
Spontaneous fibrin formation during Descemet membrane endothelial keratoplasty (DMEK; Figure 1) can prolong graft unscrolling times and contribute to increased endothelial cell loss.1 A valuable technique I learned from Dean P. Ouano, MD, is the injection of enoxaparin to facilitate graft unscrolling when fibrin is present. Additionally, we have developed a modified technique using intracameral enoxaparin. Both surgical methods are described here.
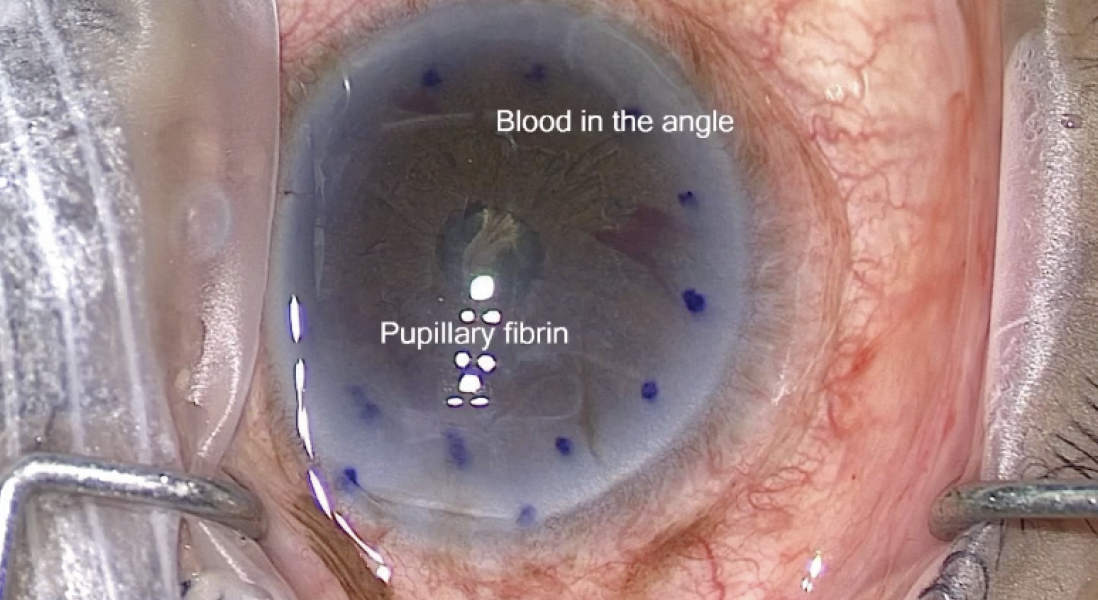
Figure 1. Blood is visible in the angle.
Courtesy of Dean P. Ouano, MD
The Challenge of Fibrin Formation
When intraoperative fibrin formation occurs during DMEK, surgeons have options: (1) continue tapping to unscroll the tissue, (2) mechanically lyse the fibrin with microscissors or a vitrector, or (3) inject a tissue plasminogen activator. Tissue plasminogen activators, however, have limitations such as a high risk of bleeding and a short half-life. Enoxaparin serves as a more practical alternative.2
Case example. A 67-year-old woman with a dislocated sulcus-fixated IOL, decompensated cornea, and iris atrophy was scheduled for DMEK. During surgery, the IOL was repositioned, and a limited vitrectomy was performed to address partially prolapsed vitreous. A single-pass, four-throw suture technique was then used to close the iris atrophy, resulting in iris bleeding.
I considered performing Descemet-stripping endothelial keratoplasty at this juncture, but I decided to irrigate and aspirate as much blood as possible and proceed as planned. The Descemet membrane was stripped, and the DMEK graft was inserted through a 2.5-mm clear corneal incision. The donor tissue was sufficiently pliable, enabling straightforward unscrolling on the right side (Figure 2A). I then typically pinch the graft on the right and use a tapping technique to unscroll it on the left. In this case, however, the graft failed to unscroll properly (Figure 2B).
