The multiple design considerations of the AcrySof IQ ReSTOR IOL with ACTIVEFOCUS optical design (Alcon) are additive in delivering overall optical performance. The lens has been specifically engineered to provide quality vision across near, intermediate, and distance while also allowing surgeons flexibility to tailor the surgical plan to the visual needs of the patient. Maintaining the final vision is then facilitated by properties that confer greater mechanical stability of the IOL placement to decrease the potential for axial rotation—and a certain margin of error should slight decentration occur.
The result of these considerations for design is that they help to provide the best conditions for ensuring patients are happy with the final visual result. Patients opting for premium lenses may be more demanding of final vision compared with monofocal patients, and so reducing the potential for visual compromises may go a long way to meeting those expectations.
Quality Distance Vision and a Full Range of Vision
The ACTIVEFOCUS design with 7-step diffractive steps is intended to provide greater energy distribution for distance vision, and more energy distribution overall, thus ensuring uncompromised distance vision with a full range of vision (Figure 1).1-3 Yet, achieving quality distance acuity does not mean having to sacrifice near vision, as the AcrySof IQ IOL with ACTIVEFOCUS provides for closer near acuity compared with other presbyopia-correcting IOL technologies (Figure 2).4,5
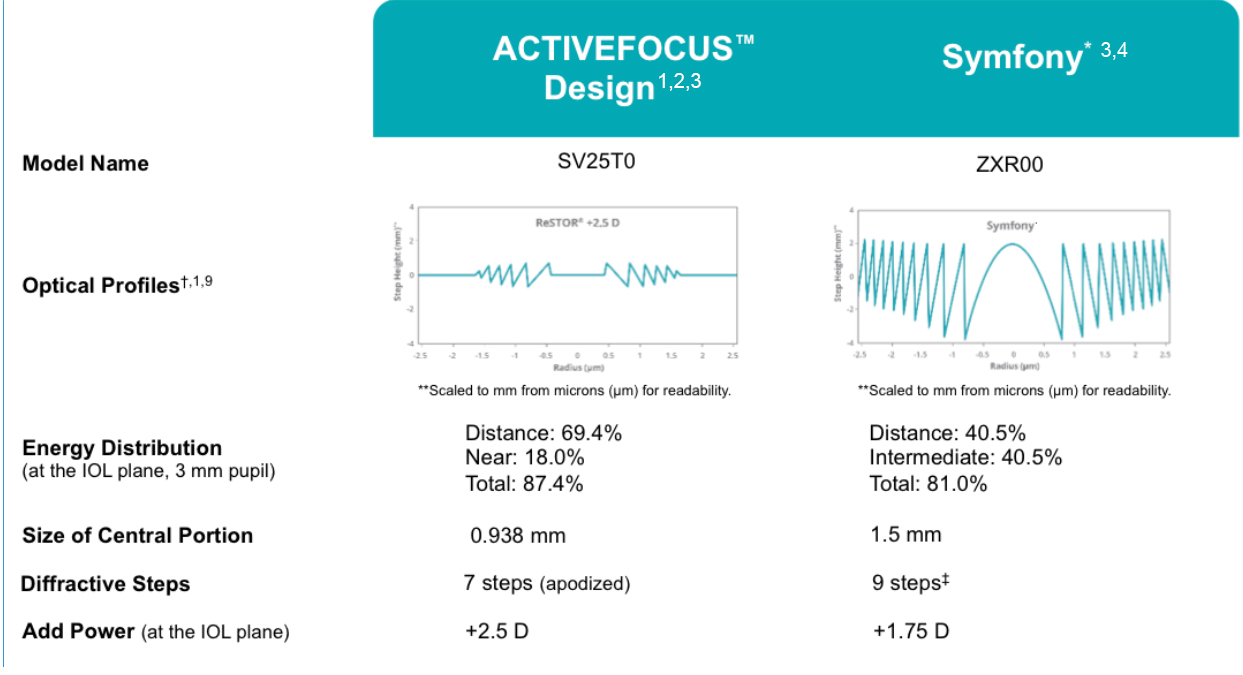
Figure 1. Overview of design profiles and implications of two presbyopia-correcting IOL platform technologies. Note: Optical profile of the ReSTOR +2.5 D model SV25T0 is based on the design profile. Surface profile of the TECNIS Symfony 28.0 D IOL (Johnson & Johnson Vision) measured using Bruker Contour white light interferometer on the posterior surface and the diffraction efficiency calculated.
