CASE PRESENTATION
An intellectually disabled 68-year-old woman in group care has become unable to perform to her usual ability. She can no longer see well enough to be safely cared for in this setting.
An examination reveals central steady and maintained fixation in both eyes, no relative afferent pupillary defect, IOP symmetric to finger tension, and densely brunescent bilateral cataracts with a poor view of the retina consistent with the media. Retinoscopy shows a myopic refraction, although the patient has never worn glasses. Glimpses of the optic nerve show a cup-to-disc ratio of 0.3 and no mass or hemorrhage in either eye. The patient’s inability to hold still makes it impossible to obtain accurate measurements for IOL power calculation in the office. She is scheduled for cataract surgery with examination under anesthesia.
How would you proceed? What would your surgical plan be? What would you do differently for this patient compared to one who is able to cooperate?
—Case prepared by Lisa Brothers Arbisser, MD.

H. BURKHARD DICK, MD, PhD
I would examine the patient under general anesthesia and, based on the results, would then perform simultaneous bilateral cataract surgery using a laryngeal mask airway during anesthesia.
For accuracy and ease, I would use a handheld autokeratorefractometer (Retinomax; Lombart Instrument).1,2 I would measure axial length via A-scan ultrasonography. If the results were uncertain, I would perform intraoperative aberrometry using the ORA System (Alcon), which I find works well under general anesthesia.
I would target mild myopia of approximately -1.75 D in one eye and -2.50 D in the other eye with monofocal IOL implantation. If the patient’s personal circumstances allowed, a multifocal IOL would be a perfectly reasonable option (according to the medical history, it seems unlikely she will drive a car).
Bilateral simultaneous surgery actually means two entirely separate interventions: two separate operating tables; hand disinfection and a change of surgical scrubs after the first operation; and the use of ophthalmic viscosurgical devices (OVDs), fluid, etc. from different lot numbers. Only if the surgery on the first eye went without any meaningful complication would I proceed with the contralateral eye.
I would create the main incision at 12 o’clock, because a temporally located incision might be compromised if the patient rubbed her eyes postoperatively. I would perform a primary posterior capsulorhexis to render posterior capsular opacification as unlikely as possible, which should be helpful in this case. An alternative would be to perform this step—once again depending on the patient’s situation—with a femtosecond laser after IOL implantation in an eye that, from a surgical standpoint, is already closed.3,4 Finally, I would instill cefuroxime intracamerally and inject a steroid-antibiotic depot mix subconjunctivally. Watch glass bandages would protect both eyes.

P. DEE G. STEPHENSON, MD
In intellectually disabled patients, the workup and care can be somewhat challenging.
This patient’s old records would be helpful, but her gross eye exam is essentially normal. I wish there were more options for preoperative A-scans or that intraoperative aberrometry could be used. Unfortunately, a patient’s cooperation is necessary, although the ORA System with VerifEye+ (Alcon) could be tried even with her under general anesthesia. My plan would therefore be straightforward, with general anesthesia and bilateral microincisional cataract surgery. I would use average keratometry readings and average axial length measurements and would use the IOLMaster (Carl Zeiss Meditec) to pick the best monofocal IOL possible. I would target mild monovision.
I would create a small self-sealing incision and implant either the Akreos AO Micro Incision or the EnVista lens (both from Bausch + Lomb), which pass through a 1.8- and a 2.2-mm incision, respectively. This approach would eliminate the need for a suture, which could be impossible to remove in the future. I would perform a primary posterior capsulorhexis and posterior optic capture. The EnVista would be a great lens for posterior optic capture to eliminate the need for a future YAG capsulotomy. I would conduct a thorough examination of each eye while the patient was under anesthesia, inclusive of a thorough retinal exam and IOP measurement.
An intracameral antibiotic-steroid combination would be an excellent choice for this patient, as it might eliminate the need for postoperative drops. Alternatively, this patient might be able to use a topical nonsteroidal anti-inflammatory drug once daily in her eyes after surgery for approximately 4 weeks.