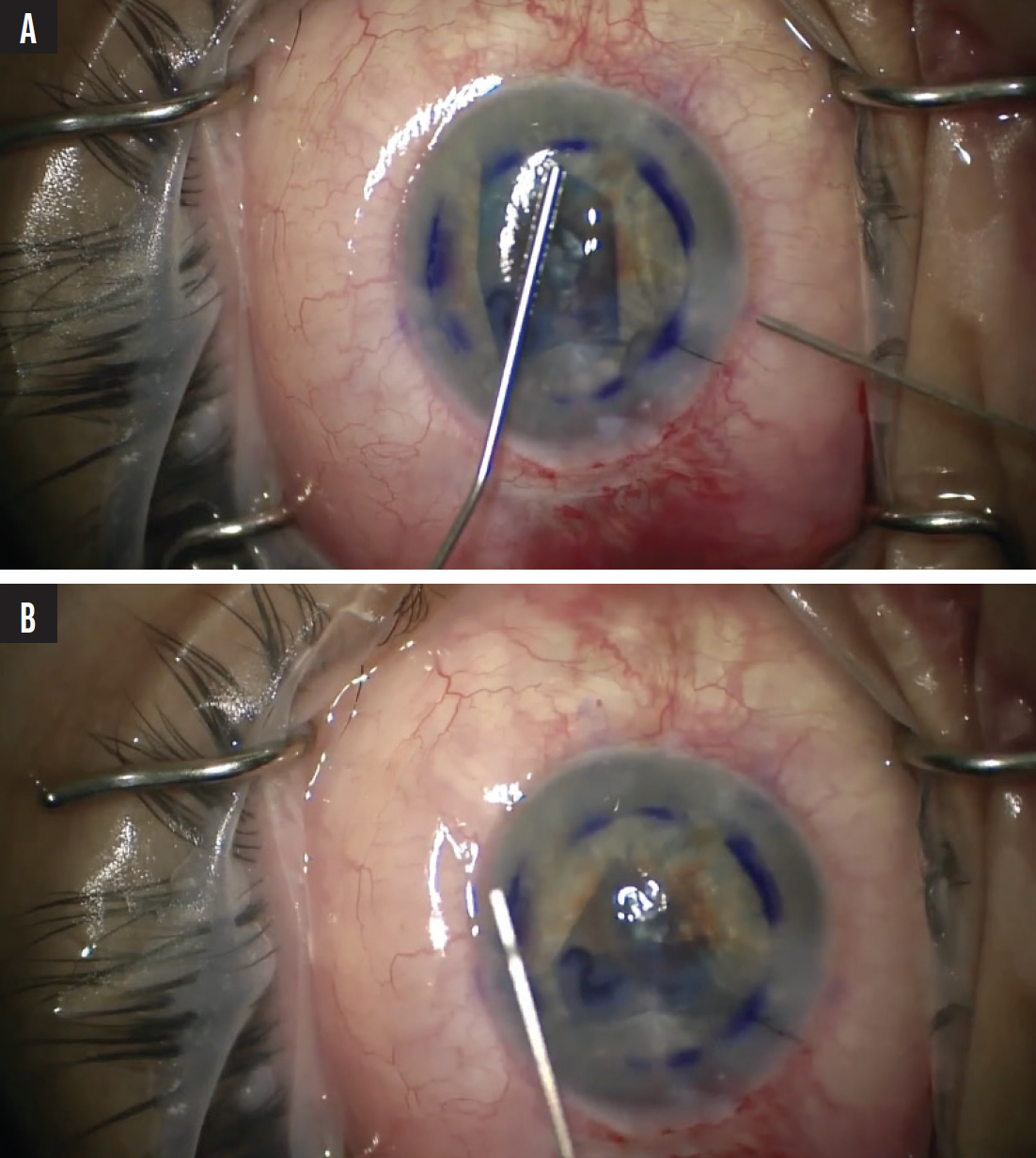
Figure 2. The donor graft cornea unscrolled properly to the right (A) but not to the left (B).
After several attempts to break the fibrin strand with a cannula—and my waning patience—I decided to deepen the anterior chamber. An anterior vitrectomy was performed with a low flow setting to detach the fibrin strand. After fibrin removal, the graft unscrolled perfectly (Figure 3). The patient’s postoperative visual acuity was 20/25.
