CASE PRESENTATION
A 52-year-old man presents for a refractive surgical evaluation. The patient has an ocular history of congenital nystagmus and a recorded BCVA of 20/30 OU. He normally wears contact lenses for distance but says he has been having difficulty with them recently. He also reports a daily struggle to focus in his null point while wearing reading glasses.
The patient is a business owner and property manager by profession and desires good uncorrected distance and near visual acuity (UDVA and UNVA). He notes that night vision is also important because he frequently works evenings. The patient states that he understands the limitations of refractive surgery and says that he does not mind wearing readers to see very fine print (J2 or smaller). His stated goal, however, is to be as independent as possible from spectacles.
On examination, refraction is -6.75 +0.25 x 6º = 20/30 BCVA OD and -10.00 +2.50 x 86º = 20/30 BCVA OS. Keratometry readings are 45.80/46.20 D @ 60º OD and 44.80/47.30 D @ 88º OS. A comprehensive ophthalmic examination confirms congenital nystagmus, a null point with gaze slightly right of center, and myopic-looking retinas without evidence of lattice degeneration or other pathology. Pachymetry readings are 544 and 549 µm in the right and left eyes, respectively. IOP is 18 mm Hg OU. Axial length is 24.53 mm in the right eye and 25.15 mm in the left eye. Topographic measurements are shown in the Figure.
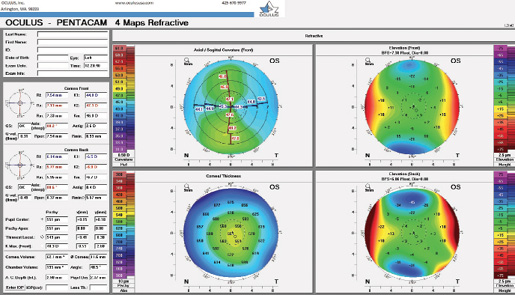
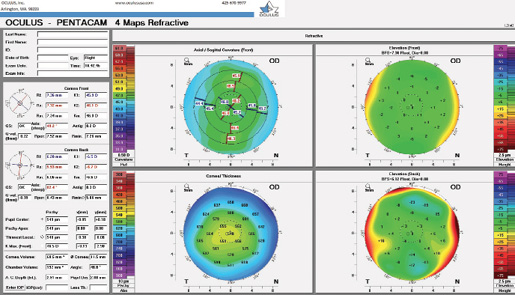
Figure. Preoperative topography of the patient’s left and right eyes.
What treatment options would you consider for this patient? Are there any special considerations for informed consent? How would you proceed?
—Case prepared by Denise M. Visco, MD, MBA

QUENTIN B. ALLEN, MD
A discussion of the risks of refractive surgery during informed consent will be important for many reasons in this case. The patient is at high risk of refractive shifts during the next 5 to 10 years due to early cataract formation. Assessing lens clarity and recent refractive stability will be important to verify surgical candidacy. The ideal procedure for a patient of this age is lens-based, but myopic men are at increased risk of retinal detachment after refractive lens exchange, especially when a posterior vitreous detachment is not already present. Cornea-based refractive surgery may therefore be a better option for this patient.
Corneal refractive surgery in a patient over 50 years old requires extensive counseling and clearly stated expectations. The risk of dry eye disease increases with age, and the condition may worsen after surgery. Thorough screening and treatment for preexisting dry eye are therefore necessary.
This patient has expressed a desire for excellent UDVA and UNVA. Given his age, these can be achieved only with some degree of monovision correction. A monovision trial would be helpful because it would demonstrate to the patient both the visual impact of losing binocular summation and the impact of monovision on night driving.
Given the patient’s high myopia and myopic astigmatism, LASIK would be my preferred procedure if corneal thickness allows. Based on this patient’s corneal thickness, the residual stromal bed should be more than 300 µm in each eye, although the percentage of tissue altered in the more myopic left eye would approach 40%. Topography shows symmetric regular astigmatism in each eye without features suggesting high risk for ectasia. I would recommend to the patient targeting distance correction for both eyes because his BCVA is already reduced by nystagmus. In my experience, patients such as this one can do very well with refractive surgery, especially when a femtosecond laser is used to create the LASIK flap and modern pupil-tracking technology neutralizes saccadic movements during the excimer laser treatment. In some cases, however, manual fixation with forceps is required.
The informed consent should address the expected variability in outcome and the increased risk of under- or overcorrection because of the patient’s high myopia and nystagmus. If the patient understands the risks and has appropriate expectations, it would be reasonable to proceed with LASIK.

