
The goal of refractive cataract surgery is to reduce patients’ dependence on glasses by correcting presbyopia and/or astigmatism at the time of lens extraction. For years, monovision and multifocal IOLs were the primary means of correcting presbyopia. Although each of these strategies can provide satisfactory outcomes for patients, each also has its drawbacks.
Monovision provides near vision but sacrifices the visual quality benefits of binocular summation. Depth perception can also be compromised, and the lack of redundancy between eyes at a particular range can exacerbate the effects of ocular surface changes. Multifocal IOLs provide excellent near vision, but they can also have visual quality tradeoffs, including reduced contrast sensitivity and the presence of nighttime dysphotopsias. Additionally, early generations of multifocal IOLs provided a near point closer than many patients desire, leaving a visual quality gap in intermediate vision.
Fortunately, more recent designs of presbyopia-correcting IOLs can provide patients with a more natural range of vision, with near focal points at a more functional intermediate range and with fewer night vision symptoms. This trend began with the introduction of low-add multifocal IOLs on both the Tecnis (Johnson & Johnson Vision) and AcrySof IQ (Alcon) platforms and has continued with the introduction of an entirely new FDA-defined category of presbyopia-correcting IOLs: extended depth of focus (EDOF) IOLs.
Rather than creating two distinct focal points as multifocal IOLs do, EDOF IOLs provide a wider continuous range of high-quality vision, extending the focus from distance into intermediate and even into near range. EDOF IOLs provide excellent intermediate vision, which can be ideal for the frequent digital device use of today’s active professionals. In the United States, the only EDOF IOLs available are the Tecnis Symfony and Tecnis Symfony Toric (Johnson & Johnson Vision), which use diffractive optics to extend the depth of focus. The IC-8 IOL (AcuFocus), which uses pinhole optics to achieve extended depth of focus, is in use in Europe and currently undergoing FDA clinical trials.
The ideal solution for presbyopia would be to provide true accommodation similar to that of a young healthy eye, but that technology does not yet exist. Until it does, I find that EDOF IOLs come closest to meeting patients’ top priorities for (1) high quality of vision, particularly in the far distance, (2) minimal visual symptoms in low light, and (3) a continuous range of good vision through intermediate and into near. For this reason, an EDOF lens is the starting point for discussions with nearly all of my cataract patients who want to reduce their dependence on glasses after surgery.
QUALITY OF VISION
The primary goal of cataract surgery is to improve the patient’s quality of vision. The quality or sharpness of vision is determined largely by the presence or absence of optical aberrations, which can be influenced by IOL material and design. Even among monofocal IOLs, there is a range of optical quality, and a presbyopia-correcting IOL introduces further potential for optical compromises.
Tecnis Symfony EDOF IOLs use aspheric optics to fully correct the 0.27 µm of positive spherical aberration (SA) of the average cornea. They are made from a low-dispersion hydrophobic acrylic material with an Abbe number of 55 to minimize the amount of longitudinal chromatic aberration (CA) in the pseudophakic eye. In optical bench testing of the various materials used in IOLs available in the United States, Henk A. Weeber, PhD, and I found that there is a nearly a threefold difference in the amount of CA introduced by the materials used in currently available presbyopia-correcting IOL (Figure 1).1 The Tecnis acrylic adds the least CA (0.47 D), at a level comparable to that of the natural human lens.
