Incorporating dry eye diagnosis and management into a busy practice is not as easy as it sounds. First, you have to buy into the concept that dry eye disease (DED) is important and realize that it matters to each and every one of your patients. (It also matters to you in the long run.) Second, you must inspire staff buy-in and ownership (not monetarily) of the same concept for your effort to be successful.
The key components are to look, listen, and treat for DED.
LOOK
We look for problems by presenting all of our patients with a symptoms questionnaire, namely the Ocular Surface Disease Index (OSDI) or the Standardized Patient Evaluation of Eye Dryness (SPEED), to identify ocular surface disorders.
Many patients are not even aware of how their eyes should feel normally (hint: they should feel like they aren’t even there), or they overlook telling us that they are bothered by symptoms until they are walking out of the exam lane. Typically, you have just finished your chart note when you hear the dreaded words: “Oh, by the way doctor, my eyes have been burning and stinging lately.” Instead, these symptoms can be identified while patients are in the waiting room and flagged by your staff so that you will be aware of them by the time you see the patient (see the heading LISTEN for more information).
I have made it a point to train staff members to look by staining the cornea with a strip of fluorescein and lissamine green. This allows them to get a clean view of the corneal surface without alterations due to the patient sitting and not blinking, with the eyes anesthetized and dilating before I get into the room. At least we can determine that there is staining before applanation for IOP checking. This helps to direct treatment approaches. A thorough examination of the bulbar and tarsal conjunctiva is necessary as well, to identify allergies and other possible comorbidities.
The last component of looking is to examine the meibomian glands directly to assess the quality and quantity of the meibum. This requires expression of the glands on both upper and lower lids, as there is often considerable variation. This can be done with a Q-tip by a trained technician, an optometrist, or you can do it yourself. This is crucial because meibomian gland dysfunction (MGD) comprises 86% of evaporative DED.1 Additionally, according to a study by Gupta et al,2 42% of children between the ages of 8 and 16 years have early evidence of meibomian gland changes on LipiView (Johnson & Johnson Vision) exams.
With easy metrics, a questionnaire like OSDI or SPEED, corneal staining, and lid expression, most patients with DED can be identified. Having the TearLab Osmolarity System (TearLab) available is also helpful because we know from the Dry Eye WorkShop II that hyperosmolarity is a key component of dry eye.3
LISTEN
I encourage staff members to listen for complaints of DED symptoms. With staff buy-in, when staff members hear these complaints, they can proactively undertake testing to help you move more quickly down the lanes. If your practice has the point-of-care tests for tear film osmolarity or for the inflammatory marker matrix metalloproteinase-9 (InflammaDry, Rapid Pathogen Screening), these tests should be done by a technician before you walk into the room. If the technician has time, he or she can also perform imaging of the meibomian glands with the LipiView II Ocular Surface Interferometer. If not, all of these tests can be performed in a specific dry eye visit scheduled for later.
In the long run, looking and listening for DED will save you a boatload of time. When a patient wants surgery and we are ready to schedule it, only to find out that he or she has signs or symptoms of DED, scheduling comes to a halt and we have to backpedal and treat the ocular surface first. Otherwise, our biometric tests are inconsistent and frankly inaccurate,4 and it is difficult to plan for toric or multifocal IOL correction.
TREAT
Deciding how to treat DED is the most difficult part of the equation, as incorporating DED treatment into your busy day is more challenging than looking for symptoms and listening for complaints. The treatments for DED are more time-consuming than the exams.
Therefore, the best way to incorporate them is to set aside treatment days specifically for DED. Whether it is the LipiFlow Thermal Pulsation System (Johnson & Johnson Vision), BlephEx eyelid cleaning (BlephEx), iLux heat and compression treatment (Tear Film Innovations), Mibo ThermoFlo heat treatment (Mibo Medical Group), or another treatment, all these procedures tack on extra time to your patient flow.
These times add up, and, unless your staff is streamlined and trained to move selected patients from the examination lane to a treatment room, you will undoubtedly begin to run behind. Also, all questions must be answered and patient education completed before treatment. DED patients tend to have a lot of questions, and they love commiserate about their symptoms. After a few visits and treatments, as long as there is some improvement, the time they require diminishes dramatically.
