Prominent topics at this year’s annual meeting of the American Glaucoma Society (AGS) included microinvasive glaucoma surgery (MIGS), new developments in visual function testing, and the practical application of clinical trial results. In this article, I review several studies I believe have the potential to change glaucoma management today or in the near future.
MIGS DEVICES
The Hydrus Microstent (Ivantis, Figure 1) is a metallic intracanalicular scaffold that provides 90º of outflow effect. Thomas W. Samuelson, MD, presented 2-year data from the device’s FDA trial, in which 556 patients were randomly assigned to cataract surgery alone or with implantation of the Hydrus.1 Preoperative mean IOP was approximately 25 mm Hg in both groups. At 24 months, mean reduction in diurnal IOP was -7.5 mm Hg in the Hydrus group and -5.2 mm Hg in the control group. Seventy-seven percent of Hydrus patients achieved the primary study endpoint of a 20% or greater reduction in IOP compared with 58% of controls. At 2 years, 78% of Hydrus patients versus 48% of controls remained medication-free. Complications associated with the stent included transient hyphema in 1.1% of patients and obstruction of the device by peripheral anterior synechiae in 3.8% of patients. No cases of hypotony occurred. In the control group, 2.7% of patients experienced a significant IOP spike and 2.1% required glaucoma surgery.

(Courtesy of Thomas Samuelson, MD)
Figure 1. Hydrus Microstent in situ.
Douglas J. Rhee, MD, presented the 12-month findings of a trial comparing the Hydrus Microstent with two current-generation iStent Trabecular Micro-Bypass Stents (model GTS100, Glaukos) as a standalone procedure.2 Investigators randomly assigned 152 patients without a history of incisional glaucoma surgery to receive the Hydrus or two iStents; approximately two-thirds of the patients were phakic. Mean unmedicated IOP was about 27 mm Hg on an average of 2.5 medications.
At 12 months, mean IOP was approximately 18 mm Hg in both groups. Patients in the Hydrus group required significantly fewer medications than those in the iStent group (mean, 1 vs 1.5). The Hydrus patients were also more likely than the iStent patients to be free of medication use (47% vs 24%). Few complications were observed. Device obstruction or adhesions occurred in 5% of the Hydrus group and 13% of the iStent group. One patient in each group required trabeculectomy.
ARTIFICIAL INTELLIGENCE
Artificial intelligence (AI) has received a great deal of attention in ophthalmology since the December publication of an article describing the use of a deep learning system to detect diabetic retinopathy in retinal images.3
Machine learning could also be used to recognize abnormal optic discs or to detect glaucomatous progression. At the AGS meeting, Osamah Saeedi, MD, reported on a project using 90,713 visual fields of 13,156 eyes from the Glaucoma Research Network collection.4 Investigators used available progression algorithms, including the Visual Field Index (VFI), to classify the fields as progressing or stable. The fields were used to train six different machine learning systems. Glaucoma specialists graded a subset of eyes for comparison.
The VFI had accuracy (defined as the ability to differentiate progression from stability) of 95%. The machine learning classifiers had accuracy ranging from 83% to 88%, sensitivity of 76% to 85%, and specificity of 89% to 94%. The investigators concluded that, in the future, machine learning could help clinicians determine which patients are experiencing disease progression.
LASER TRABECULOPLASTY
Should patients instill topical steroids or NSAIDs after undergoing selective laser trabeculoplasty (SLT)? A 2014 study of 125 eyes with baseline IOPs around 19 mm Hg found no difference in outcomes,5 but many clinicians continue to prescribe NSAID drops routinely after SLT. To study the matter further, investigators conducted a randomized multicenter trial that involved 96 eyes of 85 patients with IOP higher than 18 mm Hg. Sylvia Groth, MD, of Stanford University, presented the results at the AGS meeting.6
Patients received either prednisolone acetate 1%, ketorolac 0.5%, or artificial tears dosed four times a day for 4 days after SLT. Most patients had primary open-angle glaucoma (POAG), and baseline IOP was approximately 23 mm Hg in all three groups. At 6 weeks, there was no significant difference in IOP among the groups, but patients in both the NSAID and steroid groups showed a greater mean decrease in IOP at week 12 compared with the placebo group (-6.2, -5.2, and -3 mm Hg, respectively). The investigators concluded that the effect of SLT may depend on the activation of inflammatory signaling pathways and that modulating the impact through the short-term use of NSAID or steroid drops may improve results.
