
Patients with glaucoma deserve to have their presbyopia managed, but should they be offered presbyopia correction at the time of cataract surgery? As an experiment, I asked some ophthalmologists if they would implant a multifocal IOL in a patient with glaucoma. At the risk of sounding like a toothpaste commercial, nine out of 10 respondents cited concerns about contrast sensitivity in their responses. It made me wonder where this dogma originated.
CONTRAST SENSITIVITY IN REVIEW
Contrast refers to the difference in luminance or color that makes an object distinguishable. The ability to detect this difference is called contrast sensitivity, which is often measured on a logarithmic scale. The term spatial frequency refers to the ability to detect a difference between objects, and it is reported in cycles per degree (cpd). For context, most meaningful vision, such as seeing a street sign, takes place at spatial frequencies of 3 to 6 cpd (Figure 1).
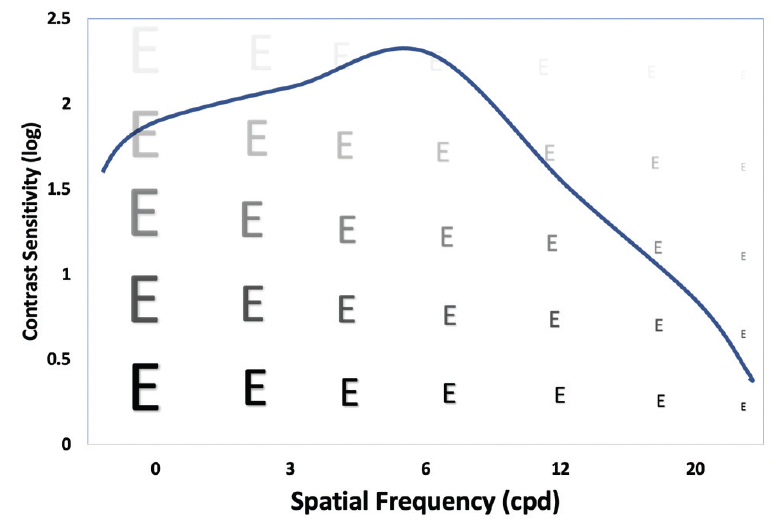
Figure 1. Contrast sensitivity function in a normal eye.
Multiple tools are available to measure contrast sensitivity; the most common is the CSV-1000 test. Although this test is validated, it is challenging to conceptualize the experiential effect of reduced contrast sensitivity. The Pelli-Robson chart (see example in Figure 2) is not a perfect method, but it permits direct visualization of changes in contrast sensitivity. This test features triplets of letters, each of which represents a different level of contrast. By reading two of the three letters in the triplet, the test subject gets credit for that contrast sensitivity score (the number adjacent to the triplet) at a spatial frequency of 1 cpd. For context, normal eyes have a contrast sensitivity score of greater than 2 at spatial frequencies of 6 cpd.
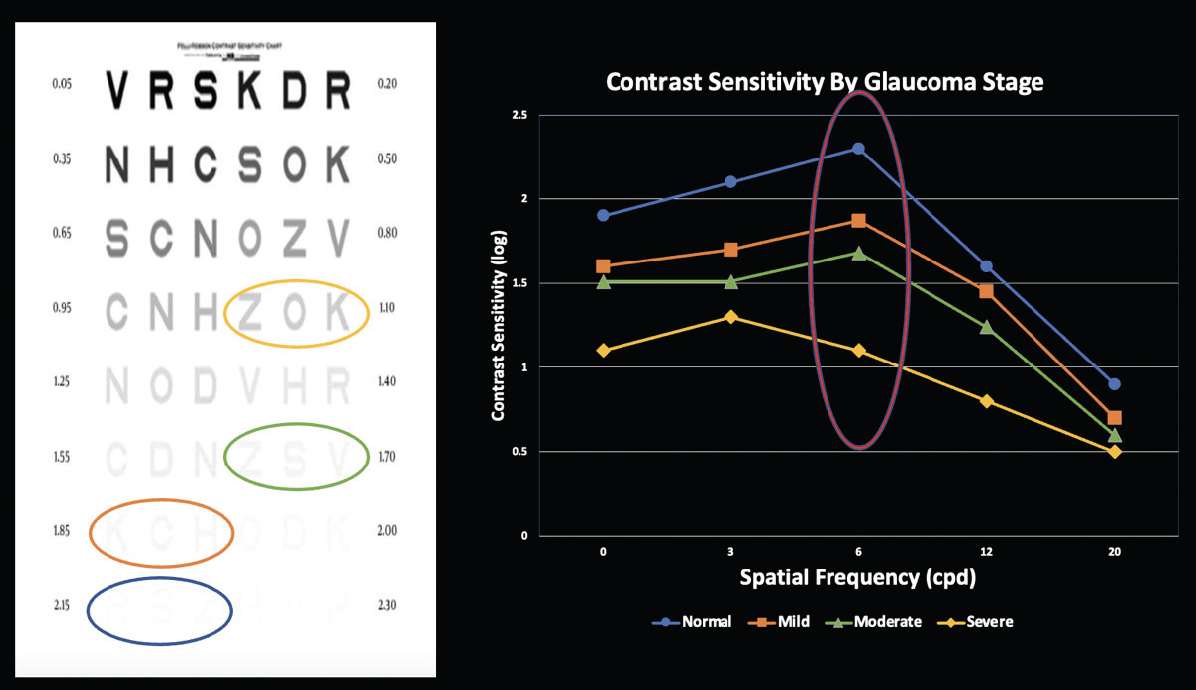
Figure 2. The Pelli Robson chart (left) and contrast sensitivity by glaucoma stage (right).