Prescribing drops such as lifitegrast ophthalmic solution 5% (Xiidra, Shire), cyclosporine ophthalmic emulsion 0.05% (Restasis, Allergan), or loteprednol etabonate ophthalmic suspension 0.5% (Lotemax, Bausch + Lomb) helps your throughput, as the patient leaves with a medication plan and can return for punctal plugs and/or further lid treatment.
Do not try to accomplish all treatments in one visit, as it will take much too long and set your schedule behind. The concept is to do one thing at a time—with a plan of action for each visit—so you can deliver a treatment option and adjust it each time the patient comes to the office.
TIMING IS EVERYTHING
To incorporate all of this into a busy practice, timing is everything. It seems to work best for us when time is set aside separately for these in-office treatments, so that regular patient flow is not backed up. As mentioned, DED patients like to talk about their problems, so a patient’s first visit should be longer to allow time for the patient to explain problems and for you to educate him or her on solutions. After that, you should be able to move them through your workflow like other patients.
If, however, a DED patient does not respond to therapy, you will either have to refer him or her out to a subspecialty cornea clinic or be prepared to take the time to work through a complex treatment paradigm with the patient.5 This is when throughput during regular office hours becomes difficult.
Early treatment of MGD can restore health to the many meibomian glands that are withering away from poor blinking during the use of digital devices or from contact lenses and bacterial biofilm buildup on the lid margin that block the meibomian orifices. These glands must be reopened, as they have shut down over time—most likely as an autoregulation effect designed to block meibum production when there is no outlet or exit. It is up to us as eye care professionals to identify MGD and salvage what we can. It has been quite shocking and sad to see the extent of meibomian gland atrophy in the spectrum of young patients (Figure).
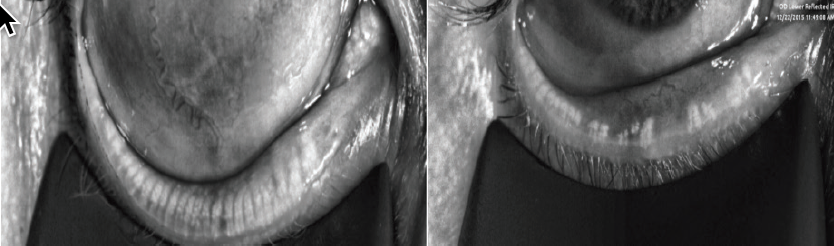
Figure. Normal meibomian glands (left). Meibomian gland dropout, atrophy, and truncation in a 26-year-old man seeking LASIK for contact lens intolerance (right).
Conclusion
Incorporating basic DED diagnostics and treatment can greatly enhance your practice and improve your patients’ lives. If you look and listen, you will not miss early treatment opportunities, and you will have the chance to treat and restore your patients’ healthy, natural ocular surface. Further, your surgical outcomes will be dramatically improved because the first refracting surface of the eye, the tear film, will be smoother, more regular, and healthier. Finally, your patients will be extremely grateful and loyal to you and your practice.
1. Lemp MA, Crews LA, Bron AJ, Foulks GN, Sullivan BD. Distribution of aqueous-deficient and evaporative dry eye in a clinic-based patient cohort: a retrospective study. Cornea. 2012;31(5):472-478.
2. Gupta PK, Stevens MN, Kashyap N, Priestley Y. Prevalence of meibomian gland atrophy in a pediatric population. Cornea. 2018;37(4):426-430.
3. Craig JP, Nichols KK, Akpek EK, et al. TFOS DEWS II Definition and Classification Report. Ocul Surf. 2017;15(3):276-283.
4. Epitropoulos AT, Matossian C, Berdy GJ, Malhotra RP, Potvin R. Effect of tear osmolarity on repeatability of keratometry for cataract surgery planning. J Cataract Refract Surg. 2015;41(8):1672-1677.
5. Milner MS, Beckman KA, Luchs JI, et al. Dysfunctional tear syndrome: Dry eye disease and associated tear film disorders–new strategies for diagnosis and treatment. Curr Opin Ophthalmol. 2017;27(Suppl 1):3-47.





