Update on Epi-On CXL
Recent studies have evaluated the role of oxygen in enhancing the results of epi-on CXL.
By Kenneth Beckman, MD, FACS
Epithelium-off (epi-off) CXL has been shown to be an effective treatment for progressive keratoconus and ectasia after refractive surgery. The Dresden protocol, an epi-off technique first introduced in 2003 by Theo Seiler, MD,1 is currently followed in the United States in conjunction with the only FDA-approved CXL suite, the KXL System, and Photrexa drug formulations (both by Avedro).
Although the efficacy of the Dresden epi-off protocol has been shown, the required removal of the epithelium can cause discomfort, raise the risk of infection, and delay visual recovery in patients who undergo treatment. Additionally, current FDA labeling for epi-off CXL requires an intraoperative corneal pachymetry reading of 400 µm or greater, potentially excluding eyes with thinner corneas from receiving treatment if adequate stromal thickening cannot be obtained with hypotonic solution.
There has long been a high level of interest in the development of transepithelial, or epithelium-on (epi-on), protocols. A challenge has been that an intact epithelium acts as a natural barrier that limits the penetration of the UV light and riboflavin needed for effective CXL. Several published studies from outside the United States have concluded that current epi-on CXL treatment approaches are not as effective at halting progression as epi-off treatment, in part due to this barrier effect.2,3 Recently published results of a US study involving a large number of eyes treated with an epi-on protocol were promising, but there were weaknesses in the study design, including the lack of a control group and a large percentage of cases lost to follow-up.4 Without documented ectatic progression at baseline and a control group, no conclusions can be drawn about whether the procedure stabilized the ectasia or the ectasia was already stable. This study also evaluated technology that is not currently FDA approved, cannot legally be marketed in the United States, and would not be covered by insurance, which significantly impedes patient access.
Therefore, to date, no epi-on protocol has been approved by the FDA.
SUPPLEMENTAL OXYGEN
The light-chemical reaction that occurs in CXL naturally depletes the corneal oxygen required to induce the free radical formation that stiffens tissue. As oxygen is depleted, anaerobic reactions begin to dominate, and CXL efficiency decreases. It has been theorized that delivering oxygen to the cornea during the procedure could result in increased biomechanical change, especially in epi-on procedures.
Investigators are now looking more closely at what happens to oxygen in the stroma during CXL. Recent study results indicate that midstromal oxygen decreases to zero under normoxic conditions (room air) during both epi-on and epi-off CXL. However, when supplemental oxygen was provided at the anterior surface during UV illumination in high-irradiance epi-on CXL, midstromal supply and demand were balanced (Figure 1).5
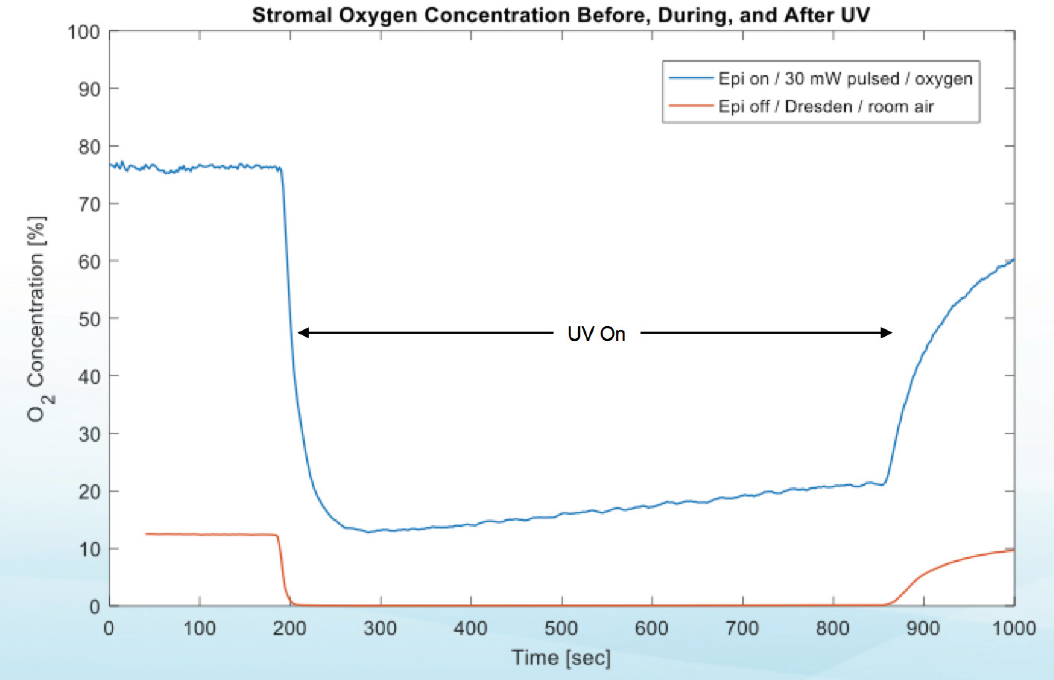
Figure 1. Stromal oxygen concentrations before, during, and after UV light application in two different CXL protocols demonstrate that the supply of and demand for oxygen are balanced when supplemental oxygen is provided.
