

Last year, we celebrated 50 years since Charles D. Kelman, MD, performed the first phacoemulsification in a human in 1967. He later said of that operation that the emulsification time was well over 1 hour, and the total time in the operating room was 4 hours (see Dr. Kelman’s accompanying excerpt below).
How things have changed in 51 years! Straightforward phaco cases now take 10 minutes or less, and the associated techniques and technologies have improved commensurately. Let’s have a look at how phacoemulsification is being done this year and where it may go in the future.
An excerpt from CRST March 2004
The Genesis of Phacoemulsification
A look back by the originator of the technique.
By Charles D. Kelman, MD
Four years after my residency, I drafted a grant proposal to study the effects of freezing on the ciliary body, retina, and choroid. I went to bed concerned that the Hartford Foundation would not find the topic of interest, awoke in the middle of the night, and, almost in a trance, added an addendum to my application that would affect the rest of my life and the lives of 100 million patients: “In addition to the freezing studies, this investigator will develop a method for removing a cataract through an incision small enough so that no hospitalization will be required.”
Mr. E. Pierre Roy, the head of the John A. Hartford Foundation, could easily have rejected my application and put an end to this matter. Instead, he had confidence in my abilities and gave me a 3-year grant, although I did not have the vaguest idea of how to realize my idea.
INITIAL STRUGGLES
Mr. Roy’s confidence was misplaced for 2 years and 8 months, while I tried everything I could imagine. I first attempted to capture the cataract within a folding lens bag, crush it inside the bag with manual disintegrators, and then remove the device containing the fragmented lens material from the eye (Figure 1). The rotating devices I tried simply spun the cataract around inside the anterior chamber. High-speed cutting needles, a miniature blender, drills, tiny meat grinders, engraving tools—nothing worked. All the devices yielded opaque corneas in animal eyes.

Figure 1. One of the many unsuccessful devices created by the author for smallincision cataract surgery involved folding lens bags and manual disintegrators.
I had, meanwhile, allowed my hair to grow down to my shoulders, and my teeth badly needed a cleaning. Sitting in my dentist’s chair, I became interested in the ultrasonic tool he was using to clean my teeth. He explained that its high-frequency vibration removed tartar without disturbing the tooth itself. I raced out of his office with the bib still hanging around my neck and returned 1 hour later with a cataractous lens. Because I was able to engrave lines on the lens without it jumping off my finger, I believed that I could break up a cataract inside the eye without it spinning or vibrating against the corneal endothelium.
Cavitron (acquired by Coopervision, followed by Alcon) ultimately made a prototype with a handpiece that incorporated I/A (Figure 2), rather than only irrigation, as with the original dental instrument. My first efforts with this device resulted in opaque corneas until I began using a physiologic solution, imported from the Centro de Oftalmologia Barraquer, in Barcelona, Spain, in place of the simple saline solution. I also realized that the high temperatures that the procedure created inside the eye would denature the corneal proteins.
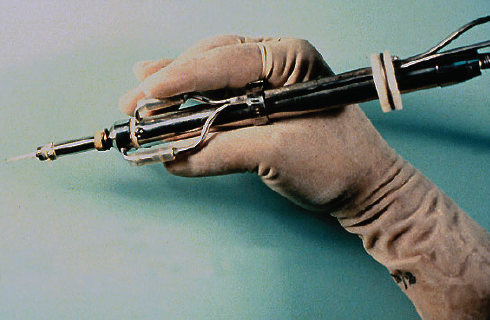
Figure 2. An early prototype of the phaco handpiece.
After several months of successful animal testing, I operated on my first human case—a man suffering from a painful blind eye due to burned-out glaucoma. The strong surge of suction caused the cornea to collapse 30 to 40 times during the 70-minute phacoemulsification. The next day, his eye was a bag of pus that had to be removed.
