Case Presentation
A 54-year-old man presents with a complaint of glare, blurry vision, and an inability to view the television or read 2 months after undergoing the repair of a ruptured right globe. The rupture was caused by an injury from a fishing weight that also induced hyphema, vitreous hemorrhage, retinal detachment, and eyelid laceration. The patient is status post anterior chamber washout for hyphema, pars plana vitrectomy for vitreous hemorrhage, and globe closure. He is currently administering IOP-lowering topical drops.
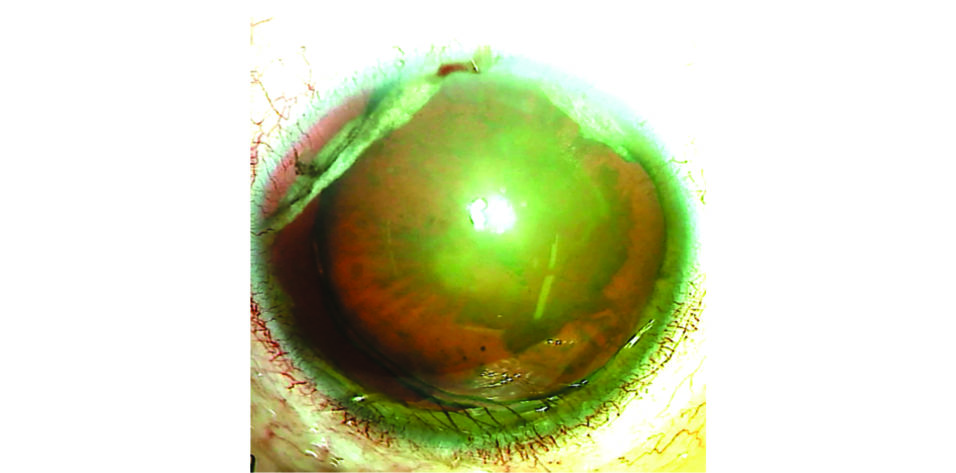
On examination, the patient’s BCVA is counting fingers OD. The IOP is 11 mm Hg OD. The eyelids exhibit mild ptosis that is more prominent on the right. The conjunctiva of the right eye exhibits scarring from previous surgeries. The cornea is clear, and the anterior chamber is deep and quiet. There is a large iridodialysis superotemporally, a second iridodialysis superonasally, and mydriasis. The lens is nasally subluxated (Figure 1), and the view to the fundus is poor because of a significant cataract.
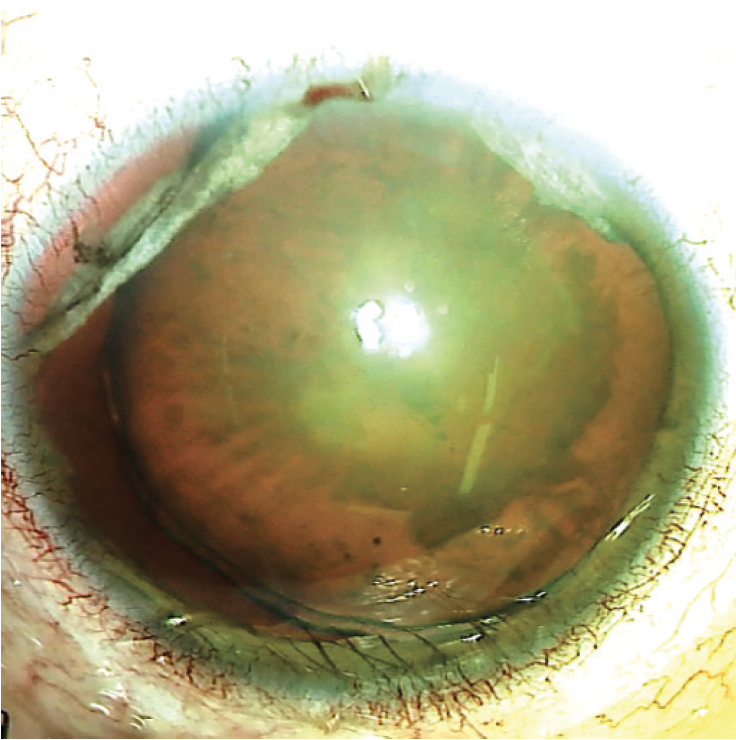
Figure 1. Preoperative appearance of the right eye. Note the superior iris defects, dilated pupil, and nasally subluxated cataract.