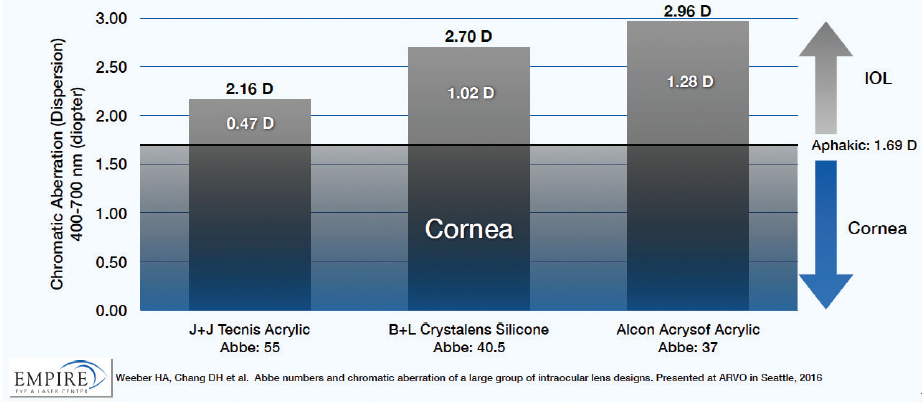
Figure 1. Comparison of chromatic aberration among available presbyopia-correcting IOL materials.
In addition to these materials factors, the Symfony uses a diffractive achromatic technology similar to that used in high-end diffractive-optics camera lenses, which actually corrects the CA of the cornea to reduce the CA of the entire eye. This further enhances contrast sensitivity and improves retinal image quality.
By leveraging the synergistic combination of SA correction, CA minimization, and active CA correction, these EDOF IOLs can increase depth of focus and still provide high visual quality. Image quality is best measured by modulation transfer function (MTF), a measure of the amount of contrast transferred by an optical system. The higher the MTF value, the better the image contrast. In laboratory bench studies in photopic conditions, the MTF value of the Tecnis Symfony EDOF IOL is at least 35% higher than any multifocal IOL platform. In mesopic conditions, the difference increases to 74% (Figure 2).2
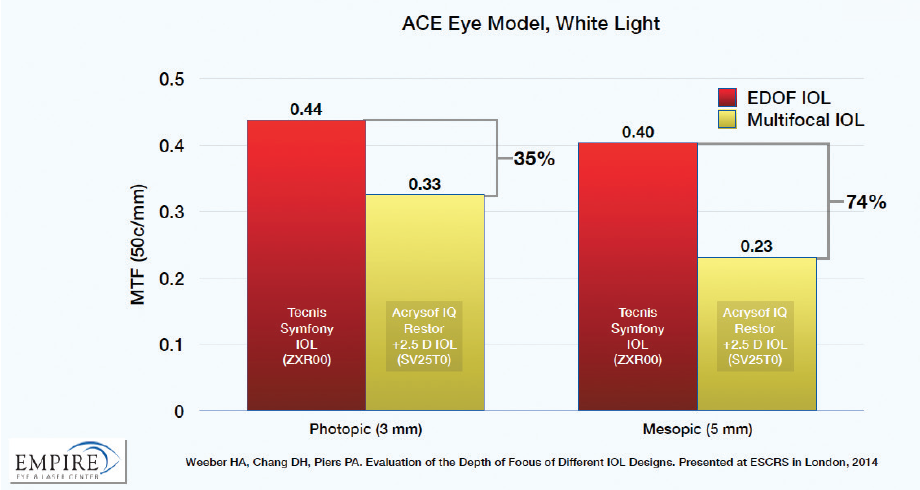
Figure 2. Comparison of image quality (MTF) between an EDOF (red) and a multifocal (yellow) IOL.
Clinically, Petrotti et al found no significant difference in contrast acuity or visual quality between the Tecnis Symfony and Tecnis monofocal IOLs, and patients with the Symfony had better uncorrected distance visual acuity and less dependence on glasses.3
It is notable that the Symfony EDOF lenses are made from a material that has not been associated with significant glistenings, which can cause light scatter and further reductions in image contrast. Glistenings continue to remain a problem in some modern high-index hydrophobic acrylics.4,5
Choosing an IOL that provides high image quality for patients not only results in the best possible visual quality; it also helps to mitigate the effects of factors that can negatively affect vision or visual quality, including residual refractive error, ocular surface problems, posterior capsular opacification, and minor retinal pathology.
NIGHT VISION SYMPTOMS
With all pseudoaccommodating IOLs, increasing depth of field is associated with some induction of dysphotopsias in low light conditions, particularly at night. These nighttime vision symptoms vary in configuration and intensity with IOL design, and they can also vary in severity and symptomatology from patient to patient.