The literature has shown that patients with glaucoma have reduced contrast sensitivity.1 A linear relationship has been reported between mean deviation on visual field testing and contrast sensitivity: The higher the mean deviation, the worse the contrast sensitivity.2 Figure 2 shows the level of contrast per disease stage circled on the Pelli-Robson chart to illustrate decreasing contrast more clearly in a patient with glaucoma.
PRESBYOPIA CORRECTION AND CONTRAST SENSITIVITY
As eye care providers, our role is to minimize harm to patients, so we should avoid any intervention that may further reduce contrast sensitivity in glaucomatous eyes. Do presbyopia-correcting IOLs meaningfully reduce contrast sensitivity? The answer to this question is nuanced.
For the first 20 years that multifocal IOL technology was available, both refractive and diffractive lens platforms appreciably reduced contrast sensitivity.3 Not only patients with glaucoma experienced waxy vision and had difficulty seeing in dimly lit environments with early multifocal lens technology. All patients who received these lenses experienced reduced contrast sensitivity. Has this changed with advances in lens technology?
To be considered a clinically meaningful reduction, the contrast sensitivity score must decrease by log 0.3 at a given spatial frequency. Figure 3 demonstrates the contrast sensitivity function for the AcrySof IQ PanOptix trifocal IOL (Alcon) compared with that of a monofocal IOL. In this analysis, no clinically meaningful difference in contrast sensitivity was observed between the PanOptix lens and the monofocal equivalent at any spatial frequency, regardless of the presence of glare and in both mesopic and photopic conditions.
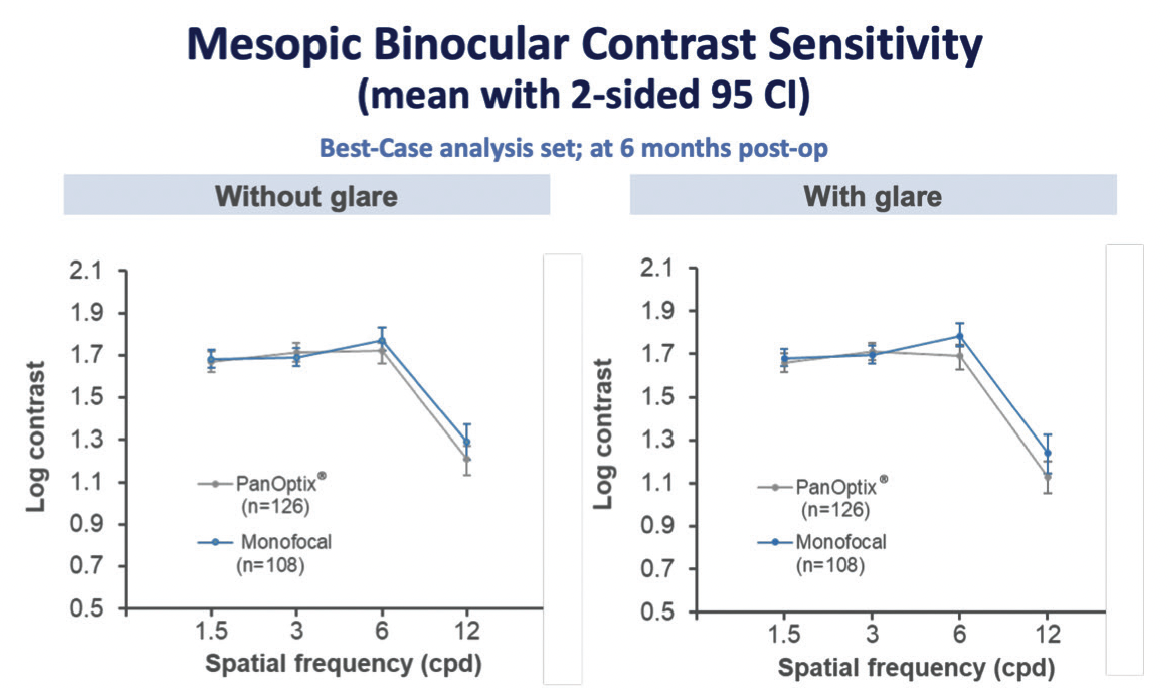
Figure 3. No clinically meaningful difference in mesopic contrast sensitivity was observed between an AcrySof IQ PanOptix IOL and a monofocal IOL across spatial frequencies, regardless of the presence of glare.4
OUTDATED DOGMA?
