
Mask wearing is common practice in the age of COVID-19, and dry eye disease (DED) is an increasing patient complaint. This increase is possibly due to turbulent air from mask-associated dry eye (MADE), a term coined by Darrell E. White, MD, as well as increased screen time and the beauty, skincare, and cosmetics practices of our patients.
This article explores five topics in eye cosmetics that can contribute to the incidence and severity of DED.
No. 1: Lipstick index is out, mascara effect is in. During the Great Recession of 2008, lipstick sales were up 11% from previous years.1 It turns out that purchasing and wearing an inexpensive tube of lip color was an affordable luxury that made wearers feel better. This trend, termed the lipstick index in a phrase coined by Leonard Lauder, Chairman of Estée Lauder, highlighted the idea that, in turbulent times, individuals find joy in small things.
With today’s mask-wearing practices, cosmetics such as lipstick, foundation, and blush are easily smeared. As a result, attention has turned to the eyes, and the use of mascara, eyelash extensions, eyeliner, and eyeshadow is on the rise. Mica and glitter-laden highlighters finish this eyes-only makeup look.
But use of these products can have deleterious side effects. We have all treated patients complaining of increased dry eye symptoms who do not realize that their makeup is to blame. Even men wear makeup today (see Guyliners, Job Interviews, and Heads of State). When advising patients on the safety of the ocular surface products they use, ophthalmologists and optometrists cannot rely on marketing claims such as hypoallergenic, natural, vegan, sensitive eye formula, suitable for contact lens wearers, suitable for dry eye, or even ophthalmologist-tested.
Guyliners, Job Interviews, and Heads of State
It is important to know that makeup for men is a rapidly growing social media topic and an increasingly accepted practice. Examples of beauty industry growth in men’s skincare and cosmetics include these:
- An entire section in Kiehl’s (the under-eye cream has nearly a dozen known ocular surface–offending ingredients);
- Abundant makeup tutorials for men on YouTube and Instagram;
- Increasing numbers of men wearing makeup on job interviews;1 and
- Makeup use by public figures such as Prime Minister Emmanuel Macron of France, who reportedly spends more than $300 a day on professional makeup artist services.2
1. Fujikawa M, Bhattacharya S. How to wear makeup for your job interview—men’s edition. The Wall Street Journal. March 20, 2019. https://www.wsj.com/articles/how-to-wear-makeup-for-your-job-interviewmens-edition-11553086802. Accessed July 22, 2020.
2. Gallagher J. More men are wearing makeup than you think—here’s why. The Wall Street Journal. April 13, 2018. https://www.wsj.com/articles/more-men-are-wearing-makeup-than-you-thinkheres-why-1523626771. Accessed July 22, 2020.
First, let’s unmask the dangers of mascara. Mascara that doesn’t smear with artificial tear use but that can be removed without harsh chemicals is difficult to find. When you can find them, such products are laden with ocular surface–unfriendly ingredients. Even formulas that rely on alternative preservative systems (different from traditional paraben and formaldehyde-donating preservative systems) are tested only for a short period, meaning sterility is not reliable beyond that time. Additionally, in the protocols I have read about, the cosmetic is inoculated once and then researchers wait for microbial growth; this does not reflect real-world use of daily application and inoculation. Patients should be encouraged to discontinue the use of products that are long past their suggested replacement dates.
One way to improve the lengthening effect of mascara is to add nylon fiber. However, nylon fibers is an ingredient that should be avoided. A 2018 case report described a woman in her 40s who presented with eye redness, irritation, and pain. She was using Moodstruck 3D Fiber Lashes (Younique) and had six 1-mm black nylon fibers lodged in the subconjunctival space with adjacent inflammatory reaction and fibrosis. Surgical removal was required.2 A $3.3 million class action settlement was brought against the company in 2019 for false claims of using natural lengthening fibers (nylon is man-made).
A wide variety of nylon fiber additives (listed as nylon66) are used in mascara. All should be avoided. Nylon has sharp edges on scanning electron microscopy; it is stiff and would have a spear-like effect at short lengths (explaining the case above); and when it is combined with other mascara chemicals including propylene glycol and preservatives, the potential for inflammation and fibrosis increases.
No. 2: Ditch the lengthy lashes. Those long, lush eyelashes that are so widely desired may come at a price. In addition to chemical-laden mascaras, the look of embellished eyelashes can be achieved with the use of prostaglandin analogue (PGA)–laced over-the-counter (OTC) eyelash growth serums, natural defense–compromising magnetic eyelashes, chemically dangerous eyelash perms, and clinically unhygienic eyelash extensions (Figure). None of these is safe. It is important for ophthalmologists to understand the pitfalls of each of these methods in order to counsel our patients. I cover a few in this article; I will tackle others in a subsequent series of articles in CRST.