How would you manage the posttraumatic subluxated cataract and large iris defect?
—Case prepared by Brandon D. Ayres, MD, and Joanne S. Francis, MD

ASHVIN AGARWAL, MS
Visualization and stabilization are crucial in situations such as this one. The conjunctival scarring and IOP indicate a history of retinal detachment surgery.
First, a standard capsulorhexis would be performed; a slight lack of countertraction may render the maneuver somewhat difficult. Next, four capsular hooks would be placed to stabilize the capsule, keep vitreous behind the bag, and prevent the iris from coming into contact with the phaco probe. The bottle height would be lowered to compensate for weak zonules, and cataract surgery would be performed.
If the capsular bag is intact, then an IOL would be placed in the bag and fixated using an Ahmed Segment (FCI Ophthalmics) and a double-flanged technique with a 6-0 polypropylene (Prolene, Ethicon) suture that passes through the islet of the segment and out of the sclera at the site of maximum subluxation. If the bag has been compromised, then fibrin glue–assisted scleral fixation of the IOL would be performed. Use of the Yamane technique1 is another option in this situation.
Because the iridodialysis is located toward the superior side, I would perform the iris repair with a hang-back or sewing machine technique from a temporal position to maximize access. The techniques are equally effective in my experience but must be coupled with a single-pass four-throw pupilloplasty to correct pupil-related defects that typically become evident after iridodialysis repair.

D. BRIAN KIM, MD
An evaluation for an afferent pupillary defect is crucial in this situation. The patient had a ruptured globe and should be evaluated for traumatic optic neuropathy and glaucoma because he is on IOP-lowering medication. These factors and an assessment of the macula and cornea should guide preoperative counseling of the patient, including the setting of realistic expectations regarding visual potential.
A big question in this situation is whether to save or sacrifice the capsular bag. As a rule, the bag should be saved whenever possible, but doing so here may require surgical gymnastics, including the use of capsule retractors, a capsular tension ring (CTR), a scleral-sutured capsular tension segment (CTS), and a careful phaco technique. If the zonules are tenuous, then the risks of a posterior capsular rupture, vitreous loss, and corneal decompensation are greater. Another issue to consider is that the tissue over the iridodialysis is white and atrophic and may be associated with tissue loss, which raises the possibility of a cheese-wiring effect if scleral fixation or pupillary cerclage is attempted.
In light of these challenges, if the bag cannot be salvaged, one approach is to perform intracapsular cataract extraction through a scleral frown incision like the one used for manual small-incision cataract surgery to minimize cylinder and promote a watertight seal. A thorough anterior vitrectomy would be executed through a pars plana trocar. A Customflex ArtificialIris (VEO Ophthalmics) would be trimmed to the appropriate diameter, and a pair of stab incisions would be made 180º apart in the midperipheral part of the artificial iris. The haptics of a CT Lucia 602 IOL (Carl Zeiss Meditec) would be threaded through and sutured to the prosthetic device with 9-0 polypropylene to prevent slippage. The entire prosthetic device–IOL complex would then be placed through the scleral incision and fixated to the scleral wall using the Yamane technique.1 Care would be taken to ensure that the prosthetic is positioned behind the iris plane.