I spent 2 years seeking a way to prevent corneal collapse and finally found a company in North Carolina that manufactured a device that sensed arterial flow. Cavitron incorporated this feature into the phacoemulsification device’s aspiration line with an air-relieved valve. If the speed of the current in the aspiration line exceeded the speed of the irrigation, then a valve opened to halt suction immediately.
One year later, I worked up the nerve to operate on the eye of a patient with central retinal artery occlusion and no light perception. If I had failed that time, I probably would have abandoned the project. A specially designed, 3D micromanipulator supported the weight of the cumbersome phaco handpiece and steadied the phaco tip inside the eye for long periods of time. I deemed the operation a success when the patient achieved light perception postoperatively. The severe striate keratitis present on the first postoperative day subsided after 6 weeks.
HURDLES
The hospital. At that time, there were no rules against performing new procedures. Nonetheless, after I had successfully completed a few more cases, the chief surgeon at Manhattan Eye and Ear Hospital in New York tried to convince the board to rescind my operating privileges. Ultimately, they established a committee to oversee the results of every patient. Fortunately, by this time, my results were quite good.
The profession. Although I began teaching phacoemulsification in 1970 with the first commercially available machine, the profession vehemently protested the adoption of the technique. Most if not all surgeons at that time used only loupes for magnification. As a result, they had to learn to use an operating microscope as well as to perform phacoemulsification. Many politically important ophthalmologists were older and either unwilling or unable to learn these new modalities.
The companies. The lead engineer at Cavitron Corporation had encoded and threatened to withhold all the critical information about the new phacoemulsification machine in a play for a promotion to vice president around 1971. Cavitron Corporation fired him, but the company was then ready to abandon the project. Fortunately, Alan McMillan, an entrepreneur from California, took over the project and developed a model that he eventually sold to Coopervision and, finally, Alcon.
The study. The American Academy of Ophthalmology initiated a major comparative study of phacoemulsification and intracapsular cataract extraction (ICCE). At my insistence, the chairman of the department of mathematics and statistics at Columbia University in New York monitored the study’s results. The investigators’ reluctant conclusion was that phacoemulsification delivered the same quality of results as ICCE, but the report’s conclusion stated that ICCE was the preferred and only method to use in many instances.
The classification. The next major attack on phacoemulsification occurred when opponents got the FDA to classify the procedure as experimental and, therefore, nonreimbursable. Reversing this decision required thousands of letters from patients and an appearance before the FDA by the renowned television doctor, Marcus Welby, MD [the actor Robert Young].
The IOL. As IOLs became more popular in the late 1970s, many surgeons gave up on phacoemulsification because implanting the lenses required larger incisions. The advent of lenses that could fit through a smaller incision, such as the Omnifit (Precision Cosmet Company and Heyer Schulte) and foldable IOLs, revived the procedure.
SURGICAL CONSIDERATIONS
Incision size for most surgeons now is between 2 and 2.4 mm, but, despite the arrival of the femtosecond laser 10 years ago, these incisions are still almost all made with knives, even by laser users. The reasons for this are that incisions made by the laser are rather corneal in position and that they may be difficult to open, particularly if there is any arcus senilis present.
Increasingly the pupil is being dilated on the operating table with products containing multiple active ingredients, such as Mydrane (tropicamide 0.2 mg/phenylephrine HCl 3.1 mg/lidocaine HCl 10 mg injectable solution, Thea; not available in the United States). This saves time preoperatively and reduces costs, according to Davey et al.1 If a femtosecond laser is to be used for the capsulotomy, however, drops will be required to maintain mydriasis.
CAPSULOTOMY ISSUES
The accuracy of sizing, circularity, and centration of the capsulotomy was revolutionized with the introduction of the femtosecond laser, and some IOL manufacturers, such as Oculentis and Morcher, have begun making lenses to take advantage of these characteristics. It seems likely that others will follow suit.
Although some studies have shown that visual outcomes are not influenced by the relationship of the capsulotomy to the IOL in the short term, early posterior capsular opacification (PCO) is likely when the IOL optic is not fully covered by the capsulotomy edge. Additionally, IOL centration is important for premium IOLs such as multifocals.