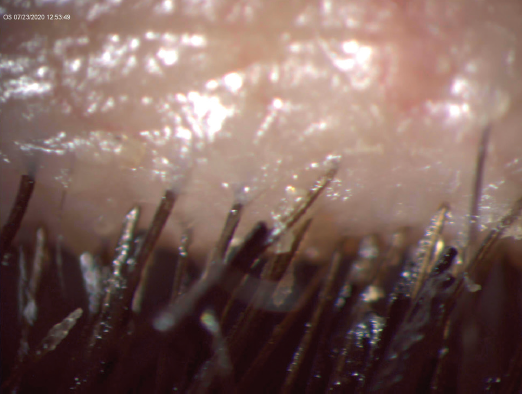
Figure. Significant blepharitis in a dry eye patient with eyelash extensions.
No. 3: Know the hairy aspects in glaucoma medication use. PGAs constitute an important class of glaucoma medications, but they have side effects, including permanent pigment changes in the iris, periorbital tissues, and eyelids; periorbital fat atrophy; intraocular inflammation; macular edema; and eyelash growth.3 A longitudinal, masked, multicenter comparative study reported that 64% to 76% of patients with elevated IOP had at least one adverse effect, mainly ocular hyperemia (ie, red eye) with daily PGA use over 12 weeks.4
Red eye is compounded by the known association of PGAs with meibomian gland dysfunction (MGD). In a prospective, cross-sectional study, 92% of glaucoma patients using a PGA had obstructive MGD, compared with 58% among patients taking other classes of IOP-lowering medications.5
Further, approximately 40% of postchemotherapy patients taking bimatoprost 0.03% for eyelash hypotrichosis had at least one adverse event at 16 weeks, and 47% had at least one adverse event after 1 year.6 These events included conjunctival hyperemia, eyelid pruritus, pigmentation, hyperemia, punctate keratitis, and DED.
No. 4: Beware of OTC eyelash growth serums. Knowing the side effects of prescription PGAs can help you to recognize the same patterns in patients who are using unregulated, drug-laced OTC eyelash growth serums. Nearly one-third of OTC eyelash growth serums contain a PGA, and some do not disclose it on the ingredients label,7 which poses significant issues for consumers (not to mention, PGAs are pregnancy category C). The frequent lacing of OTC eyelash growth serums with PGAs occurs despite FDA warning letters to cosmetics manufacturers for “misleading statements regarding the product’s safety and [failure] to reveal material facts with respect to consequences that may result from the use of the product.”8
In a survey on OTC eyelash growth serum use, among 154 respondents, 43% said they had stopped using an eyelash growth serum, and, of these, 67% reported that the reason for dropout was side effects (burning, stinging, itching, eyelid pigment change, eyelash loss, sunken eyes).9 If you see any of the telltale side effects of PGA use, ask patients if they are using an eyelash growth serum. If they are, advise them to stop.
No. 5: There is a difference between real science, secret science, and pseudo-science. Prescription medications require transparent, robust, placebo-controlled studies to gain FDA approval. Head-to-head randomized studies in identical cohorts allow us to make claims such as safer, noninferior, or even better than.
Unfortunately, there is no such data transparency or basis for clinical safety and efficacy claims in cosmetics.10,11 The days of ocular irritancy testing in Draize rabbit models are over, thankfully. These inhumane tests have been replaced by in vitro EpiOcular MatTek assays designed to test ocular irritancy from industrial and household chemicals, personal care products, and cosmetics. In cosmetics studies, a liquid cosmetic is exposed to cultured human keratinocytes for 30 to 254 minutes and assayed for cell death against positive and negative controls (not a typical time frame in expected human use of 8, 16, or 24 hours of wear). If 60% or more of the cells survive that short in vitro, non–real world exposure, the product is considered nonirritating.12
Said in another way, up to 40% of the cells can die from exposure to the cosmetic and the product will still be reported as nonirritating.
This raises significant concerns around cosmetics safety, especially in our ocular surface disease patients. The lab assay protocol versus real-world exposures may help to explain cosmetics users survey data indicating higher Standard Patient Evaluation of Eye Dryness (SPEED) questionnaire scores in patients who do not remove their makeup.13 Cosmetics companies are not required to perform EpiOcular testing; and if they do, the data results are not made public.
Cosmetics performance claims are based on subjective consumer surveys with pooled satisfaction data sets to artificially elevate the percentages. Performance claim studies are performed by cosmetics marketing companies with explicit designs to support cosmetics marketing claims.10 These cosmetics clinical studies are nonscientific marketing studies that do not confer assurances of ocular surface safety because they do not even ask questions around ocular safety and side effects. Even in the European Union, where cosmetics safety regulations are more up to date, cosmetics organizations develop consumer use surveys (often misrepresented as clinical trials) to “undergo the scrutiny of the advertising standards agency” and “instill consumer’s confidence.”10 Subjective performance does not equal consumer safety. We expect to see safety data for the prescriptions we write, so shouldn’t we expect it for the eye cosmetics we recommend?