KENNETH A. BECKMAN, MD, FACS
This interesting case poses several dilemmas. Nystagmus and difficulty finding the null point in glasses make functioning with spectacles problematic, but the patient is also becoming intolerant of his contact lenses. It is no wonder that he desires as much independence from spectacles and contact lenses as possible, but, unfortunately, he faces several limitations with regard to surgical options.
Topography for the left eye is suspicious, and my concern about possible post-LASIK ectasia would be high, particularly considering the amount of treatment required. High myopia signals potential problems even with PRK (eg, haze). Additionally, the patient’s inability to fixate may make him a poor candidate for any form of laser vision correction. Because he wants to minimize his dependence on readers, the target of any corneal refractive procedure would likely be monovision. In a patient whose distance visual acuity is already limited by nystagmus, targeting myopia in one eye to provide good UNVA may render driving at night difficult. Because the patient is in his early 50s, he may retain some accommodative ability and not require as much myopia in the near eye as a pseudophakic patient would.
Lens-based surgery is another option, but his high myopia increases the risk of retinal complications. Additionally, planned monovision could present problems because he may require more myopia in the near eye to read as a pseudophakic patient, further compromising his ability to see at distance. Moreover, his irregular corneas and significant astigmatism may negatively affect his ability to tolerate multifocal IOLs.
That said, I believe he could benefit from refractive lens exchange. One approach would be to place a monofocal lens in the right eye and a monofocal toric lens in the left. An alternative would be an extended depth of focus IOL implanted either bilaterally or in the nondominant eye only, which could offer some near visual acuity while disturbing distance visual acuity less than a standard multifocal lens would. A third approach would be to implant enVista monofocal lenses (Bausch + Lomb) and target distance in the right eye and mini-monovision in the left. Many surgeons have reported anecdotally that this IOL seems to improve UNVA and extend depth of focus. Bilateral implantation of enVista IOLs could therefore be the best choice for this patient because it would avoid the possibility of his perceiving aberrations from the rings of a multifocal or traditional extended depth of focus IOL.
If operating on a clear lens in a high myope is a concern and a nonsurgical approach is preferred, I would try to optimize the ocular surface as much as possible and refit the patient’s contact lenses. I suspect that he could do well with a scleral lens. He might then consider monovision, leaving his near eye slightly myopic. With-the-rule astigmatism might afford him a slightly multifocal cornea and perhaps somewhat improved near visual acuity. This strategy would also allow him to obtain a distance contact lens for the nondominant eye, which he could wear when he desires optimal distance visual acuity.

WHAT I DID: DENISE M. VISCO, MD, MBA
Although this patient was not an ideal candidate for refractive surgery, he was highly motivated, had an agreeable personality, and possessed a good understanding of the potential limitations of surgical intervention. Because of the congenital nystagmus, I did not consider monovision to be a viable option for this patient. He already struggled with his null point, and I find that patients like this one generally have better binocular than monocular vision. We therefore decided to proceed with lens-based surgery with a target good of UDVA and UNVA using multifocal IOLs. The process of informed consent was extensive, and the patient understood that, if neural adaptation did not occur, I would exchange the lens implants for monofocal IOLs targeted for best corrected distance visual acuity in each eye.
I implanted an AcrySof IQ Restor +2.5 D IOL with Activefocus optical design (Alcon) in the patient’s right eye and an AcrySof IQ Restor +2.5 D Multifocal Toric IOL with Activefocus optical design (Alcon) in the patient’s left eye. I performed arcuate incisions to reduce the small amount of cylinder present in the right eye. The patient and I chose a multifocal IOL design that prioritizes superior night vision over reading fine print because driving at night was very important to him and he did not mind wearing glasses to read print finer than J3. Additionally, I wanted to use the same IOL technology in both eyes, with a toric model in one eye.
The nystagmus made accurately measuring and treating the steep axis of astigmatism with lensectomies challenging. Small amounts of residual astigmatism can have a significant effect on neural adaptation and quality of vision with multifocal IOLs. Iris registration technology was critical to my ability to mark the steep axis accurately. Using the Cassini (Cassini Technologies), my technician performed topography with the patient’s head positioned as close to his null point as possible. Using those images with the Lensar Laser System (Lensar), I made arcuate incisions in the right eye and refractive capsulorhexis marks in the left eye that were exactly on axis with a successful iris registration maneuver. I also attempted to use the ORA System (Alcon) during cataract surgery. The patient, however, had trouble fixating under anesthesia, so I was glad to have marks at the lenticular plane for the toric IOL. Overall, there were no complications.
One week after surgery, UCVA was 20/25-1 and J2 OD, and a prescription of -0.50 D sphere improved visual acuity to 20/25+ and J2. UCVA was 20/25 and J2 OS with a plano prescription. The patient was pleased with his result, as was I, and he reported good reading vision and no problems driving at night.
Subsequent development of early posterior capsular opacification reduced UCVA to approximately 20/40 and J5. The patient underwent Nd:YAG laser capsulotomy in each eye approximately 120 days after surgery, and UCVA returned to the earlier level in each eye.




