Minimally invasive glaucoma surgery (MIGS) has transformed the way we approach glaucoma becoming a preferred management strategy for patients with open angle glaucoma. MIGS is particularly appealing to patients undergoing concurrent cataract surgery. The goal of all glaucoma surgery is to lower eye pressure to prevent or reduce damage to the optic nerve. The introduction of the OMNI Surgical System (Sight Sciences) allows surgeons to intervene earlier in the disease state. The OMNI indications for use do not restrict utilization by disease severity. It can be used to treat mild, moderate, low tension, ocular hypertension on medication, and advanced glaucoma. Another benefit of the device is that it is not limited for use combined with cataract surgery only but can be used as a standalone glaucoma procedure.
All Three Points of Resistance in the Conventional Outflow Pathway
Glaucoma is a disease of fluid flow obstruction. I often use the analogy of a clogged sink drain to explain glaucoma to my patients. We don’t know whether the blockage is at the drain screen or under the sink in the “P trap” or in the pipes under the house. Testing for the specific site of blockage does not exist today. OMNI will flush the entire system from the sink to the street.
We know that traditional glaucoma incisional surgeries (trabs and tubes) offer high efficacy but at the price of high rates of complication, failure, and reoperation.1 A challenge is that many patients may need to receive these types of surgeries at some point in the future. Utilizing OMNI spares conjunctiva and sclera that may be involved with a traditional surgery.
One of the surprising discoveries from performing OMNI is that you can often start channeling Schlemm’s canal with the microcatheter without making a large incision in the trabecular meshwork (TM). This leads to minimal hemorrhage and, in many cases, no hemorrhage. In my practice, OMNI procedures are producing excellent outcomes to reduce medication burden and/or reduce or maintain acceptable IOP.
MY First OMNI Case: Complex Cataract/Glaucoma Case Using OMNI
An 85-year-old Hispanic female presented with cataract and a history of mild to moderate open angle glaucoma cataract grade 2 NS, grade 3 ASC with vertical C/D 0.8 inferior notch and Ta 12 mm Hg on fixed combination brinzolamide/brimonidine twice daily.
Patient Goals
The goals we set for this patient were to improve vision with glasses and maintain her IOP control without the need for medication. I performed a manual basic lens replacement and OMNI viscocanaloplasty superior 180°.
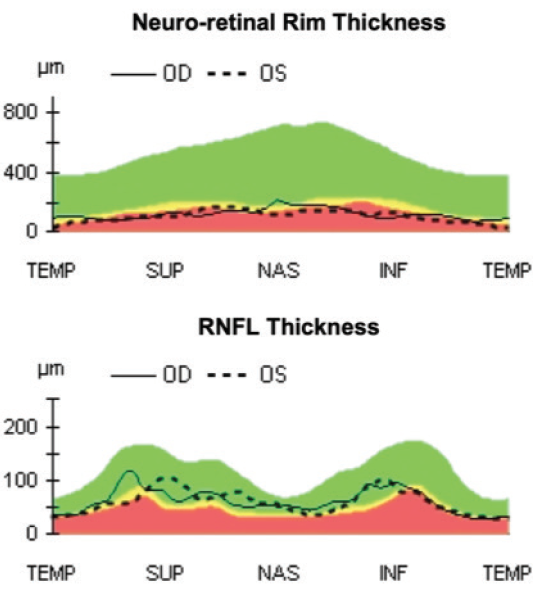
Figure 1. Nerve fiber layer analysis demonstrated mild to moderate defects.
Patient Presentation
- BCVA of 20/30 with glare reduction 20/60
- IOP of 12 mmHg
- Nuclear sclerosis was grade 2, anterior subcapsular grade 3
- C/D ratio was 0.8 with thin inferior rim
- Nerve fiber layer analysis demonstrated mild to moderate defects (Figures 1, 2)
- Humphrey visual field demonstrated mild superior arcuate defects (Figure 3)
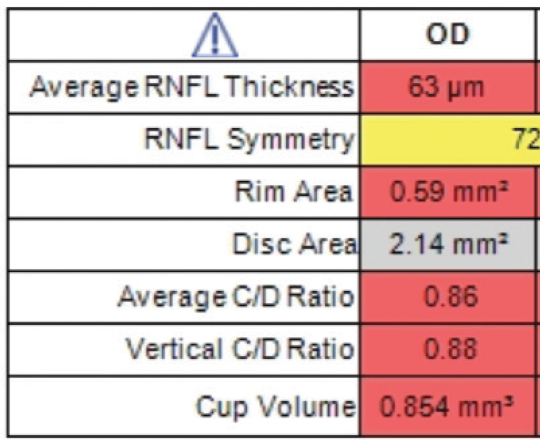
Figure 2. Nerve fiber layer analysis demonstrated mild to moderate defects.