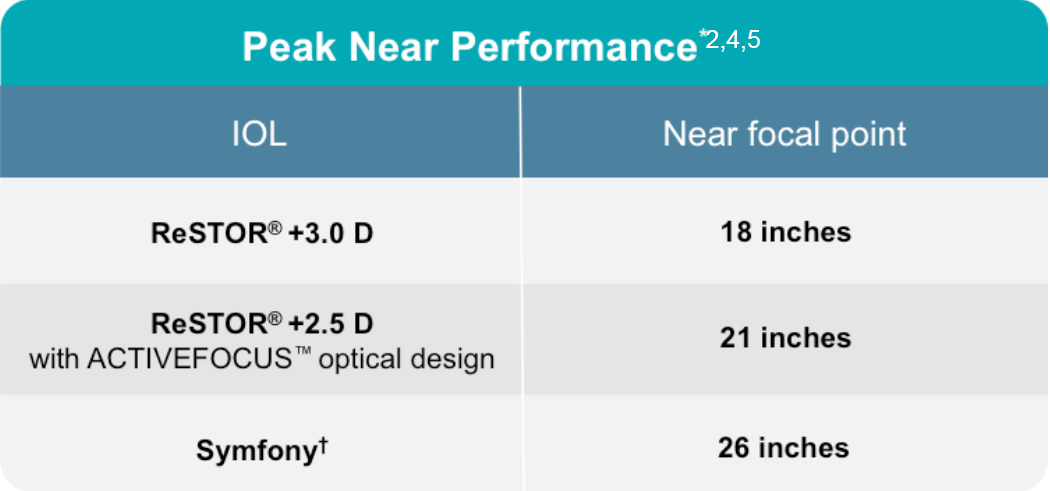
Figure 2. Peak near performance with on-market presbyopia-correcting IOLs.
*Derived from the defocus curve in each respective products direction for use.
Designed for Low Incidence of Visual Disturbances
The unique distance-dominant ACTIVEFOCUS optical design was added to the AcrySof IQ platform using the same spherical aberration correction factor in monofocal models and, therefore, provides comparable contrast sensitivity (Figure 3).6
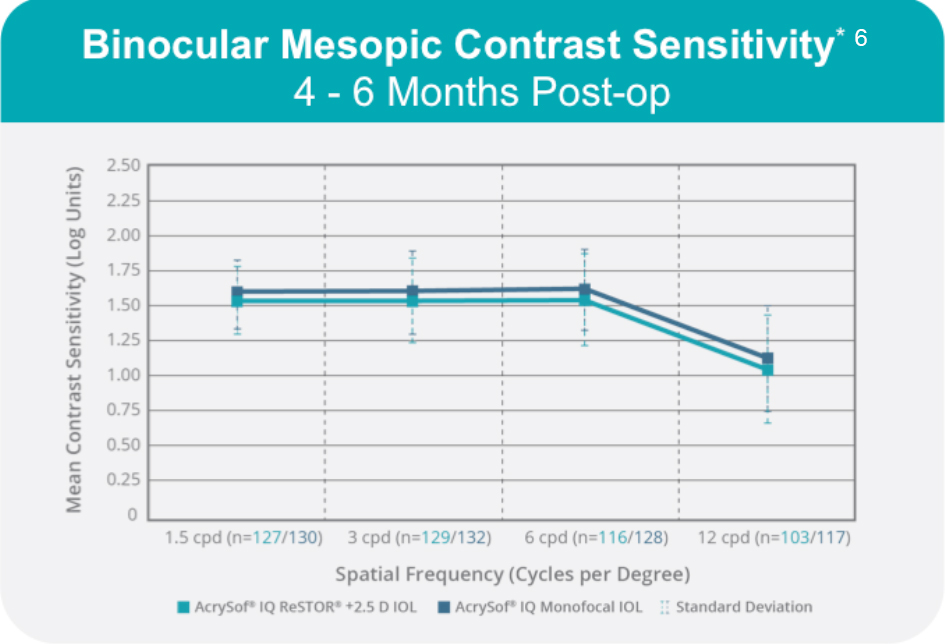
Figure 3. Binocular mesopic contrast sensitivity with the AcrySof IQ ReSTOR +2.5 D IOL with ACTIVEFOCUS versus AcrySof IQ ReSTOR monofocal IOL.
*With glare.