CATHLEEN M. MCCABE, MD
A complete evaluation of the patient’s ocular health is the first step in planning for cataract surgery after blunt trauma—especially after an open globe repair with evidence of intraocular sequelae of trauma. Information from OCT of the macula and optic nerve, gonioscopy, an endothelial cell count, and anterior segment ultrasound can help to determine which procedures must be performed, which complications may be encountered during surgery, and what to discuss with the patient regarding reasonable expectations. In the current situation, the surgical plan would include repairing the iris, managing traumatic mydriasis by decreasing the pupil size, supporting the capsule during cataract removal, supporting the IOL after cataract removal, and managing the IOP. The specifics of the plan and the instruments and devices required should be noted in the medical record to ensure that the necessary supplies are on hand on the day of surgery.
A retrobulbar block would be administered. After the instillation of a copious amount of a dispersive OVD, one or more paracenteses would be created in the areas of iridodialysis. Iris hooks would be placed to protect the iris during phacoemulsification. The use of a double-flanged technique with polypropylene sutures to repair the iridodialysis (6-0 polypropylene) and fixate a CTS (5-0 polypropylene) can be helpful in the setting of conjunctival scarring.2,3 Using low-temperature cautery, a flange would be created that is small enough to bury in the superficial layers of a scleral tunnel to minimize the risk of late extrusion of the flange through the conjunctiva.
In similar situations, after completing the capsulorhexis, I have used the Canabrava double-flanged technique3 to support the capsule in the area of zonular dialysis. This strategy can allow a CTS to function as a support system during phacoemulsification and for the IOL over the long term.
After cataract removal, atraumatic insertion of a CTR into the capsular bag would be facilitated with a Sinskey hook to keep the leading end of the CTR central in the bag until the device is fully deployed. Iridodialysis repair would be performed using the technique described by Eric D. Rosenberg, DO.4 Next, the mydriatic pupil would be addressed by either performing a pupillary cerclage or placing interrupted 10-0 polypropylene sutures with the four-throw pupilloplasty technique described by Priya Narang, MS.5
I would consider performing a goniotomy or endocyclophotocoagulation to improve IOP control. An intraocular depot steroid and intracameral antibiotics would be administered. Postoperatively, I would advise the patient not to rub the eye and to return as required for IOP monitoring.

SIMONETTA MORSELLI, MD
A photograph of the contralateral eye would be obtained to allow an IOL with a color-matched iris diaphragm to be ordered. The IOL would be placed in the capsular bag or sulcus or fixated to the sclera depending on the integrity of the capsular bag and zonules. I would have a standard and a Cionni CTR (Morcher) on hand.
Surgery would be performed under general anesthesia. A 2.2-mm clear corneal incision would be created. A cohesive OVD would be injected into the anterior chamber, and the capsulorhexis would be performed. Gentle hydrodissection and phacoemulsification would be performed with a low bottle height and aspiration rate. The anterior chamber and stability of the capsular bag would be assessed during these maneuvers.
Assuming the capsular bag is intact, it would be stabilized by placing a standard CTR after irrigation and aspiration are complete. The IOL would then be placed in the capsular bag. If the capsular bag is not centered, then a Cionni CTR would be used to fixate the bag to the limbus, and the IOL would be placed in the bag. If vitreous is detected in the anterior chamber, then triamcinolone acetonide would be instilled to aid visualization, and an anterior vitrectomy would be performed. If the capsular bag is unstable and scleral fixation is not possible, then the capsular bag would be removed, after which an iris diaphragm IOL would then be fixated to the sclera.


WHAT WE DID: BRANDON D. AYRES, MD, AND JOANNE S. FRANCIS, MD
The surgical options for the patient included complex cataract extraction by phacoemulsification and IOL placement with iridoplasty or staged as a secondary iris repair. Important points to consider in this situation are the likelihood of zonular instability because of the history of trauma and a nasally displaced lens. An intraoperative assessment is required to evaluate the quality of the remaining iris tissue. Alternatively, an artificial iris implant could be considered for placement in the sulcus. If the eye has no capsular support, an IOL could also be secured to the back of an artificial iris implant and sutured to the sclera.
The risks and benefits of and alternatives to surgery were discussed extensively with the patient. He elected to undergo cataract extraction combined with IOL placement and iridoplasty. He was informed that an anterior vitrectomy would be performed and the IOL sutured to the sclera if necessary.