Postoperative Results
- Same day postoperative (about 6 hours later) IOP of 9 mm Hg with no hyphema
- At 1 week, IOP was 12 mm Hg off glaucoma drops
- At 1 month, her pressure had stabilized at 11 mm Hg off glaucoma drops
- At 3 months, her pressure was 10 mm Hg off glaucoma drops
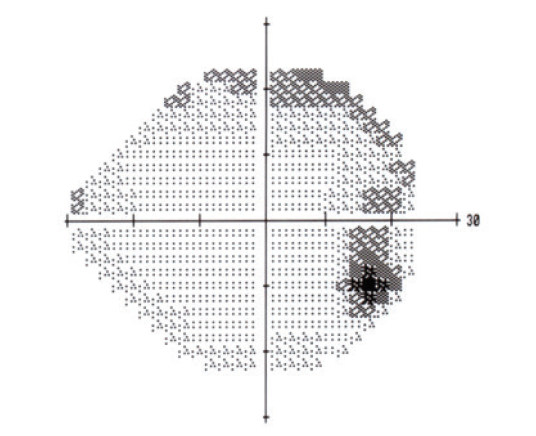
Figure 3. Humphrey visual field demonstrated mild superior arcuate defects.
Case Conclusion
OMNI Surgical System performed successfully in this patient who had a reduction in preoperative medicated IOP to postoperative unmedicated IOP.
Getting Started with OMNI: MY First 10 Patients
As with other MIGS technologies, learning to operate using a gonioprism in the non-dominant hand offers approximately 90% of the additional skillset a surgeon would need to perform OMNI. As you prepare for the first OMNI case, I recommend practicing intraoperative gonioscopy at the end of cataract cases until comfort is reached. Take time to reposition the patient’s head, reposition the microscope, get comfortable with the seating position, and all the maneuvers required to obtain and maintain a centered, clear, close-up view of the angle.
For the first 10 OMNI cases, recruit cooperative patients who are motivated to reduce their eye drop medication burden. In terms of anatomical set up for excellent visualization, look for open angles with visible pigmentation in the TM. The more heavily pigmented the TM, the easier it will be to perform OMNI initially. The advanced skillset of a modern cataract surgeon should allow adoption of OMNI into their tool kit.
Conclusion
My experience with OMNI has been both beneficial and rewarding. Early outcomes include delighted patients with significantly lower IOP and/or reduced medication burden. The ability to address two problems (cataract and glaucoma) in one surgical session is nearly universally accepted by patients; this means one trip to the OR, one anesthetic administration, and one postoperative course with one set of postoperative medications, visits, and expenses. As a result, most patients enjoy clear vision postoperatively as if we had performed lens replacement alone.
1. Gedde SJ. Results from the tube versus trabeculectomy study. Middle East Afr J Ophthalmol. 2009;16(3):107-111.
IMPORTANT SAFETY INFORMATION
INDICATIONS FOR USE: The OMNI® Surgical System is a manually operated device for the delivery of small amounts of viscoelastic fluid, for example Healon or HealonGV* from Abbott Medical Optics (AMO), Amvisc* from Bausch & Lomb, or PROVISC* from Alcon, during ophthalmic surgery. It is also indicated to cut trabecular meshwork tissue during trabeculotomy procedures.
DISCLAIMER: The OMNI® Surgical System is cleared (indicated) by FDA for the uses set forth above. While the OMNI® Surgical System is not specifically cleared for transluminal canal dilation, there is support for its use in transluminal canal dilation in the literature and medical textbooks, and ab interno trabeculotomy, for which it is FDA-cleared, is referred to as a MIGS procedure in the literature and medical textbooks and dictionaries. A current list of references/publications is available through Sight Sciences, Inc.
*Trademarks are property of their respective owners.
For important safety information including contraindications, warnings, precautions and adverse events, please visit omnisurgical.com.
© 2020 Sight Sciences, Inc. 7/20 OM-1093-US.v1



