
Movement of an IOL that leaves the pupil only partially covered by the optic is a serious complication of cataract surgery that almost always requires surgical management. I am seeing a growing number of patients at my practice who present with decreased or fluctuating vision because of a highly mobile IOL or a visual axis obstructed by an opacified capsule and retained cortex in the periphery of the capsular bag. The causes of these complications vary, and the surgical solution can be technically challenging.
Some complications associated with primary cataract surgery are incomplete placement of the IOL in the capsular bag, prolapse of vitreous through a posterior capsular tear or in an area of zonular dehiscence, zonular instability or dehiscence and dislocation of the IOL–capsular bag complex, migration of a haptic through an anterior capsular tear, and damage to a haptic during surgery. More often, however, lens subluxation occurs later in the postoperative period as a result of progressive zonular laxity in an eye with pseudoexfoliation (Figure 1), trauma (Figure 2), or subsequent intraocular surgery including pars plana vitrectomy.
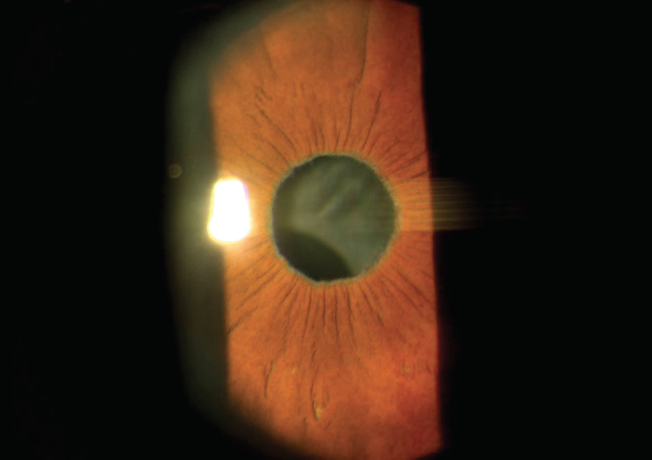
Figure 1. Inferiorly subluxated one-piece acrylic IOL–capsular bag complex in a patient with pseudoexfoliation. Note the exfoliative material at the pupillary margin.
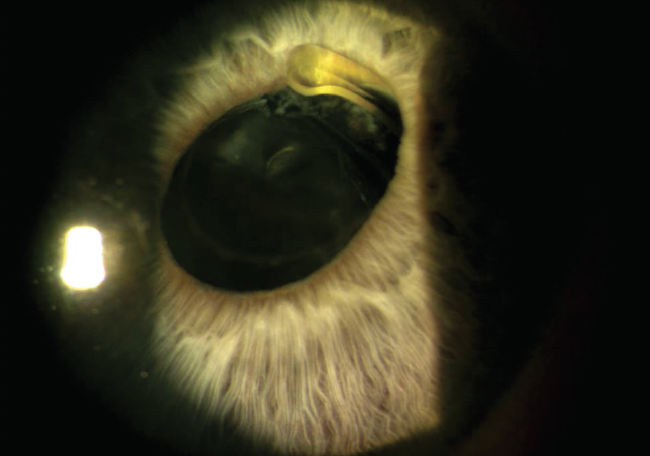
Figure 2. The haptic of a one-piece acrylic lens and surrounding vitreous strands can be seen entrapped in the iris. This patient had a recent history of blunt trauma to the right side of the head as a result of a fall.
Additionally, IOLs fixated with a 10-0 or 9-0 polypropylene suture (Prolene, Ethicon) can move if the suture breaks because of trauma or even eye rubbing or because the suture degrades over time.
This article covers several of these scenarios in detail and shares approaches to their management.
EARLY IOL SUBLUXATION OR DECENTRATION
A patient who presents early in the postoperative period with a subluxated or decentered IOL due to incorrect placement or zonular instability may be taken back to the OR. If one of the haptics was incorrectly placed in the sulcus, the IOL may be repositioned so that both haptics are located in the capsular bag, with or without the addition of a capsular tension ring and/or a capsular tension segment fixated to the sclera (see Preventive Measure; watch the video below).
PREVENTIVE MEASURE
As the number of subluxated lenses I see in my practice seems to increase, my threshold for placing a capsular tension ring during cataract surgery decreases when there is evidence of zonular laxity or of an unstable IOL–capsular bag complex. The use of capsular tension rings and segments at the time of cataract surgery can decrease the risk of future IOL subluxation in eyes with identified zonular dehiscence or instability.