Limbal paracenteses were created peripherally. Lidocaine and an OVD were injected into the anterior chamber. A 2.4-mm clear corneal incision was created temporally. The anterior capsule was stained with trypan blue dye. A continuous curvilinear capsulorhexis was initiated with a cystotome and completed with Utrata forceps. Two microsurgical capsule retractors were placed through limbal paracenteses to secure the temporal capsule. Hydrodissection and phacoemulsification were performed to remove the nucleus. Bimanual irrigation and aspiration were used to remove the remaining cortical material. Stability of the capsule was found to be sufficient to support a one-piece IOL in the capsular bag (Figure 2).
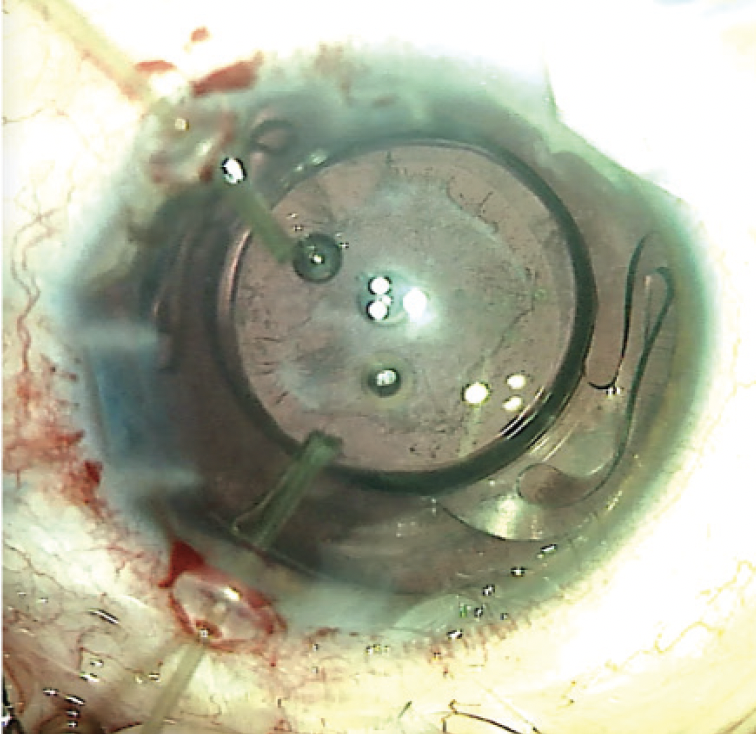
Figure 2. Microsurgical capsule retractors provide adequate stability for standard phacoemulsification to be performed and a one-piece IOL to be placed.
Iris repair followed. Microholding forceps were used to test the integrity of the iris tissue. Atrophic tissue was noted superiorly with atonic iris tissue inferiorly that was better suited to hold suture material. A 10-0 polypropylene suture on a long, curved needle was inserted through the temporal wound, threaded through the remaining superior iris tissue, and externalized through a superonasal paracentesis (Figure 3A). The Siepser technique was used to tie a knot with the assistance of an intraocular snare.6,7
Next, a conjunctival peritomy was created superotemporally for iridodialysis repair. A double-armed 10-0 polypropylene suture on a long, straight needle was inserted through the inferotemporal paracentesis, threaded through the superior iris, and externalized through a 27-gauge needle (Figure 3B). The second needle was passed through adjacent iris tissue and tied. A second double-armed iridodialysis suture was placed to secure the rest of the superotemporal iris (Figure 3C). Two additional 10-0 polypropylene iridoplasty sutures on a curved needle were placed superotemporally and inferiorly and tied (Figures 3D and 3E). The conjunctival peritomy was closed with an 8-0 polyglactin suture (Vicryl, Ethicon). Irrigation and aspiration were performed to remove the residual OVD, and the incisions were hydrated with balanced salt solution.
