CASE PRESENTATION
A 75-year-old man presents with a desire for cataract surgery on his right eye. At age 15, he sustained a bungee cord injury that led to a progressive loss of vision in his left eye, and he says he was previously told that surgery on this eye would be too risky (Figure). Now, this functionally monocular patient has failed a driving test because of diminished vision from cataract formation in his right eye.
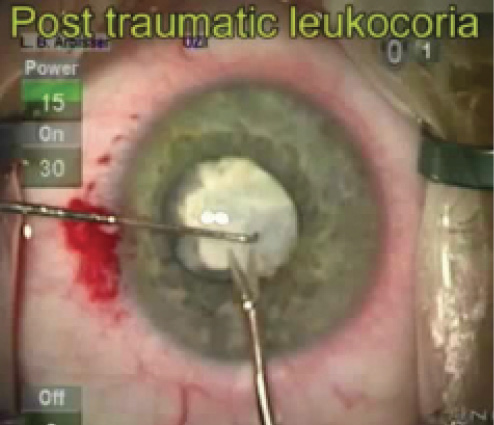
Figure. As a teenager, this now elderly patient’s left eye was injured by a bungee cord.
Upon examination, visual acuity measured 20/60 in his right eye and hand motion in his left with accurate light projection in all quadrants. IOP is symmetric between the two eyes (19 mm Hg). The patient’s right eye has a normal appearance, aside from a 3+ nuclear sclerotic cataract. His left eye exhibits mild anisocoria associated with a small defect in the iris sphincter, and a 0.3 log unit relative afferent pupillary defect (APD) is evident. The patient’s left eye has leukocoria associated with a dense, white, membranous anterior capsule. A poor view of the retina seems to show red reflex in all quadrants of his left eye, but the cup-to-disc ratio is difficult to determine. The patient also demonstrates a small sensory exotropia of his left eye. How would you rehabilitate this patient?
—Case prepared by Lisa Brothers Arbisser, MD

JAMES FREEMAN, MD
The most rapid, safe, and effective way to rehabilitate this patient would be to perform routine cataract surgery on his right eye. He has adapted well to monocular vision, and this procedure is likely to return him to his baseline.
If the patient desires rehabilitation of his left eye after removal of the cataract in his right, the first step would be preoperative counseling regarding the uncertain prognosis and the likelihood that, even with an excellent surgical outcome, the visual acuity of his left eye will never be as good as that of his right. Preoperative testing should include ultrasonography and an attempted assessment of macular function via color discrimination or entoptic phenomena. If available, flash electroretinography might also be useful.
Cataract surgery on the patient’s left eye would involve dissection of any posterior synechiae, followed by careful, sharp dissection of the pupillary membrane. A two-handed approach with forceps and intraocular scissors would likely be required to remove the tissue surrounding the visual axis.
Eyes with a history of injuries like the one in this case are at particularly high risk of iatrogenic retinal tears. A careful but thorough anterior vitrectomy is necessary, and the surgeon should be careful to avoid engaging or pulling on any vitreous strands. Once the visual axis was open and the surgeon was confident that no vitreous was present in the anterior chamber, there would likely be room for an IOL in the sulcus.

ANITA NEVYAS-WALLACE, MD
I would operate on the patient’s left eye first. Its visual potential is decent, given the mildness of the relative APD and the presence in all quadrants of good light projection and a red reflex.
A blunt injury significant enough to damage the anterior lens capsule probably injured the zonule as well, even if phacodonesis was not obvious on slit-lamp examination. The combination of a fibrotic capsule and damaged zonule presents its own challenges. First, if trypan blue dye travels through a zonular defect, it will stain the outside of the posterior capsule and interfere with the red reflex. To prevent this problem, I would instill a high-viscosity cohesive OVD over the peripheral bag/zonule and then inject a shallow layer of balanced salt solution over the central 7 to 8 mm of the anterior capsule so that I could paint only that area with dye.
Second, a poor zonule might be further damaged by the blunt trauma of attempting to puncture and tear the anterior capsule, especially if it is fibrotic. I would not use a femtosecond laser to create the capsulotomy for several reasons: the likelihood of an incomplete cut, the constricted pupil, and the risk of radial capsular tears, particularly if iris or capsular hooks are needed to support the bag.