I submit that the dogma of “Do not implant a multifocal IOL in a patient with glaucoma because of decreased contrast sensitivity” is outdated and should be retired. To be clear, I am not advocating for the placement of multifocal IOLs in patients with glaucoma. I do not recommend implanting a presbyopia-correcting lens in a patient with moderate or severe disease. I do, however, believe that IOL selection should be centered on the following question: Is it worth it for a given patient to pay for a premium IOL if that person may not experience its full benefits? In other words, what is the value to the patient?
When evaluating whether a glaucoma patient is a candidate for a multifocal IOL, I consider the trajectory of vision loss (Figure 4). If a patient is likely to lose central vision from glaucoma, that person’s visual satisfaction will be low whether a multifocal or a monofocal IOL is implanted. This brings us back to value. Is it worth paying for a form of vision correction that may not achieve its goals owing to a comorbidity?
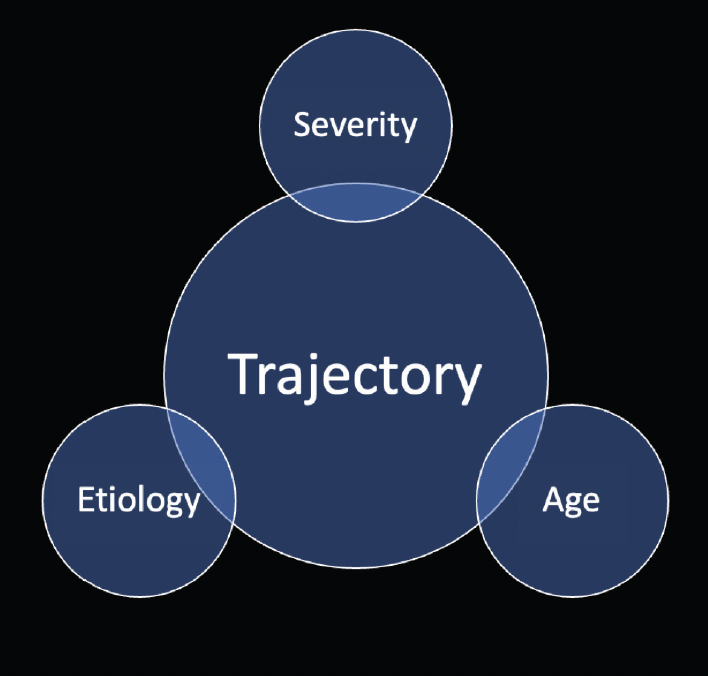
Figure 4. Trajectory: a combination of etiology, age, and severity of glaucoma.
Let us consider a patient with mild, intermittent angle-closure glaucoma who is now pseudophakic and has not demonstrated glaucomatous progression for 20 years. Based on a flat trajectory, this patient could benefit from premium IOLs and would likely enjoy the value of spectacle independence for a lifetime.
Contrast that scenario with that of a 60-year-old patient who has bilateral central islands and 20/400 visual acuity from severe pseudoexfoliation glaucoma. Would this patient experience any value from a premium IOL? Most likely not.
As physicians, it is our responsibility always to do what is best for the patient. This conversation becomes simple if we ask ourselves, “What would I want if it were my eye?”
CONCLUSION
The year 2020 delivered many valuable lessons, including how to let go. Maybe in 2021 we can finally dismiss the old adage of avoiding multifocal IOLs in certain patients because of contrast sensitivity and redirect the conversation to what matters most: patient value.
1. Ichhpujani P, Thakur S, Spaeth GL. Contrast sensitivity and glaucoma. J Glaucoma. 2020;29(1):71-75.
2. Wilensky JT, Hawkins A. Comparison of contrast sensitivity, visual acuity, and Humphrey visual field testing in patients with glaucoma. Trans Am Ophthalmol Soc. 2001;99:213-218.
3. Ravalico G, Baccara F, Rinaldi G. Contrast sensitivity in multifocal intraocular lenses. J Cataract Refract Surg. 1993;19(1):22-25.
4. FDA premarket approval for PanOptix. FDA. Accessed July 22, 2021. www.fda.gov/cder/approval/index.html