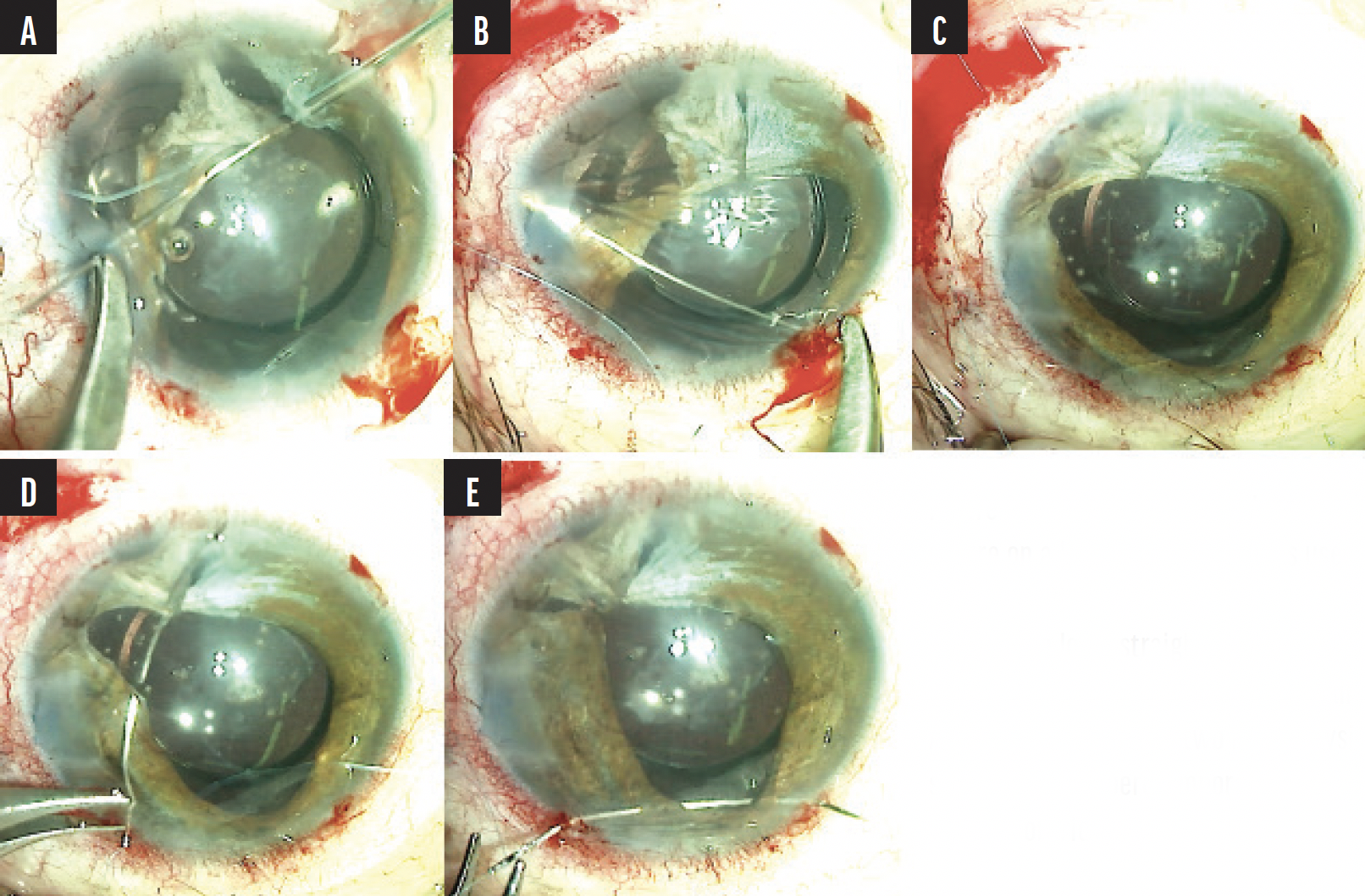
Figure 3. Iris repair. A 10-0 polypropylene suture on a long, curved needle is used for a superior iridoplasty (A). A 10-0 polypropylene suture on a long, straight needle is used for superotemporal iridodialysis repair (B). After the placement of two iridodialysis sutures (C). A superotemporal iridoplasty (D). An inferior iridoplasty (E).