Halos have commonly been associated with multifocal IOLs. When patients look at a distant object, out-of-focus energy directed at the near focal point creates the halo, and larger halos are associated with higher add powers. Patients with the diffractive EDOF IOLs report a different appearance from traditional halos; they tend to note radial starbursts with multiple fine concentric halos in a spiderweb pattern (Figure 3). In my experience, patients with the Symfony have fewer and milder nighttime vision complaints compared to patients with multifocal IOLs.

Figure 3. Simulated profile of low-light dysphotopsias.
Appropriate preoperative counseling plays a key role in how patients perceive their symptoms after surgery. Patients should be counseled that there will likely be some night vision symptoms but that they are usually quite tolerable. When you describe low-light dysphotopsias, it is best to use as much precision as possible. A simple comment about glare and halos does not convey the same level of preparation as a more detailed description like the one in the preceding paragraph. The more specific the description, the better patients seem to adapt to night vision symptoms postoperatively. For the rare patient who is not willing to consider tolerating any low-light dysphotopsias, it would be best to steer him or her away from presbyopia-correcting IOL solutions.
FUNCTIONAL RANGE OF VISION
With expected uncorrected binocular visual acuity of 20/20 for distance and intermediate and around 20/40 for near, patients with EDOF IOLs can function well without glasses for most daily activities, including driving, using a computer, and looking at their tablet or smartphone. Near vision may not be quite as strong as with a fully diffractive multifocal IOL. I find, however, that when I ask patients preoperatively, most say they would not mind an occasional need for low-powered reading glasses for small print or dim lighting, particularly if they have high-quality vision for far distance.
The range of vision provided by the EDOF platform gives us the opportunity to personalize vision based on each patient’s needs. In most cases, I implant the dominant eye first with an EDOF lens and then specifically assess the patient’s satisfaction with near and night vision at the 1-day and 1-week postoperative visits to guide IOL choice for the second eye. If the patient is happy with the outcome of the first eye, I implant another EDOF IOL in his or her second. If the patient wants better near vision—typically low myopes who are accustomed to removing their glasses to read—I can implant a low-add multifocal in the contralateral eye.
For the occasional patient who has a primary concern about low-light dysphotopsias, I implant the nondominant eye first with an EDOF lens and then assess satisfaction with near and night vision postoperatively. If the patient complains about night vision symptoms related to the EDOF IOL, I would then consider a monofocal or monofocal toric IOL in his or her contralateral dominant eye.
CONCLUSION
The availability of EDOF IOLs has simplified my discussions with patients because I know that I can generally achieve a patient’s goals with bilateral EDOF lenses or with a personalized combination of two different lens types. Additionally, with toric EDOF lenses also available, I no longer have to choose between correcting presbyopia and astigmatism.
I believe that EDOF IOLs are the best options available to patients today for correcting vision at all distances while minimizing night vision symptoms and tradeoffs in quality of vision.
1. Chang DH, Weeber HA, Lowery MD, Piers PA. Chromatic aberration of intraocular lenses measured in vitro and in vivo. Invest Ophthalmol Vis Sci. 2016;57(12):E-abstract 3110.
2. Weeber HA, Chang DH, Piers PA. Evaluation of the depth of focus of different IOL designs. Paper presented at: ESCRS Annual Meeting; September 13-17, 2014; London.
3. Pedrotti E, Bruni E, Bonacci E, et al. Comparative analysis of the clinical outcomes with a monofocal and an extended range of vision intraocular lens. J Refract Surg. 2016;32:436-442.
4. Nagata M, Matsushima H, Mukai K, et al. Clinical evaluation of the transparency of hydrophobic acrylic intraocular lens optics. J Cataract Refract Surg. 2010;36(12):2056-2060.
5. Van der Mooren M, Franssen L, Piers P. Effects of glistenings in intraocular lenses. Biomed Opt Express. 2013;4(8):1294-1304.