Even cosmetics safety-in-use studies (ophthalmologist-tested) lack scientific rigor, scientific methodology and transparency. I’ve searched for months and have been unable to find industry standards or FDA regulations for making cosmetic label claims such as safe for contact lens wearers and appropriate for dry eye. These claims as well as suitable for sensitive eyes are based on nontransparent protocols from ophthalmologist testing companies, one of which has had indictments of data fraud.14
Cosmeceutical brands that claim the space between cosmetics and pharmaceuticals have an even higher burden of safety, transparency, performance, and consumer safety expectations.15 In a 2017 study analyzing cosmetic-related adverse events reported to the FDA, researchers suggested that a form of premarket approval should be considered for such products.16 But since cosmeceuticals are neither recognized nor defined under FDA law, the legal requirements in this blur zone are the same as those for the weak FDA cosmetics regulations. Cosmetics companies do not perform studies with pharmaceutical rigor because they don’t have to,11 and yet ocular complications from their products are seen in our clinics often, and the complications are well described in the literature.17 Also, cosmetics are not allowed to make treatment claims,11 so we should view cosmetics claims of suitable for dry eye with healthy skepticism. Finally, when you see cosmetics claims of better than or safer than, ask yourself, “Better and safer than what?”
1. Prance-Miles L. Goodbye lipstick effect, hello mascara index — How COVID-19 may have changed the beauty landscape for good. Global Cosmetics News. https://www.globalcosmeticsnews.com/goodbye-lipstick-effect-hello-mascara-index-how-covid-19-may-have-changed-the-beauty-landscape-for-good/. Accessed July 20, 2020.
2. Dowlut MS, Ahmed Y, Knox A. Ocular inflammation associated with fibers from eyelash extensions. JAMA Ophthalmol. 2018;136(1):e175723.
3. Zioptan [package insert]. Merck Sharp and Dohme. 2012. accessdata.fda.gov/drugsatfda_docs/label/2012/202514s000lbl.pdf. Accessed July 22, 2020.
4. Parrish RK, Palmberg P, Sheu WP, for the XLT Study Group. A comparison of latanoprost, bimatoprost, and travoprost in patients with elevated intraocular pressure: a 12-week, randomized, masked-evaluator multicenter study. Am J Ophthalmol. 2003;135:688-703.
5. Mocan MC, Uzunosmanoglu E, Kocabeyoglu S, Karakaya J, Irkec N. The association of chronic topical prostaglandin analog use with meibomian gland dysfunction. J Glaucoma. 2016;25(9):770-774.
6. Wirta D, Pariser DM, Yoelin SG, et al. Bimatoprost 0.03% for the treatment of eyelash hypotrichosis: a pooled safety analysis of six randomized, double-masked clinical trials. J Clin Aesthet Dermatol. 2015;8(7):17-29.
7. Pharmaceutical ingredients in one out of three eyelash serums [press release]. Swedish Medical Products Agency. April 15, 2003. dr-jetskeultee.nl/jetskeultee/download/common/artikel-wimpers-ingredients.pdf. Accessed July 22, 2020.
8. Warning letters address drug claims made for products marketed as cosmetics. US Food and Drug Administration. June 26, 2019. https://www.fda.gov/cosmetics/warning-letters-related-cosmetics/warning-letters-address-drug-claims-made-products-marketed-cosmetics. Accessed July 20, 2020.
9. Doll T, Schwartz S, Hom M, et al. Over the counter eyelash growth serum use: Self-reported pervasiveness and user satisfaction. Poster presented at: The Association for Research in Vision and Ophthalmology; 2020.
10. Clinical testing of cosmetics and personal care products. Intertek. https://www.intertek.com/consumer-healthcare-trials/. Accessed July 20, 2020.
11. FDA authority over cosmetics: how cosmetics are not FDA-approved, but are FDA-regulated. US Food and Drug Administration. July 24, 2018. https://www.fda.gov/cosmetics/cosmetics-laws-regulations/fda-authority-over-cosmetics-how-cosmetics-are-not-fda-approved-are-fda-regulated. Accessed July 20, 2020.
12. EpiOcular eye irritation test (OCL-200-EIT). MatTek. mattek.com/wp-content/uploads/EpiOcular-Eye-Irritation-Test-EIT.pdf. Accessed July 20, 2020.
13. O’Dell LE, Periman LM, Sullivan AG, Halleran CC, Harthan JS, Hom MM. An evaluation of cosmetic wear habits correlated to ocular surface disease symptoms. Poster presented at: The Association for Research in Vision and Ophthalmology Annual Meeting; May 6-11, 2017; Baltimore.
14. Owner Of AMA, A Rockland based consumer products testing company, arrested for fraud scheme involving fabricated test results [press release]. US Department of Justice. August 9, 2019. https://www.justice.gov/usao-sdny/pr/owner-ama-rockland-based-consumer-products-testing-company-arrested-fraud-scheme. Accessed July 31, 2020.
15. Pandey A, Jatana GK, Sonthalia S. Cosmeceuticals. In: StatPearls. StatPearls Publishing; 2020.
16. Kwa M, BA, Welty LJ, Xu S. Adverse events reported to the US Food and Drug Administration for cosmetics and personal care products. JAMA Intern Med. 2017;177(8):1202-1204.
17. Wang MT, Craig JP. Investigating the effect of eye cosmetics on the tear film: current insights. Clin Optom (Auckl). 2018;10:33-40.




