CASE PRESENTATION
As I mentioned in the August installment of this series (bit.ly/rscase1118),1 the question I have heard from doctors most often lately goes something like this: “I have a postrefractive surgery patient on whom I performed cataract surgery, and now I have a refractive surprise. What can I do to fix it?” I am therefore devoting a few articles to this subject and have recruited experts to provide some answers. Although the panel will discuss fixes for refractive surprises, the goal is prophylaxis for future cases. How can ophthalmologists fine-tune cataract surgery planning in patients with a history of refractive surgery, and how can surgeons prevent postoperative problems with quality of vision? In each case, the presumption is that no refractive information is available prior to the intervention. This month’s case for evaluation follows.
A 59-year-old woman presents with decreased vision in her right eye. The patient underwent uneventful bilateral myopic LASIK for a -2.75 D spherical treatment in 2006, and she wishes to undergo surgery to address a nuclear sclerotic and cortical cataract in her right eye. She tried monovision in the past, and a recent contact lens trial failed. In the patient’s right eye, the UCVA is 20/50, and the BCVA is 20/40 with a manifest refraction of +0.50 +0.75 x 165º. Glare visual acuity is 20/70 (Figures 1–4).
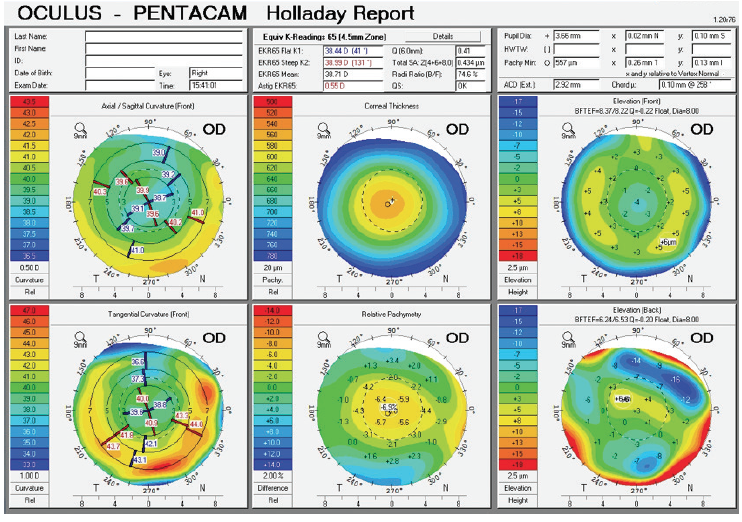
Figure 1. Holladay report on the Pentacam (Oculus Optikgeräte) prior to cataract surgery.
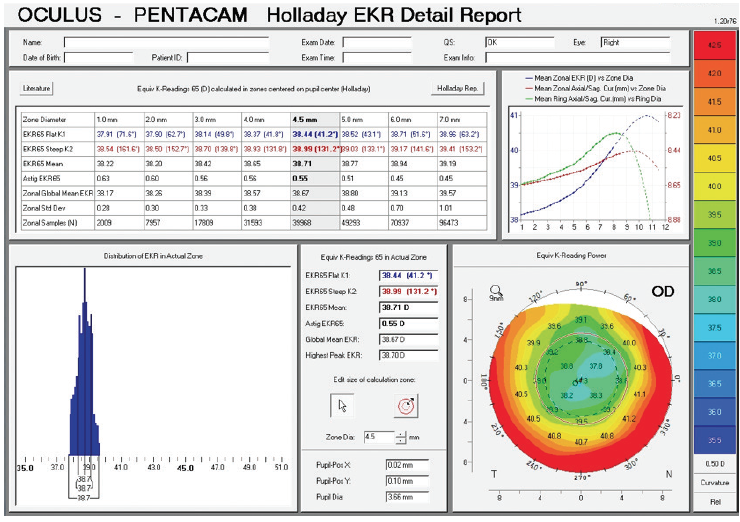
Figure 2. Holladay EKR report on the Pentacam prior to cataract surgery.
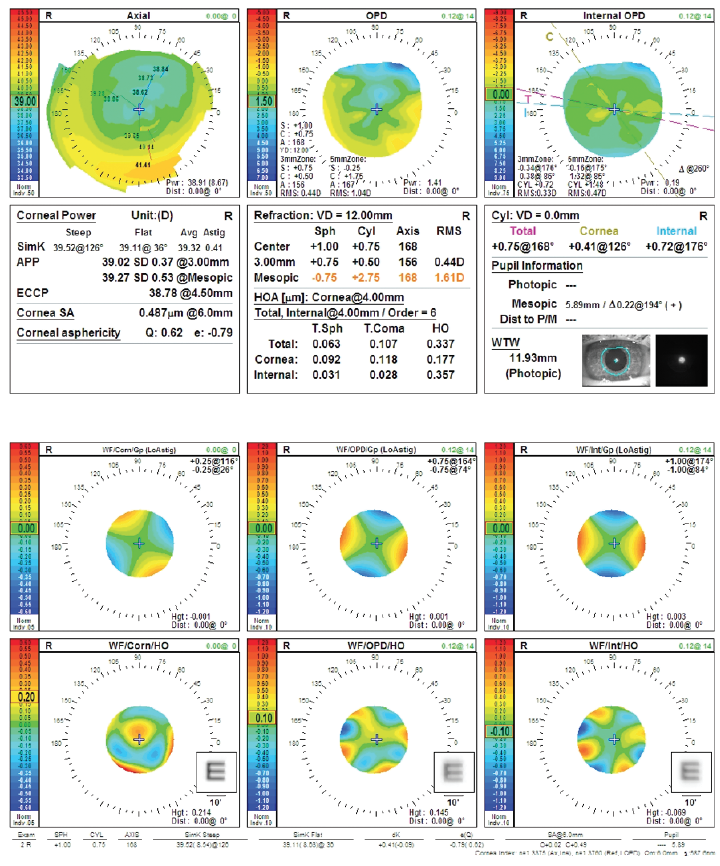
Figure 3. Preoperative measurements (top and bottom) with the OPD-Scan III (Nidek).
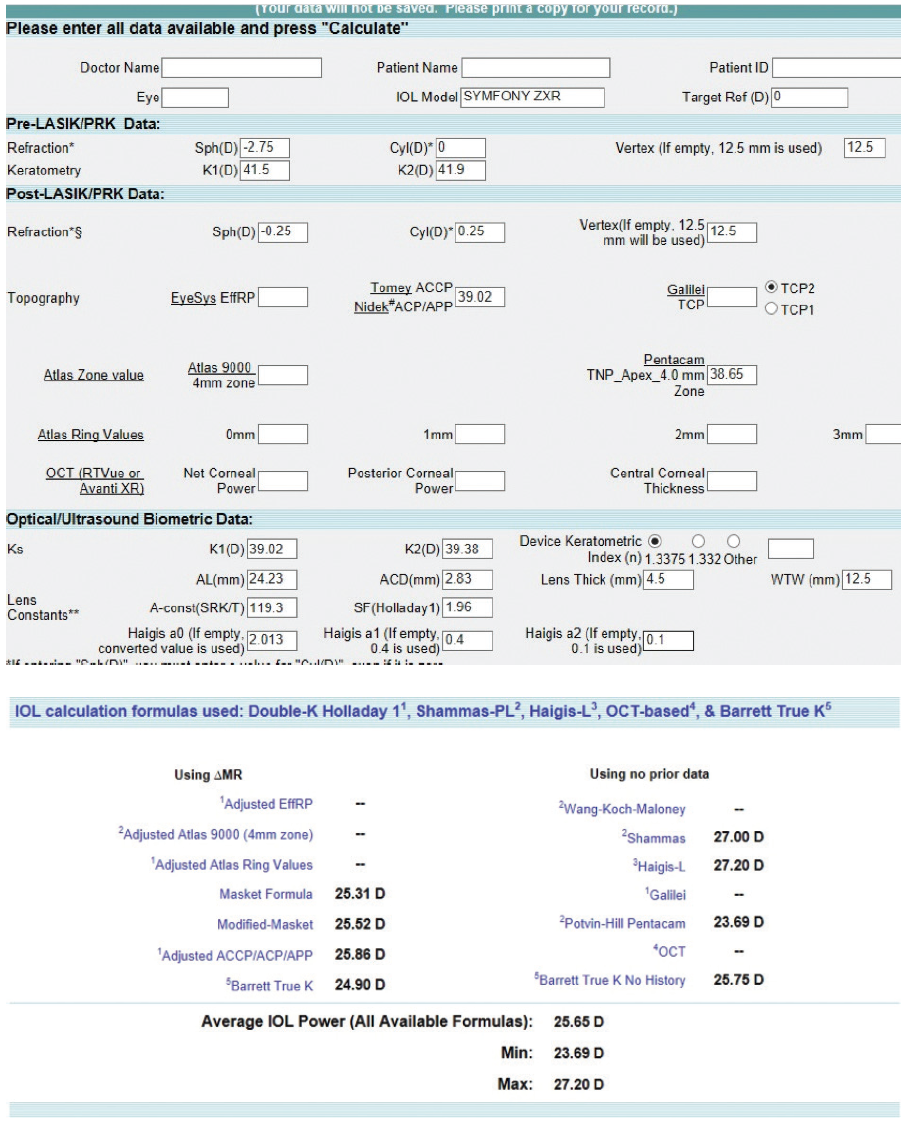
Figure 4. Preoperative calculations using the ASCRS IOL power calculator for prior myopic LASIK/PRK.
Would a multifocal or extended depth of focus IOL be appropriate for this patient?
—Case prepared by Karl G. Stonecipher, MD