If an IOL placed in the sulcus has dislocated, one option for correction is to reposition the lens in a different axis with better capsular support. Another option is to increase the stability of centration by capturing the optic anteriorly through an anterior capsulotomy or posteriorly through a posterior capsulotomy that is created intra- or preoperatively with an Nd:YAG laser.
DISLOCATION OF THE IOL–CAPSULAR BAG COMPLEX
Surgical planning. Dislocation of the IOL–capsular bag complex is one of the most challenging scenarios to manage in cataract surgery. The surgical plan depends on the type of IOL, the severity of zonular weakness, whether vitreous is present in the anterior segment, and the health of the eye. Preoperative testing includes an endothelial cell count, an examination of the peripheral retina, OCT scans of the macula and optic nerve, and topography. An examination of the sulcus with a gonioprism through a dilated pupil or with ultrasound biomicroscopy can also be helpful by revealing the extent of zonular loss and the amount of retained cortical material.
Take note of iris atrophy, dialysis, and iridodonesis to determine if iris fixation of the IOL is an option. If scleral fixation is planned, an examination of the conjunctiva to identify areas of scarring from prior surgery and areas to avoid such as a filtering bleb can help determine the best surgical approach.
The more information that is gathered before surgery, the more accurate the planning can be. I like to develop at least three plans, with the appropriate lens selected and available for each. Proper preparation of my surgical team is also important. I will review online videos with my surgical staff and talk them through the surgical steps and review which instruments and devices will be required for each option. If the planned technique is new or not one performed recently, it can be helpful to practice the more complicated steps such as suturing (watch the first video below) before surgery with a surgical training eye model such as SimulEye (watch the second video below).
Certain instruments should be on hand such as trocars and vitrectors, iris hooks or pupil expanders, hooks or snares for retrieving a suture, and intraocular microinstrumentation. An intraocular steroid such as triamcinolone acetonide, acetylcholine chloride, sutures, and appropriate needles should also be available.
Anesthesia. Because I plan several possible approaches, I find it best to perform a retrobulbar or peribulbar block with a combination of lidocaine and bupivacaine. Studies have shown that it is not necessary to stop blood thinners before surgery.1,2 I prefer to do so, however, if it can be cleared with the patient’s cardiologist; excessive bleeding of the conjunctival vessels can compromise visualization for scleral fixation. This potential difficulty must, of course, be weighed against the systemic risks of stopping blood thinners.
Another option for anesthesia is to combine the intracameral administration of preservative-free lidocaine with a subconjunctival injection of lidocaine, depending on the procedure being performed. Intravenous sedation can help the patient to remain comfortable during a procedure that is typically longer than the average cataract surgery.
Vitrectomy. An anterior vitrectomy is often required in these cases to free the IOL from vitreous, remove residual cortex and the capsular bag, and remove vitreous that has prolapsed into the anterior chamber. I prefer to use a 23-gauge trocar system inserted through the pars plana. This strategy is easiest to execute when the eye is pressurized before corneal incisions are created. Infusion may be placed through the pars plana or a paracentesis incision with either an anterior chamber maintainer or a cannula on the infusion tubing. I favor a hands-free approach because it allows me to perform the intricate maneuvers needed for suturing lenses more easily.
If the IOL will be retained for fixation, the trocars are placed first, followed by the paracentesis. The anterior chamber is filled with a dispersive OVD, and the lens can then be held with an intraocular microforceps, freed from surrounding vitreous with the vitrector, and prolapsed into the anterior chamber for further cleanup of cortical material and the capsular bag. To minimize the risk of a peripheral retinal tear, care is taken to use a high vitrector cut rate, visualize the cutter through the pupil during cutting, and remove vitreous attachments without pulling on vitreous when manipulating the IOL.
Refixation. IOL material and design often dictate the best approach for refixation. Patients who received premium IOLs and who had good visual acuity and quality of vision before subluxation may be eager to keep their current lenses. One-piece acrylic lenses are not appropriate for placement where the sharp edge of the haptic may rub against the iris (Figure 3). Closed-loop IOLs such as an Akreos (model AO60, Bausch + Lomb) can be fixated to the sclera with a PTFE suture (Gore-Tex, W.L. Gore & Associates) or a 5-0 or 6-0 polypropylene suture using a four-flange technique (watch the video below).

Figure 3. Iris atrophy caused by a one-piece hydrophobic acrylic lens placed in the sulcus.
Plate-haptic lenses often require removal. Sometimes, however, they can be fixated with a suture placed through the eyelet with either a loop of polypropylene (watch the first video below) or a PTFE suture (off-label use) passed through the eyelet or by using a 5-0 or 6-0 polypropylene suture with a flange technique (watch the video below).