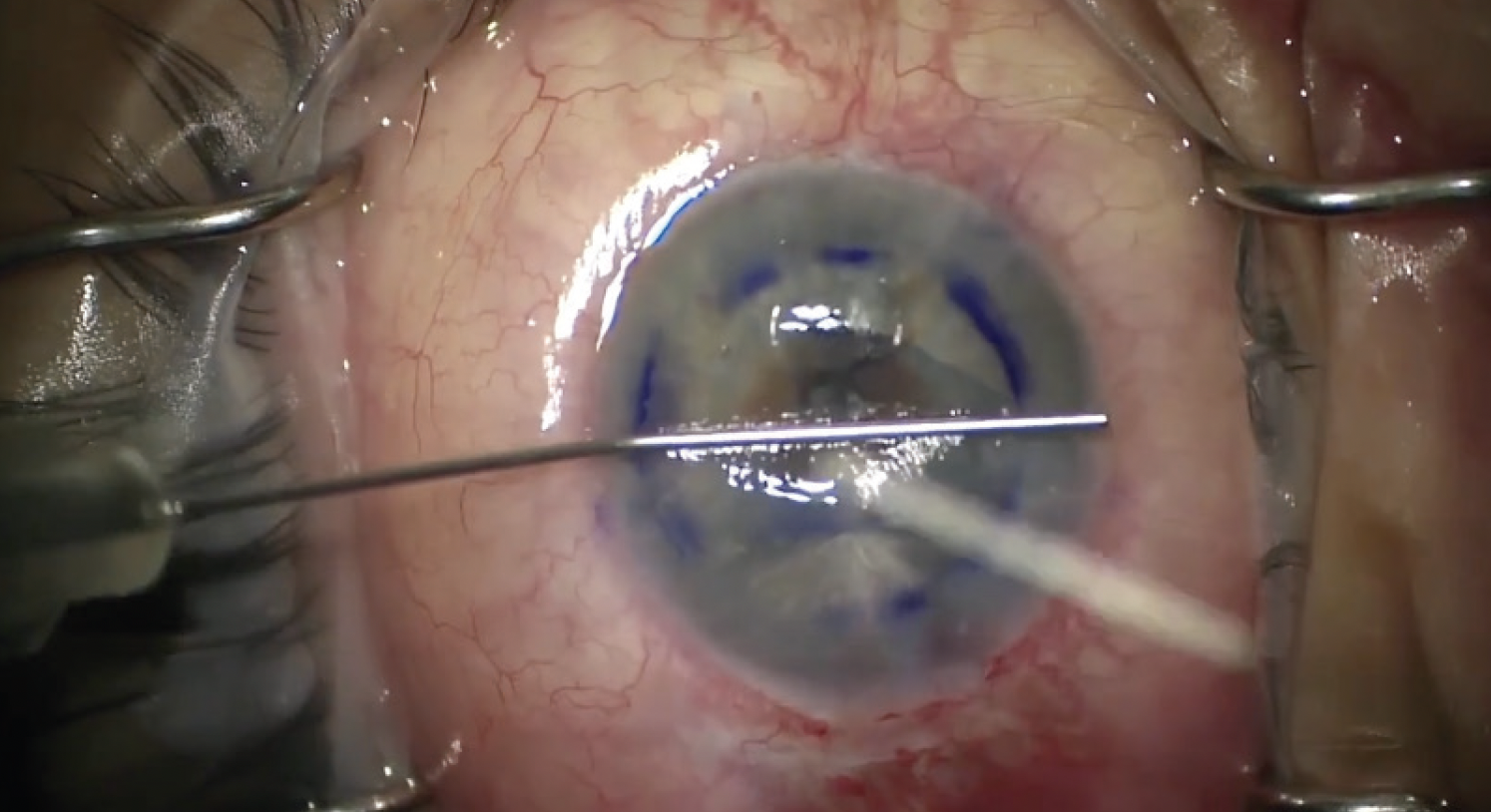
Figure 3. After the fibrin was removed, the graft unscrolled fully.
INTRACAMERAL ENOXAPARIN
Dr. Ouano and I have explored the application of enoxaparin as an additive to the irrigating solution, akin to the use of heparin during diabetic or uveitic cataract surgery, to mitigate fibrinoid reactions. Unlike heparin, enoxaparin is available at most pharmacies, shelf-stable, and packaged in a subcutaneous syringe for direct injection into the irrigating solution. Moreover, enoxaparin may offer a marginally greater antiinflammatory effect and potentially a lower risk of bleeding. It is both time-efficient and cost-effective, requiring a single 40-mg dose priced at approximately $16. The solution is introduced into 500 mL balanced salt solution irrigating fluid, which is then used during or after stripping the host Descemet membrane. In all cases where the technique was used, there were no instances of hemorrhage, bleeding, or fibrin formation.
Furthermore, enoxaparin imparts a negative charge to the graft, facilitating its self-repulsion and enabling problem-free unscrolling.