WHAT I DID: LISA BROTHERS ARBISSER, MD
After a consultation with the patient’s caregivers and power of attorney, I recommended bilateral sequential cataract surgery under general anesthesia with bilateral primary posterior capsulotomies and posterior optic capture into Berger’s space. This is the only solution that will allow one anesthesia for lifelong vision. My plan was for minimonovision with a refractive target of plano OD and -1.50 D OS, assuming the patient’s axial length showed previous emmetropia versus axial myopia, but near and intermediate vision if I found intraoperatively that she was previously a lifelong myope. I gave the caregivers samples of artificial tears just to accustom the patient to the instillation of eye drops for a few days before beginning the usual preoperative and subsequent postoperative topical regimens.
According to the dictates of the International Society of Bilateral Cataract Surgeons, care was taken to ensure no duplication of lot numbers for all intracameral compounds, and there were two separate back tables and a separate prep and drape for each eye. The plan was to postpone surgery on the second eye if any complications occurred during the cataract procedure on the first eye.
After the induction of intubation and general anesthesia, keratometry readings and A-scan ultrasonography were obtained, and three-piece acrylic IOLs of the proper powers were selected. Using a Perkins tonometer, IOP measured 10 mm Hg OD and 11 mm Hg (typically low under general anesthesia).
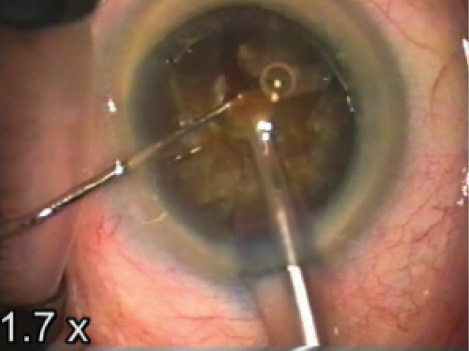
Figure. Dr. Arbisser debulks dense endonucleus during circumferential disassembly and vertical cross-action chopping.
The patient was prepped and draped in the usual manner. After instilling trypan blue under a soft shell of a dispersive and a cohesive OVD, I created a 5-mm anterior capsulorhexis. Using my circumferential cross-action vertical chop technique,5 I completed nuclear removal with under 40 seconds of cumulative dispersed energy and good endothelial protection (Figure). I performed cortical cleanup and polished the capsule.
Because of the flaccid capsule associated with the advanced cataractous change, I inserted a capsular tension ring. I measured the anterior capsulorhexis to ensure that it would allow optic capture if necessary. Placing an OVD in the sulcus to collapse the anterior and posterior capsules together, under high magnification, I incised the posterior capsule with a bevel-up 30-gauge needle. I instilled a cohesive OVD behind the capsule to define Berger’s space delimited by Wieger’s ligament and to push back the intact hyaloid face. I performed a continuous curvilinear posterior capsulorhexis of 4 to 5 mm. Next, I opened the cul-de-sac of the bag with an OVD and inserted the IOL into the capsular bag. I then captured the optic, with pressure on each optic pole 90º from the optic-haptic junction, through the posterior capsulorhexis into Berger’s space. I used automated coaxial irrigation and aspiration to remove the OVD from the anterior chamber only, while taking care not to render it shallow, which could have released the lens.
I performed an intracameral injection of moxifloxacin (off label and according to the precepts of the International Society of Bilateral Cataract Surgeons) and verified that the incisions were watertight. Next, I instilled intracameral triamcinolone acetonide (Triesence, diluted 1:10 with BSS [both products from Alcon]) to confirm that no vitreous had prolapsed (despite my certainty that the hyaloid face remained intact) and for its immediate anti-inflammatory effect.
WATCH IT NOW
Lisa Brothers Arbisser, MD, performs surgery on the patient’s first eye.
After completing the cataract procedure on the patient’s first eye without complication, Dr. Arbisser immediately performs cataract surgery on the second eye.
While the new scrub nurse gowned and gloved and prepared the entirely separate setup for surgery on the second eye, I performed indirect ophthalmoscopy and documented a normal fundus exam. The second eye was prepped and draped in the same manner, and the surgery and fundus exam were repeated for the second eye in the same fashion.
Because of the brunescent lens and the patient’s inability to communicate, at the conclusion of the case, intravenous acetazolamide (Diamox; Duramed Pharmaceuticals) 250 mg was given as prophylaxis against an IOP spike. The patient was bilaterally shielded only until she was fully awake, at which time the coverings were removed. As I find typical of such patients, she did not molest her eyes. I have never seen a wound dehiscence in this setting.
The patient’s caregivers reported that she had resumed her prior behaviors on the day of surgery, that she had reintegrated into her group environment, and that she was immediately easier to care for. The visual axes were clear on postoperative day 1, with deep and nearly clear chambers and globes that were normal to finger tension. Plus, the patient offered a big smile. The visual axes have been clear now for years, and I expect them to remain so for her lifetime.
1. Wesemann W, Dick B. Accuracy and accommodation capability of a handheld autorefractor. J Cataract Refract Surg. 2000;26(1):62-70.
2. Wesemann W, Dick B. Experiences with the hand-held autorefractometer “Retinomax” in adults and children [in German]. Klin Monbl Augenheilkd. 1997;211(6):387-394.
3. Dick HB, Schultz T. Primary posterior laser-assisted capsulotomy. J Refract Surg. 2014;30(2):128-133.
4. Haeussler-Sinangin Y, Schultz T, Holtmann E, Dick HB. Primary posterior capsulotomy in femtosecond laser-assisted cataract surgery: in vivo spectral-domain optical coherence tomography study. J Cataract Refract Surg. 2016;42(9):1339-1344.
5. Arbisser LB. Advanced phaco techniques for brunescent nuclei: cross-action chop circumferential disassembly. In: Chang DF, ed. Phaco Chop and Advanced Phaco Techniques. 2nd ed. Thorofare, NJ: Slack; 2013:221-228.