SYSTEMIC FACTORS
Blood flow dysregulation has been described extensively in patients with glaucoma. One way to directly assess the systemic vasculature is through video measurement of blood flow in the nailfold. Jonathan Chou, MD, presented the results of a study evaluating this technique in 67 POAG patients and 63 control patients (Figure 2).7
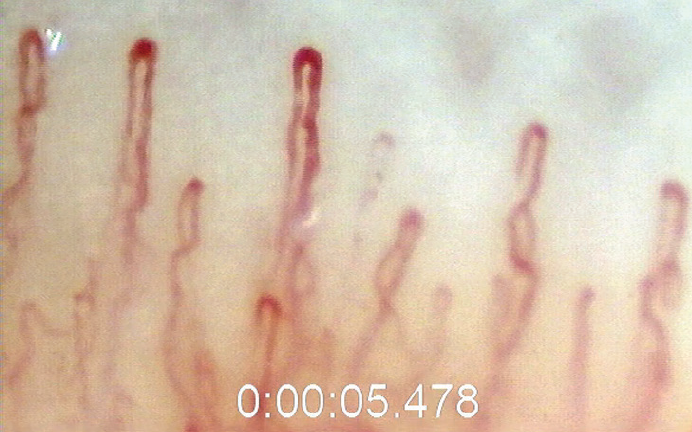
(Courtesy of Jonathan Chou, MD)
Figure 2. Nailfold capillaroscopy.
Mean blood velocity was 328.3 µm/s in POAG patients and 573.9 µm/s in controls. In a multivariate analysis that controlled for multiple factors (eg, age, blood pressure, smoking, diabetes), every 1 µm/s increase in blood velocity was associated with a 0.55% reduction in risk of POAG. Mean blood flow was much lower in POAG patients than in controls (27 vs 50 pL/s). Every pL/s increase in blood flow was associated with a 6% decrease in risk of POAG. This association was strong in both high- and normal-tension glaucoma. The investigators postulated that a better understanding of decreased peripheral capillary perfusion in POAG could improve diagnosis and treatment.
1. Samuelson TW. Results from the HORIZON trial: A multicenter, randomized study of a Schlemm’s canal microstent for reduction of IOP in primary open-angle glaucoma. Paper presented at: the 28th Annual AGS Meeting; March 1, 2018; New York.
2. Rhee D, Ahmed I, Fea A, et al. Prospective, multicenter, randomized comparison of stand-alone Hydrus versus two iStents for reduction of IOP in open-angle glaucoma. Paper presented at: the 28th Annual AGS Meeting; March 1, 2018; New York.
3. Ting DSW, Cheung CY, Lim G, et al. Development and validation of a deep learning system for diabetic retinopathy and related eye diseases using retinal images from multiethnic populations with diabetes. JAMA.2017;318:2211-2223.
4. Saeedi O, Boland M, D’Acunto L, et al. Development of a machine learning algorithm to determine visual field progression. Paper presented at: the 28th Annual AGS Meeting; March 2, 2018; New York.
5. Jinapriya D, D’Souza M, Hollands H, et al. Anti-inflammatory therapy after selective laser trabeculoplasty. Ophthalmology.2014;121:2356-2361.
6. Groth S, Goldberg J, Albeiruti E, et al. Steroids After Laser Trabeculoplasty (SALT) trial: Impact of short-term use of anti-inflammatory treatment on the efficacy of SLT. Paper presented at: the 28th Annual AGS Meeting; March 3, 2018; New York.
7. Chou J. Resting nailfold capillary blood flow in primary open-angle glaucoma. Paper presented at: the 28th Annual AGS Meeting; March 3, 2018; New York.