My first choice would be to use the »Zepto Capsulotomy System (Mynosys Cellular Devices). My second would be a manual approach; after placing iris hooks, I would use the crossed-swords technique described by Michael E. Snyder, MD, in which two 30-gauge needles perforate the anterior capsule simultaneously from opposite directions to counterbalance the traction exerted. I would then use scissors to cut a capsulotomy and enlarge the pupil with iris hooks before using either the Zepto or a manual approach.
Regardless of the approach chosen, I would size the capsulotomy to permit posterior optic capture of a sulcus-fixated IOL. In posterior optic capture, the haptic-optic junctions of a three-piece IOL stent open the capsulotomy, thereby helping to prevent progressive phimosis of the capsulotomy, which exerts traction on zonular fibers and may lead to dislocation of the lens-bag complex. Next, I would aspirate loose cortex to aid visualization and to create space in the bag for iris or capsular hooks, hydrodissection, and viscodissection.
A cataract more than 6 decades old is apt to be quite dense. I would use the »miLoop (Iantech), and I would ignore the small iris defect.
Placing a capsular tension ring (CTR) would not necessarily prevent phimosis, but it could facilitate suturing a dislocated lens-bag complex to the sclera in the future. To insert the CTR without further stressing the zonule, I would corral the device’s leading loop with an instrument such as a Fenzl hook, with its trigger pointed skyward, or a nylon suture so that no part of the CTR touched the bag until most of the ring was within the bag.
I would implant either the »LI61AO (Bausch + Lomb) or the CT Lucia 602 (Carl Zeiss Meditec). The LI61AO emerges planar from its cartridge and is relatively forgiving of decentration, but silicone oil—if required one day—will stick to a silicone IOL more tenaciously than to other materials. For that reason, my preference would be the CT Lucia 602, which is made of heparin-coated hydrophobic acrylic and features sturdy, thin PVDF haptics. This IOL is suitable for placement in the sulcus with posterior optic capture or for intrascleral fixation such as with the Yamane flanged haptic technique.

SATHISH SRINIVASAN, MBBS, FRCS(Ed), FRCOphth, FACS
The anisocoria in the patient’s left eye may have been caused by iris sphincter injury, or it may result from the mild APD. I would be inclined to operate on the patient’s left eye first after a detailed conversation with him regarding the risks and potential benefits.
The iris sphincter tear indicates potential trauma or damage to the zonules as well. The Figure shows a dense leukocoria and a densely fibrotic anterior capsular plaque. Prior to surgery, I would hold a detailed discussion with the scrub nurse regarding the challenges posed by this case. I would also ensure that the OR was equipped with all the tools I might need, including high-viscosity OVD, trypan blue dye, CTRs, »Ahmed Capsular Tension Segments (Morcher, US distributor FCI Ophthalmics), 23-gauge anterior segment instrumentation (micrograspers, scissors, and forceps), a three-piece backup IOL, and a setup for anterior vitrectomy.
Given the potential complexity of the case, retrobulbar anesthesia would be my preference. After making two paracentesis incisions 180º apart, I would stain the anterior capsule with trypan blue dye. A commercial preparation of trypan blue recently became available in Europe that has a very high concentration of 0.4% in 0.15 mL (Capsulblue, Excel-Lens; not available in the United States) compared to the traditional concentration of 0.06%. I find that the former produces better staining in cases of dense anterior capsular plaque.
Next, I would fill the anterior chamber with high-viscosity OVD. I would attempt to initiate a continuous curvilinear capsulorhexis (CCC) with a cystotome placed through the paracentesis. In eyes with dense anterior capsular plaque, however, I often find it necessary to use 23-gauge scissors to make the initial cut through the anterior capsular plaque so as to raise an edge. I would then use 23-gauge CCC forceps through the paracentesis to cautiously complete a smaller CCC and constantly replenish the OVD to ensure adequate tamponade. I would wait to construct the main corneal incision until I had completed the CCC.
