CASE PRESENTATION
A 57-year-old man undergoes uneventful cataract surgery on his right eye. Two weeks later, he undergoes cataract surgery on his left eye and is told immediately postoperatively that an incorrectly powered IOL was implanted. He returns to the OR the same day and undergoes an IOL exchange.
One day after surgery, his visual acuity is hand motions OS due to severe corneal edema. A regimen of topical difluprednate ophthalmic emulsion 0.05% (Durezol, Alcon), bromfenac, sodium chloride hypertonicity ophthalmic ointment 5% (Muro 128, Bausch + Lomb), and netarsudil ophthalmic solution 0.02% (Rhopressa, Alcon; off-label use) is initiated to restore the cornea.
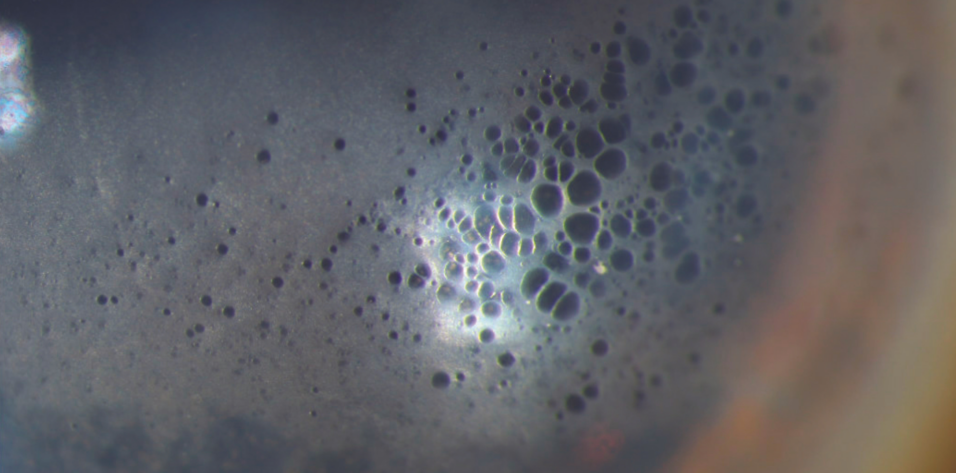
The vision in the patient’s left eye improves, but he notices a crescent-shaped image that corresponds to the temporal edge of the IOL optic. The cornea develops epithelial changes akin to those previously reported with netarsudil (Figure 1).1 Slight nasal decentration of the IOL optic becomes evident, but the lens appears to be well positioned overall.

Figure 1. Stromal and epithelial edema associated with endothelial recovery after surgery and characteristic epithelial changes associated with the use of netarsudil.
The patient has a history of laser treatment for retinal tears in both eyes. For most of his life, he has considered his left eye to be his better-seeing eye. He decides to seek a second opinion on his left eye.
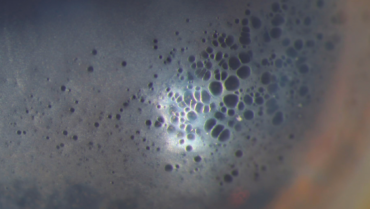
On presentation, the patient’s BCVA (plano OU) is 20/20 OD and 20/25+2 OS. He can see around the corneal edema in the left eye in a dark room. A dilated examination of the left eye confirms nasal decentration of the IOL. The temporal IOL loop is within the intact capsular bag. Visualization of the nasal loop is poor, but it appears to be in the sulcus (Figure 2).
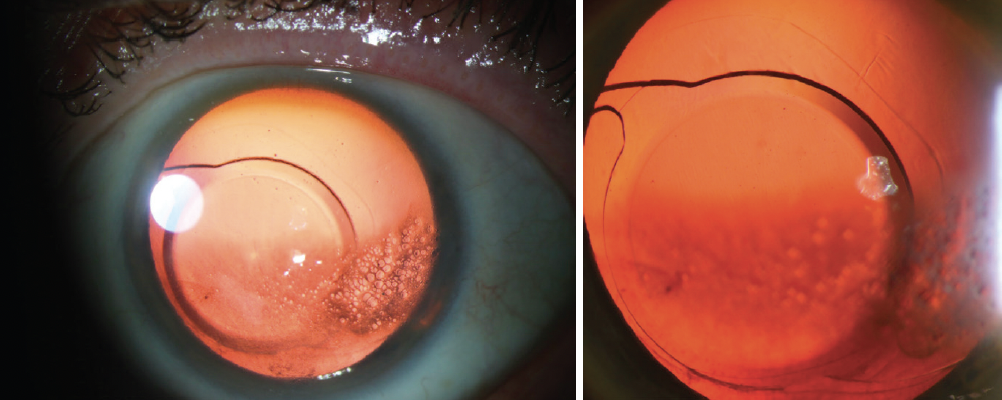
Figure 2. IOL decentration associated with a large capsulorhexis and asymmetric fixation. The temporal IOL loop is in the bag. The nasal loop appears to be in the sulcus.
Figures courtesy of Christiaan Lopez-Miro, Ophthalmic Photographer, Duke Eye Center
How would you rehabilitate the left cornea? How and when would you address the decentered IOL?
—Case prepared by Alan N. Carlson, MD