STUDY RESULTS
Six surgeons at six sites participated in a clinical study of the use of intracameral enoxaparin in DMEK. A total of 160 eyes were included, and the outcomes were compared to the results from eyes enrolled in well-known, multicenter DMEK studies and meta-analyses.3,4
The postoperative visual acuity outcomes for eyes in this study were comparable or slightly better. More than half (60%) achieved a visual acuity of 20/25, and 90% achieved 20/40 UCVA. Additionally, the complication rate was lower in our patient cohort. Most notably, there were no instances of hemorrhage in our study population. Additionally, in our laboratory analysis, exposure to high concentrations of enoxaparin showed no significant change in endothelial cell apoptosis or cell death rates when compared to control (Figure 4).
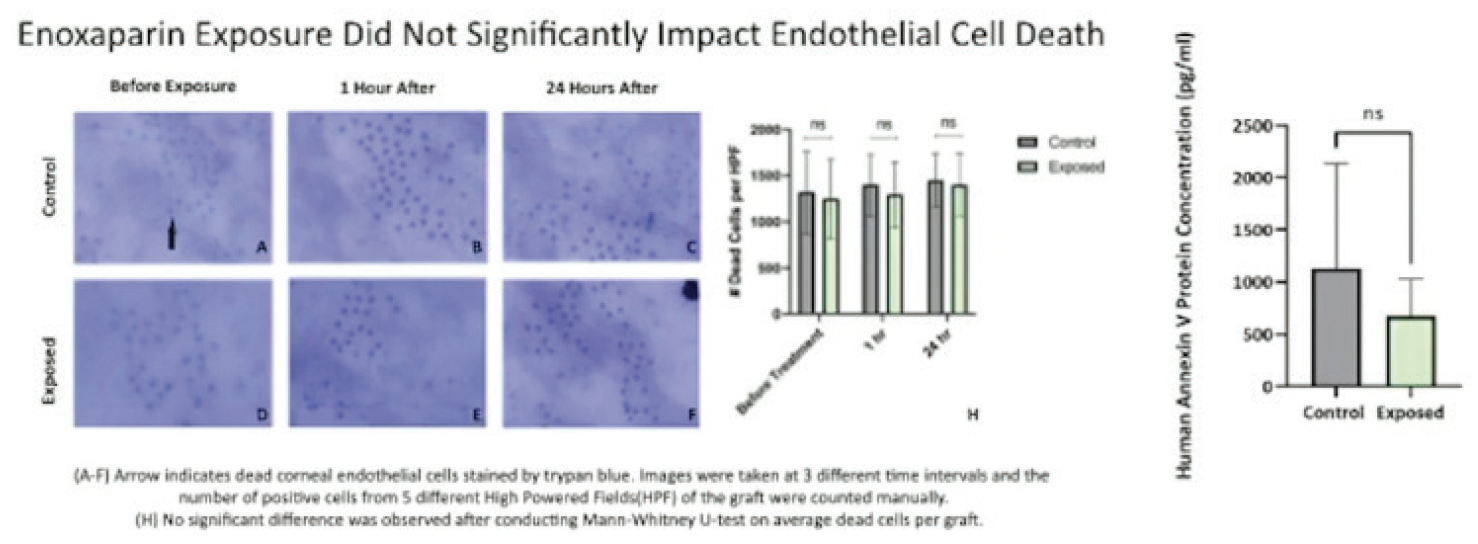
Figure 4. Enoxaparin exposure did not lead to endothelial cell death or apoptosis.
CONCLUSION
Intracameral enoxaparin represents a significant advance in the DMEK procedure. This is especially true for complex cases, including uveitic cataracts and eyes with synechiae. The simple addition of enoxaparin to the irrigating solution can facilitate the unscrolling of a DMEK graft. It can also be used for Descemet stripping endothelial keratoplasty with thicknesses below 50 µm.
1. Benage M, Korchak M, Boyce M, et al. Intraoperative fibrin formation during Descemet membrane endothelial keratoplasty. Am J Ophthalmol Case Rep. 2020;18:100686.
2. Ferguson TJ, Traboulsi EI, Goshe JM. Successful pediatric DMEK facilitated by intracameral tissue plasminogen activator to mitigate anterior chamber fibrin reaction. Am J Ophthalmol Case Rep. 2020; 19: 100812.
3. Deng SX, Lee WB, Hammersmith KM, et al. Descemet membrane endothelial keratoplasty: safety and outcomes: a report by the American Academy of Ophthalmology. Ophthalmology. 2018;125(2):295-310.
4. Spaniol K, Hellmich M, Borgardts K, et al. DMEK outcome after one year – results from a large multicenter study in Germany. Acta Ophthalmol. 2023;101(2):e215-e225.