I would perform gentle hydrodissection in multiple quadrants and then assess the mobility of the nucleus, which could be dense or a liquefied cortex. Depending on my findings, I would prolapse the nucleus into the anterior chamber and perform supracapsular phacoemulsification if zonular integrity were in doubt, or I would simply aspirate the liquefied nucleus and cortex. If the bag and zonules were intact, I would place a one-piece acrylic IOL in the bag. If I saw signs of zonular dehiscence, then I would place a three-piece acrylic IOL in the sulcus with optic capture.

WHAT I DID: LISA BROTHERS ARBISSER, MD
The patient and I decided to delay surgery on his right eye until after an attempt at maximizing the visual acuity of his injured left eye. I emphasized the possibility of complicated surgery, the potential need for more than one procedure, and the chance of bothersome postoperative diplopia that would require intervention. The prognosis for 20/20 visual acuity was guarded, and I decided to implant a monofocal IOL because there was no significant astigmatism. The patient was comfortable with the safety glasses he had been wearing for years.
Twenty minutes before surgery, he received mannitol 0.25 g/kg as a bolus intravenous push. Because the patient could not fixate on the muscle light in the lane easily, I opted for peribulbar anesthesia. I injected »Healon GV (Johnson & Johnson Vision) under a soft shell of a dispersive OVD to avoid a convex lens in case of high intralenticular pressure. I easily removed a fine pupillary membrane and then used trypan blue to stain the capsule under the viscous cohesive OVD to improve visualization.
With a cystotome, I opened the anterior capsule. Despite my care with the forceps, schisis of the fragile and scarred capsule made it impossible for me to create a true CCC. With repeated efforts from various angles, I achieved the semblance of a round opening. A thick, leathery, white plaque—seemingly fused anterior cortex and capsule—remained, and I dissected and sectioned it with intraocular forceps and scissors in order to explant the material though the unenlarged clear corneal incision. I subsequently performed minimal, delicate, multidirectional hydrodissection but made no attempt to rotate the nucleus because of the unknown state of zonules and the discontinuous capsulorhexis.
To prevent a wraparound tear, from this point forward I was cautious not to overly deepen the anterior chamber or allow it to become shallow. Because the eye was myopic, before initiating irrigation I inserted the phaco tip into an OVD-controlled anterior chamber while on foot position zero. With my second hand, I used a Rosen splitter instrument through the sideport incision to lift the iris off the anterior capsule. Only then did I engage irrigation. In my experience, this technique prevents reverse pupillary block, which can cause a sudden deepening of the chamber.
Using an in situ vertical chop technique, I liberated the first quadrant, thus mobilizing the rest of the dense nucleus and allowing me to complete phacoemulsification. I stabilized the chamber by injecting an OVD through the sideport incision before removing the phaco handpiece. To avoid turbulence and the risk of extending any anterior capsular tear, I performed dry cortical removal using a 26-gauge cannula on a 5-mL syringe under control of the chamber and lens fornix with a less-viscous cohesive OVD (ProVisc, Alcon).
WATCH IT NOW
Inspection revealed enough remaining anterior capsule to cover and control the haptics of a one-piece hydrophobic acrylic IOL, which I placed atraumatically. I performed careful manual removal of the OVD with irrigation through the incision to ensure a tight seal and burped the OVD by pressing on the posterior lip. Next, I brought down the pupil with acetylcholine (Miochol-E, Bausch + Lomb) to increase IOL support. In addition to my routine off-label intracameral injection of moxifloxacin, I administered a small amount of triamcinolone acetonide (Triesence, Alcon) to confirm that no vitreous had surreptitiously presented and to increase antiinflammatory protection. Upon discharge, the patient received oral acetazolamide to avert an IOP spike associated with any retained OVD.
One day after surgery, the patient’s UCVA was 20/25. Visual field testing showed a small peripheral area of depression corresponding to a pigmented area, probably due to commotio retinae from the injury causing the relative APD. A scleral indented examination showed no tears or detachment. The patient recovered uneventfully with only transient diplopia and proceeded with routine cataract surgery on his fellow eye.