In a porcine eye study, scientists evaluated an epi-on protocol that included high-irradiance pulsed light, a riboflavin formulation containing benzalkonium chloride, and supplemental oxygen.6 Treatment using this protocol resulted in significantly more corneal stiffening (P = .013) than alternative epi-on protocols with no supplemental oxygen.
Supplemental oxygen application with a new CXL system and epi-on protocol is currently being studied in patients with progressive keratoconus. Enrollment of 275 individuals across 14 sites is complete. This randomized, controlled phase 3 clinical trial is being conducted by Avedro to support an application for FDA approval of a drug-device combination product for epi-on CXL.
In the investigational procedure, an epi-on drug formulation is used, and the UV portion of the procedure is performed while patients wear oxygen goggles that create an oxygen-rich environment at the corneal surface (Figure 2). Two-thirds of patients in the trial receive the investigational epi-on CXL treatment, and the other one-third receive a sham treatment. After 6 months of follow-up, the sham control group is eligible to receive treatment; rescue treatment is also available during the initial 6 months, if needed.
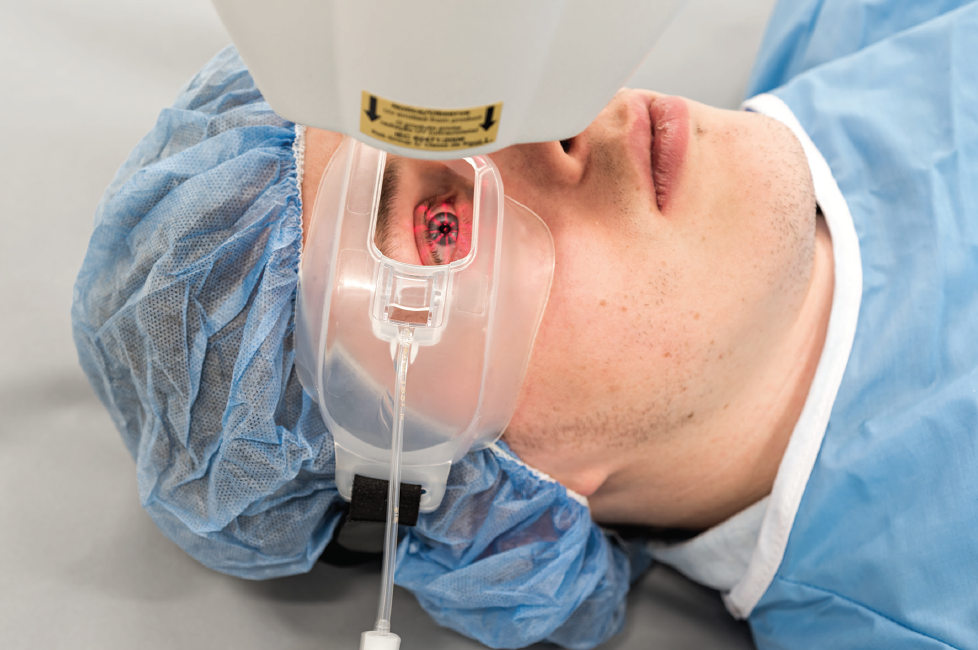
Figure 2. Goggles deliver supplemental oxygen to the corneal surface during CXL.
CONCLUSION
Theoretical evidence supports a role for supplemental oxygen in CXL, and investigations of the clinical outcomes of this procedure are under way. Explorations of this and other protocols may eventually lead to the development of an effective epi-on treatment to offer to patients with progressive keratoconus.
1. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-a-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135(5):620-627.
2. Rush SW, Rush RB. Epithelium-off versus transepithelial corneal collagen crosslinking for progressive corneal ectasia: a randomised and controlled trial. Br J Ophthalmol. 2017;101(4):503-508.
3. Kobashi H, Rong SS, Ciolino JB. Transepithelial versus epithelium-off corneal crosslinking for corneal ectasia. J Cataract Refract Surg. 2018;44(12):1507-1516.