RICHARD POTVIN, MASc, OD
Warren E. Hill, MD, FACS, and I looked at this issue specifically, and the calculations are available at no cost on the ASCRS website for interested surgeons.2
Using regression analysis, we estimated post-LASIK/PRK corneal power using the TNP_Apex_Zone4.0 value from the Pentacam rotating Scheimpflug camera (Oculus Optikgeräte), the axial length, and, if available, the anterior chamber depth value obtained with standard biometry.
The ultimate IOL power calculation was based on the Shammas-PL formula.3 Based on this no-history method, 34%, 66%, and 91% of eyes were within ±0.25, ±0.50, and ±1.00 D of the refractive target, respectively.
This method is an accurate option for determining IOL power after LASIK for myopia.

DAVID COOKE, MD
There is a high likelihood of prediction success in this eye for four reasons: (1) the amount of the prior laser correction is known; (2) the amount of laser correction was small (< 3.00 D); (3) there is essentially no astigmatism; and (4) the left column of the ASCRS calculator (known pre- and post-LASIK data) has consistent predictions.
This patient is not likely to have a refractive surprise. I would be quick to consider a premium IOL. I have been impressed with the distance clarity achieved with the Tecnis Symfony IOL (ZXR00; Johnson & Johnson Vision). I would readily suggest a toric IOL to a patient such as this one, even if only mild astigmatism were present.
Of note, we know that the ideal IOL power for emmetropia was 26.00 D. The right column of the ASCRS online calculator (no known preoperative data) shows wildly variable predictions, ranging from 23.69 to 27.20 D. This example illustrates the value of knowing the perirefractive surgical data. These data are often no longer available, yet perhaps half of my patients are able to estimate their preoperative reading distance (before and after laser vision correction). From this information, I generate approximate pre- and postoperative glasses prescriptions for my technicians to enter into the ASCRS calculator.

BRET L. FISHER, MD
I routinely implant monofocal, toric, and multifocal IOLs in patients who have undergone myopic LASIK. If they had success with monovision in the past, I am willing to re-create monovision for them with monofocal or toric IOLs, but I explain to them the greater range of vision that would be possible with bilateral multifocal IOLs. At present, my recommendation for these patients when they want to minimize their need for glasses is the AcrySof IQ ReStor +2.5 D IOL with Activefocus (Alcon), in either the toric or nontoric model, as required. In my experience, this IOL gives patients monofocal-like distance vision and good intermediate and near vision with little night vision disturbance.
In a retrospective review of 44 eyes of 31 post-LASIK patients undergoing cataract surgery, Dr. Potvin and I found equivalent uncorrected distance visual acuity among those who received the low-add distance-dominant AcrySof IQ ReStor +2.5 D Multifocal Toric IOL with Activefocus optical design and a monofocal control group.4 The refractive accuracy using either intraoperative aberrometry or the Barrett True K calculation from the ASCRS calculator for IOL power selection was high for both the multifocal and monofocal group and equivalent between them.
Measuring and treating astigmatism in these patients is often more difficult than in eyes with unoperated corneas. Preoperatively, I typically obtain Placido ring–based topography measurements with the Atlas 9000 Corneal Topography System (Carl Zeiss Meditec), Pentacam imaging, and measurements with the IOLMaster (Carl Zeiss Meditec), Lenstar LS900 (Haag-Streit), and an autokeratometer to make a preliminary estimate of astigmatism. These readings frequently conflict. If I can find no overall pattern or congruence, I treat the patient for dry eye and ocular surface disease and repeat the measurements several weeks later, typically on the day of surgery. In every case, however, I obtain intraoperative aberrometry measurements and most often use this information to finalize the IOL selection. I find that I am using more toric and multifocal toric IOLs now than in the past, whereas my frequency of astigmatic keratotomy has decreased because it must be performed before aberrometry.

UDAY DEVGAN, MD, FACS, FRCS
The preoperative corneal values are reasonable. On the OPD-Scan III WF/Corn/HO printout, the convolved E looks pretty good (Figure 3), indicating a low degree of corneal aberrations after the LASIK treatment. An extended depth of focus or multifocal IOL could work well in this eye. The odd thing is that the patient had very flat corneas before LASIK but nonetheless achieved a good result.

WHAT I DID: KARL G. STONECIPHER, MD
I implanted a 26.00 D Tecnis Symfony IOL (model ZXROO, Johnson & Johnson Vision) in the patient’s right eye. Six months after surgery, UCVA was 20/20, and BCVA was 20/20 with a manifest refraction of -0.25 +0.25 x 157º. Results were similar for the contralateral eye, which underwent cataract surgery and implantation of a Tecnis Symfony IOL 1 week after the right eye. No additional intervention has been indicated for either eye.
1. Stonecipher KG, Holladay JT, Cooke D, et al. Previous eight-incision radial keratotomy. Cataract & Refractive Surgery Today. August 2018;18(8):35-42.
2. Potvin R, Hill W. New algorithm for intraocular lens power calculations after myopic laser in situ keratomileusis based on rotating Scheimpflug camera data. J Cataract Refract Surg. 2015;41(2):339-347.
3. Shammas HJ, Shammas MC. No-history method of intraocular lens power calculation for cataract surgery after myopic laser in situ keratomileusis. J Cataract Refract Surg. 2007;33:31-36.
4. Fisher B, Potvin R. Clinical outcomes with distance-dominant multifocal and monofocal intraocular lenses in post-LASIK cataract surgery planned using an intraoperative aberrometer. Clin Exp Ophthalmol. 2018;46(6):630-636.