MINAS CORONEO, BSC(MED), MBBS, MSC, MD, MS, FRACS, FRANZCO, FARVO
A more specific management timeline, IOP measurements, and corneal pachymetry readings would assist with decision-making. Given the inferotemporal iris transillumination defect, it is possible that this haptic is also in the sulcus. My colleagues and I have reported the consequences of sulcus impingement by a one-piece acrylic IOL.2 If the time frame is short and only one haptic is out of the bag, I would consider dialing the haptic into the bag under topical anesthesia and antisepsis at the slit lamp. A bilateral endothelial cell count would be obtained, and peripheral counts would be interpreted as an indicator of the cornea’s capacity to recover.3,4
If the endothelial cell count is low or both haptics appear to be in the sulcus, repositioning at the slit lamp would not be attempted. In the OR, the IOL would be rotated under topical anesthesia and an OVD. A flat Drysdale blade would be passed under the anterior capsule and lifted in the region of the haptic to confirm its location. Resistance would indicate that the haptic is in the sulcus. In this situation, eyes are typically treated with an intensive course of topical and subconjunctival steroids. Experimental research, however, suggests that topical steroid use may inhibit endothelial wound healing,5 whereas NSAID therapy may facilitate the process.6 In a recent study, netarsudil-associated reticular corneal edema resolved after the drug was halted—consistent with most reported cases.7
I would consider discontinuing the steroids and hypertonic ointment while maintaining therapy with topical bromfenac and antibiotics. Topical hyperosmolar agents were recently shown to be ineffective for the treatment of early morning corneal edema associated with Fuchs endothelial dystrophy.8 Furthermore, experimental evidence suggests that, in activating cell volume regulatory mechanisms by anisosmotic challenge, the intervention may worsen the edema.9


RHEA NARANG AND AMAR AGARWAL, MS, FRCS, FRCOPHTH
Netarsudil-related keratopathy10-12 seems to have developed in the left eye postoperatively. The Rho kinase inhibitor can cause reticular corneal epithelial edema that resembles a honeycomb because the epithelial bullae are usually arranged in a reticular pattern. Rho kinase inhibitors are prescribed off-label to treat corneal edema, but studies have found that some eyes are predisposed to develop keratopathy as a result.10-12
We would immediately discontinue netarsudil therapy and wait for the cornea to clear. In the interim, therapy with topical difluprednate, bromfenac, and sodium chloride hypertonicity ophthalmic ointment would be maintained. After the corneal edema resolves, we would consider repositioning the IOL because leaving one haptic in the sulcus could incite an inflammatory response. A new paracentesis incision would be made. The IOL would be dialed into the capsular bag under an OVD, which would then be removed with irrigation and aspiration.
Netarsudil-related corneal epithelial changes are reversible,1 so we would expect the edema to resolve within a few weeks.

C. ELLIS WISELY, MD, MBA
The case presents two unusual postoperative complications of IOL exchange. I agree with Dr. Carlson’s assessment that the nasal haptic of the one-piece IOL is in the ciliary sulcus (Figure 2). I also agree with his assessment of reticular bullous corneal epithelial edema (Figure 1); the epithelial predilection of the small bullae is expertly demonstrated with the slit beam in Figure 2. This specific pattern of corneal edema is a side effect of netarsudil use,13 typically in patients who have corneal edema before starting the drug.1
One-piece IOLs tend to be poorly tolerated in the sulcus and may induce uveitis-glaucoma-hyphema syndrome and cystoid macular edema.14 The patient would therefore be taken back to the OR as soon as logistically feasible, and the haptic would be repositioned in the capsular bag. Prompt surgical intervention would also prevent scarring or adherence of the lens capsule to itself or the IOL, which could complicate repositioning of the haptic.
In my experience,1 reticular bullous epithelial edema resolves with no intervention apart from discontinuing the netarsudil. The time to resolution has ranged from 2 to 12 weeks. That said, I would take a more aggressive approach here. I would discontinue the netarsudil and perform a superficial keratectomy at the time of IOL repositioning and place a bandage contact lens for the first postoperative week. This would simplify lens repositioning by improving surgical visualization and would likely be therapeutic for the reticular bullous edema, which I have anecdotally found unlikely to recur after an eye heals from superficial keratectomy.