4. Stulting RD, Trattler WB, Woolfson JM, Rubinfeld RS. Corneal crosslinking without epithelial removal. J Cataract Refract Surg. 2018;44(11):1363-1370.
5. Hill J, Liu C, Deardorff P, et al. Stromal oxygen dynamics during high-irradiance epi-on corneal crosslinking. Paper presented at: The Association for Research in Vision and Ophthalmology Annual Meeting; May 3-7, 2019; Baltimore.
6. Hill J, Liu C, Deardorff P, et al. Biomechanical impact of drug formulation, supplemental oxygen, and UV delivery on epi-on CXL. Poster presented at: The Association for Research in Vision and Ophthalmology Annual Meeting; May 3-7, 2019; Baltimore.
CXL Combined With Topography-Guided PRK
Moving beyond spectacle independence.
By Eric D. Donnenfeld, MD
The excimer laser was invented in the 1970s. Stephen Trokel, MD, introduced PRK in 1987, and the FDA approved the procedure in 1995. CXL was introduced internationally in 2003, and it received FDA approval in 2016. In recent years, surgeons have been exploring how the two technologies and procedures can be combined to benefit patients, and that work points to a coming shift in keratoconus management.
SIMULTANEOUS OR SEQUENTIAL CXL AND TOPOGRAPHY-GUIDED SURFACE ABLATION
Debate continues over whether better refractive results are achieved with a simultaneous or a sequential approach to combining CXL with topography-guided PRK (TG-PRK). My colleagues and I favor a sequential approach, and we recently published the largest US case series showing the safety and efficacy of TG-PRK in keratoconus patients after CXL. Our goal was to evaluate visual and astigmatic outcomes after the two procedures, and we suggested a treatment algorithm based on our observations and results.1
Our study included 56 patients (62 eyes), 34 of whom received both topographic and refractive treatment and 28 of whom underwent treatment of topographic irregularities only. Six months after TG-PRK, we observed a significant improvement in both uncorrected and corrected distance visual acuity in the refractive group compared to the nonrefractive group. No patient in the study experienced a significant adverse event.
Watch it Now

Dr. Donnenfeld speaks with Neda Shamie, MD, about visual and astigmatic outcomes after CXL and TG-PRK.
FUNDAMENTALLY CHANGING KERATOCONUS MANAGEMENT
Research and experience with combined CXL and TG-PRK, both ours and others’,2,3 suggest that it may become possible not just to reduce the number of corneal transplantation procedures performed but also to conquer keratoconus. No longer will surgeons have to observe patients until they require a corneal transplant. Instead, ophthalmologists will be able to use CXL technology and a topography-guided excimer laser, respectively, to halt the progressive ectasia of keratoconus and to smooth irregularities in corneal curvature.
In so doing, surgeons will augment patients’ quality of life by improving their BCVA with glasses and, in certain cases, allowing them to wear contact lenses more comfortably. After TG-PRK, some will be able to transition from rigid gas permeable or scleral contact lenses to soft contact lenses.
SYNERGIES
TG-PRK after CXL is a beautiful example of how technologies can be combined synergistically to improve patients’ quality of life. As these particular technologies improve and ophthalmologists’ experience with pairing the procedures grows, I expect results to get better.
I also anticipate further melding of refractive and therapeutic modalities. Research on protocols for and applications of CXL continues. The ACP-KXL-308 Study is a phase 3 multicenter study evaluating the safety and efficacy of epi-on CXL in eyes with progressive keratoconus. In it, investigators from 14 centers are combining Avedro’s UV light source and new drug formulation that is designed to penetrate the epithelial layer of the cornea with supplemental oxygen. This procedure has the potential to enhance the effects of CXL and to reduce recovery time and patients’ discomfort postoperatively. This platform to strengthen, stabilize, and reshape the cornea may become a means of treating patients with corneal ectatic disorders and correcting refractive conditions.
1. Nattis A, Donnenfeld ED, Rosenberg E, Perry HD. Visual and keratometric outcomes of keratoconus patients after sequential corneal crosslinking and topography-guided surface ablation: early United States experience. J Cataract Refract Surg. 2018;44(8):1003-1011.
2. Krueger RR, Kanellopoulos AJ. Stability of simultaneous topography-guided photorefractive keratectomy and riboflavin/UVA cross-linking for progressive keratoconus: case reports. J Refract Surg. 2010;26(10):S827-832.
3. Kanellopoulos AJ, Binder PS. Collagen cross-linking (CCL) with sequential topography-guided PRK: a temporizing alternative for keratoconus to penetrating keratoplasty. Cornea. 2007;26(7):891-895.