One day after surgery, the patient’s UCVA was hand motions in the right eye. The IOP was 24 mm Hg. The cornea exhibited mild, diffuse microcystic edema, and the anterior chamber was deep with moderate cell. The polypropylene suture was well covered by conjunctiva superiorly. The pupil was relatively round and approximately 4 mm in diameter. The IOL was well centered (Figure 4). A postoperative regimen of topical steroids and antibiotics was initiated. One week after surgery, the patient’s UCVA was 20/70, and the corneal edema had resolved.
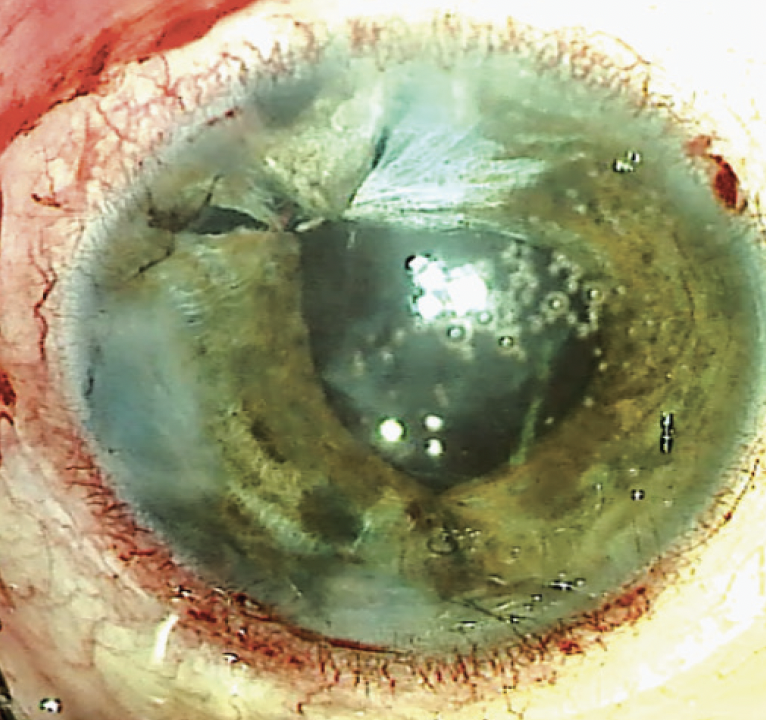
Figure 4. After surgery, the IOL is well positioned, and the pupil is smaller and round.
1. Yamane S, Sato S, Maruyama-Inoue M, Kadonosono K. Flanged intrascleral intraocular lens fixation with double-needle technique. Ophthalmology. 2017;124(8):1136-1142.
2. Kusaka M, Miyamoto N, Akimoto M. Repairing iridodialysis by riveting with a double-flanged polypropylene suture. J Cataract Refract Surg. 2019;45(11):1531-1534.
3. Canabrava S, Bernardino L, Batisteli T, Lopes G, Diniz-Filho A. Double-flanged-haptic and capsular tension ring or segment for sutureless fixation in zonular instability. Int Ophthalmol. 2018;38(6):2653-2662.
4. Rosenberg ED, Levi MA, Nattis AS. In-N-out technique: double-flanged suture ab interno iridodialysis repair. J Cataract Refract Surg. 2022;48(2):251-253.
5. Narang P, Agarwal A. Single-pass four-throw technique for pupilloplasty. Eur J Ophthalmol. 2017;27(4):506-508.
6. Osher RH, Snyder ME, Cionni RJ. Modification of the Siepser slip-knot technique. J Cataract Refract Surg. 2005;31(6):1098-1100.
7. Lian RR, Siepser SB, Afshari NA. Iris reconstruction suturing techniques. Curr Opin Ophthalmol. 2020;31(1):43-49.