WHAT I DID: ALAN N. CARLSON, MD
Why the wrong IOL power was inserted is unknown. The IOL exchange was performed on the same day, suggesting the unexpected complication weighed heavily on the mind of the surgeon. When the patient sought a second opinion, asymmetric IOL fixation and substantial corneal edema were discovered.
The addition of netarsudil when the corneal edema did not resolve was a reasonable step. I have been pleasantly surprised by a few cases in which this form of treatment cleared the cornea and obviated the need for a keratoplasty procedure.
Ultrasound biomicroscopy can be helpful when visualization of the IOL fixation is inadequate. A direct view after adequate pupillary dilation, however, confirmed that the nasal IOL loop was in the sulcus. Prioritizing repositioning of the IOL into the intact capsular bag would offer the best opportunity for restoring the integrity and health of the eye. In cases like this, visualization and patient cooperation during surgery are essential to success. I would remove the epithelium only if necessary for visualization.
I appreciate that the panelists would not offer endothelial keratoplasty unless necessary. Several weeks of recovery may be required before the need for a corneal transplant can be determined.
The patient is currently postponing further intervention to allow the cornea time to clear.
1. Wisely CE, Liu KC, Gupta D, Carlson AN, Asrani SG, Kim T. Reticular bullous epithelial edema in corneas treated with netarsudil: a case series. Am J Ophthalmol. 2020;217:20-26.
2. Micheli T, Cheung LM, Sharma S, et al. Acute haptic-induced pigmentary glaucoma with an AcrySof intraocular lens. J Cataract Refract Surg. 2002;28(10):1869-1872.
3. Choi SO, Jeon HS, Hyon JY, et al. Recovery of corneal endothelial cells from periphery after injury. PLoS One. 2015;10(9):e0138076.
4. Kopecny LR, Lee BWH, Coroneo MT. A systematic review on the effects of ROCK inhibitors on proliferation and/or differentiation in human somatic stem cells: a hypothesis that ROCK inhibitors support corneal endothelial healing via acting on the limbal stem cell niche. Ocul Surf. 2023;27:16-29.
5. Singh G. Corticosteroids in corneal endothelial wound healing. Ann Ophthalmol. 1985;17(4):238-243.
6. Joyce NC, Meklir B, Neufeld AH. In vitro pharmacologic separation of corneal endothelial migration and spreading responses. Invest Ophthalmol Vis Sci. 1990;31(9):1816-1826.
7. Lindstrom RL, Lewis AE, Holland EJ, et al. Phase 2, randomized, open-label parallel-group study of two dosing regimens of netarsudil for the treatment of corneal edema due to Fuchs corneal dystrophy. J Ocul Pharmacol Ther. 2022;38(10):657-663.
8. Zander DB, Böhringer D, Fritz M, et al. Hyperosmolar eye drops for diurnal corneal edema in Fuchs’ endothelial dystrophy: a double-masked, randomized controlled trial. Ophthalmology. 2021;128(11):1527-1533.
9. Coroneo MT, Helbig H, Korbmacher C, Wiederholt M. Effect of hypotonic media on the membrane voltage of cultured bovine corneal endothelial cells. Curr Eye Res. 1989;8(9):891-899.
10. Tran JA, Jurkunas UV, Yin J, et al. Netarsudil-associated reticular corneal epithelial edema. Am J Ophthalmol Case Rep. 2022;25:101287.
11. Chen H, McMillin JC, Frankfort BJ, Al-Mohtaseb Z. Reticular epithelial edema: an uncommon side effect of ROCK/NET inhibitor netarsudil. J Glaucoma. 2020;29(11):e124-e126.
12. LoBue SA, Moustafa GA, Vu A, Amin M, Nguyen T, Goyal H. Transient reticular cystic corneal epithelial edema with topical netarsudil: a case series and review. Cornea. 2021;40(8):1048-1054.
13. Liu KC, Gupta D. Netarsudil-associated reticular corneal epithelial edema with raised intraocular pressure. Ophthalmol Glaucoma. 2019;2(3):166.
14. Chang DF, Masket S, Miller KM, et al; ASCRS Cataract Clinical Committee. Complications of sulcus placement of single-piece acrylic intraocular lenses: recommendations for backup IOL implantation following posterior capsule rupture. J Cataract Refract Surg. 2009;35(8):1445-1458.