Two other recently introduced technologies can also provide accurate capsulotomies. The Zepto (Mynosys) is a device inserted into the eye and placed on the anterior capsule. Suction is applied via a silicone ring, and a nitinol ring inside the device thermoelectrically cuts the capsule to create a 5.2-mm capsulotomy. Another device, which requires its own special laser, is the CapsuLaser (Excel-Lens). The anterior capsule is stained with a special formulation of trypan blue dye to provide a target for the laser, the wavelength of which is in the red-orange part of the spectrum. The laser fires in a continuous manner for 1 sec to create the capsulotomy, which can be sized from 4.5 to 5.5 mm in 0.1-mm steps. A target light for the patient assists in centration. CapsuLaser now has the CE Mark. Both it and the Zepto produce strong capsulotomies.
NUCLEUS REMOVAL
For removal of the nucleus, again the femtosecond laser brings advantages. Studies have shown that the amount of ultrasound energy for phacoemulsification can be reduced by creating fragmentation patterns in the nucleus. Although this is certainly an advantage for the surgeon, studies so far comparing this laser with standard phacoemulsification have shown no benefit for patient outcomes.2
When this lack of effect on outcomes is considered along with the capital and ongoing maintenance costs of these expensive devices, it is not surprising that initial enthusiasm has not translated into more widespread adoption of femtosecond laser platforms. Use of these lasers has been seen almost entirely in the private (ie, nonstate-supported) sector of ophthalmic care. In fact, a recently completed French investigation confirmed that use of laser cataract surgery is not cost effective.3

IMPROVING SAFETY
The manufacturers of phaco machines have tried over many years to improve the safety of cataract removal. A number of technologies have been introduced, including power modulation modalities, such as microburst mode, the WhiteStar technology (Johnson & Johnson Vision), and Intelligent Phaco on the Infiniti Vision System (Alcon), and alternative tip movements such as Ozil (Alcon) and Ellips (Johnson & Johnson Vision).
These innovations have been available for some years, but development continues in the area of fluidics technologies. The capability to use some form of positive irrigation pressure is now found on many phaco machines. The Stellaris (Bausch + Lomb), the Eva (Dutch Ophthalmic), and the R-Evolution R (Optikon; not available in the United States) all have pumping mechanisms that push out irrigation fluid to maintain anterior chamber stability.
The disadvantage of this approach is that IOP is very high. The Centurion (Alcon) uses a different approach, wherein the IOP level is chosen by the surgeon. It can be as low as the high 20s mm Hg or as high as 80 mm Hg—the equivalent of a bottle height of 110 cm H2O. The IOP is maintained by a bag within the machine that is squeezed by a paddle as required to ensure anterior chamber stability.
MORE CHANGES AHEAD
Several companies, including Johnson & Johnson Vision, Bausch + Lomb, and Carl Zeiss Meditec, have plans in place to introduce new phaco machines, all with the aim of trying to minimize the use of ultrasound power and maximize anterior chamber stability during nucleus removal.
Phacoemulsification in 2018 continues to move forward, bringing better, safer outcomes for our patients. If he were here today, Charlie Kelman would be delighted.
1. Davey K, Chang B, Purslow C, Clay E, Vataire AL. Budget impact model of Mydrane, a new intracameral injectable used for intra-operative mydriasis, from a UK hospital perspective. BMC Ophthalmol. 2018;18(1):104.
2. Popovic M, Campos-Möller X, Schlenker MB, Ahmed II. Efficacy and safety of femtosecond laser-assisted cataract surgery compared with manual cataract surgery: a meta-analysis of 14 567 eyes. Ophthalmology. 2016;123(10):2113-2126.
3. Schweitzer C. Evidence from the French FEMCAT study. Presented at: the European Society of Cataract and Refractive Surgeons meeting; October 7-11, 2017; Lisbon, Portugal.