These features mean that patients are less likely to experience reduced compromise in the quality of the final vision. Importantly, though, the AcrySof IQ is specifically designed to minimize the potential for problematic visual disturbances, such as halo and glare (Figure 4).7
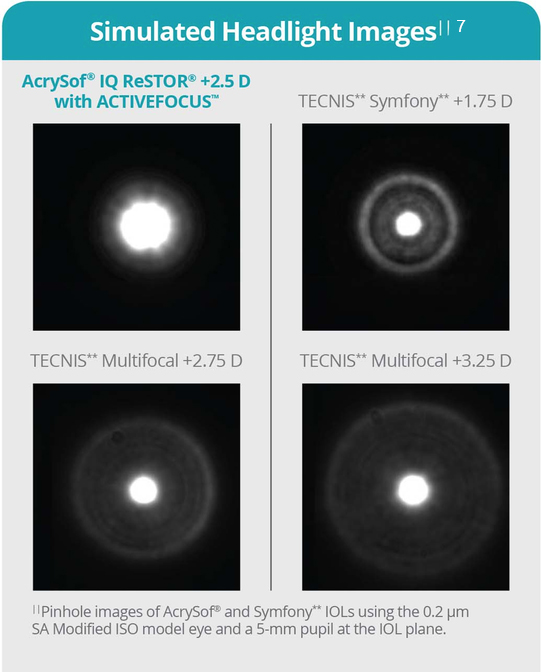
Figure 4.
Conclusion
IOL options that are designed to reduce visual disturbances address an issue that has historically been a point of consternation with presbyopia-correcting IOLs. Visual disturbances due to light diffracting through multiple optical zones on multifocal lens designs are rightly considered a safety issue, as they may affect patients’ ability to function with their postoperative vision—for example, interfering with nighttime vision.
Above and beyond the potential to help avoid positive dysphotopsias after cataract surgery, modern IOL designs are also engineered to provide the overall quality of vision that patients expect. The combination of features on the AcrySof IQ ReSTOR IOL with ACTIVEFOCUS helps deliver overall optical performance to help patients achieve postoperative vision goals and wind up happy with the result.
AcrySof, ACTIVEFOCUS, and ReSTOR are trademarks of Novartis. © Novartis 2018. All other brand/product names are the trademarks of their respective owners.
© 2018 Novartis 9/18 US-RES-18-E-1916
1. Alcon Data on File (April 11, 2016).
2. AcrySof IQ ReSTOR +2.5 D IOL Directions for Use.
3. Alcon Data on File (Oct 17, 2016).
4. TECNIS Symfony Extended Range of Vision IOL Directions for Use.
5. AcrySof IQ ReSTOR +3.0 D IOL Directions for Use.
6. Alcon Data on File (Aug 7, 2013).
7. Alcon Data on File (May 17, 2016).
AcrySof® IQ ReSTOR® Family of Multifocal IOLs Important Product Information
CAUTION: Federal (USA) law restricts this device to the sale by or on the order of a physician.
INDICATIONS: The AcrySof® IQ ReSTOR® Posterior Chamber Intraocular Multifocal IOLs include AcrySof® IQ ReSTOR® and AcrySof® IQ ReSTOR® Toric and are intended for primary implantation for the visual correction of aphakia secondary to removal of a cataractous lens in adult patients with and without presbyopia, who desire near, intermediate and distance vision with increased spectacle independence. In addition, the AcrySof® IQ ReSTOR® Toric IOL is intended to correct pre-existing astigmatism. The lenses are intended to be placed in the capsular bag.
WARNINGS/PRECAUTIONS: Careful preoperative evaluation and sound clinical judgment should be used by the surgeon to decide the risk/benefit ratio before implanting a lens in a patient with any of the conditions described in the Directions for Use labeling for each IOL. Physicians should target emmetropia, and ensure that IOL centration is achieved. Care should be taken to remove viscoelastic from the eye at the close of surgery.
The ReSTOR Toric IOL should not be implanted if the posterior capsule is ruptured, if the zonules are damaged, or if a primary posterior capsulotomy is planned. Rotation can reduce astigmatic correction; if necessary lens repositioning should occur as early as possible prior to lens encapsulation.
Some patients may experience visual disturbances and/or discomfort due to multifocality, especially under dim light conditions. A reduction in contrast sensitivity may occur in low light conditions. Visual symptoms may be significant enough that the patient will request explant of the multifocal IOL. Spectacle independence rates vary; some patients may need glasses when reading small print or looking at small objects.
Posterior capsule opacification (PCO), when present, may develop earlier into clinically significant PCO with multifocal IOLs. Prior to surgery, physicians should provide prospective patients with a copy of the Patient Information Brochure available from Alcon informing them of possible risks and benefits associated with the AcrySof® IQ ReSTOR® IOLs.
Do not resterilize; do not store over 45° C; use only sterile irrigating solutions such as BSS® or BSS PLUS® Sterile Intraocular Irrigating Solutions.
ATTENTION: Reference the Directions for Use labeling for each IOL for a complete listing of indications, warnings and precautions.





