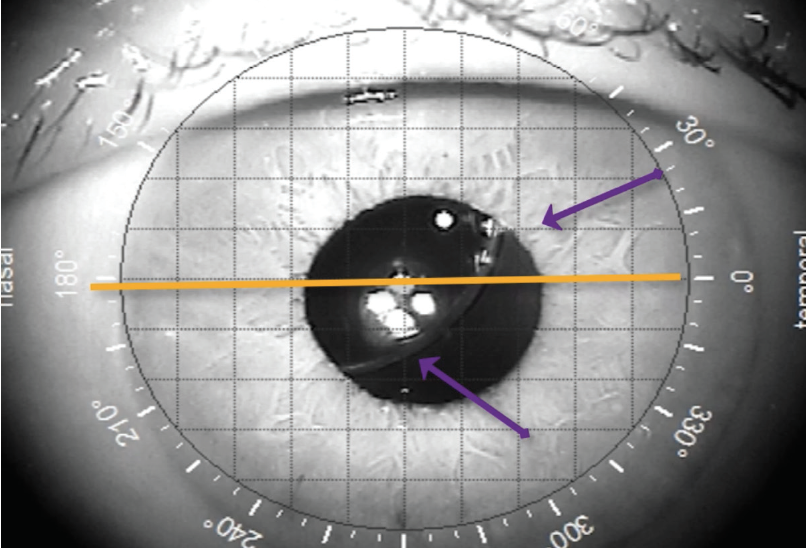
Figure 3. The IOL has dislocated 8 months after surgery.
1. Thornton IL, McMains BK, Snyder ME. Long-term safety and efficacy of single-port pars plana anterior vitrectomy with limbal infusion during anterior segment surgery. J Cataract Refract Surg. 2018;44(7):878‐883.
2. Stephenson PDG. Retinal image simulations of aspheric toric IOLs under conditions of tilt and decentration. Poster presented at: AAO Annual Meeting; October 12-15, 2019; San Francisco.