Subluxated three-piece IOLs permit the greatest versatility in surgical management. These lenses can often be refixated in lieu of an exchange.
Techniques that preserve conjunctiva include intrascleral haptic fixation. One technique is to externalize the haptics with 30-gauge thin-walled needles, create a flange with low-temperature cautery, and push the flange back into sclera3 (watch the video below). More on the Yamane technique can be learned in the article, “Yamane Tips and Tricks".
In an alternative technique, the haptics are externalized under scleral flaps, the ends of the haptics are pushed into pockets in the sclera, and the scleral flaps are glued in place (watch the video below).
A third alternative is iris fixation. For this strategy, I prefer to use the double-needle technique described by John C. Hart Jr, MD, to prevent distortion of the pupil (watch the video below). Using sutureless needles passed through the peripheral cornea and iris to elevate the haptic and IOL before the placement of permanent iris sutures allows more peripheral suture placement and decreases the chance of creating an oval pupil (Figure 4). The IOL can also be fixated to the sclera with polypropylene or PTFE sutures through a Hoffman corneoscleral pocket,4 also preserving conjunctiva.5
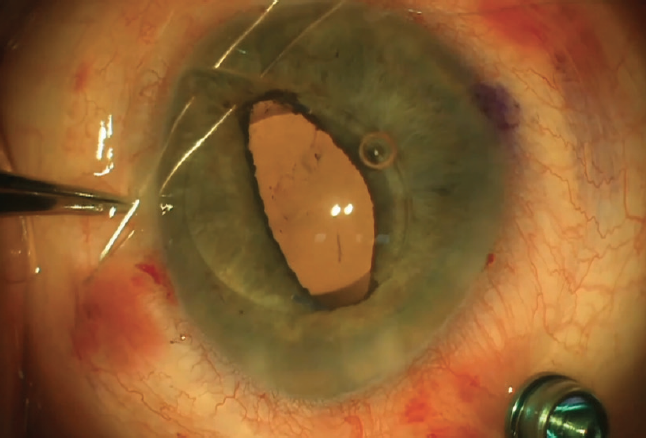
Figure 4. A double-needle technique developed by John C. Hart Jr, MD, allows more peripheral placement of iris sutures and reduces the risk of creating an oval pupil.
Suturing through the capsular bag to the sclera can eliminate the need for an anterior vitrectomy. A polypropylene or PTFE suture needle is docked into 26- or 27-gauge needles passed in front of and behind the haptic (watch the first video below) by retrieving a piece of PTFE suture with a snare in a 26-gauge needle. This can be done using either a technique developed by Soon-Phaik Chee, MD,6 or an ab externo technique described by Garry P. Condon, MD, that also avoids the need for needle docking (click here to watch the video on the ASCRS website).
Alternatively, a 5-0 or 6-0 polypropylene suture in a flanged belt loop configuration may be used to support the IOL–capsular bag complex (Figure 5; watch the first video below).7 In cases of late dislocation, the fibrotic margin of the capsule may be sutured to the iris using an innovative technique developed by Dr. Condon (watch the second video below). In this technique, a 10-0 polypropylene suture on a long, curved needle is passed through a paracentesis, the pupillary margin, the fibrotic margin of the anterior capsulotomy, and the iris and then back through peripheral cornea. Tying the suture with a sliding knot secures the capsular bag to the underside of the iris.
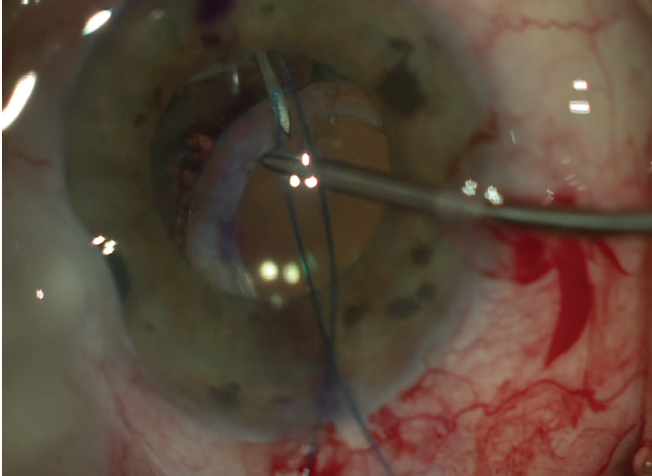
Figure 5. Belt loop technique for fixation of a subluxated IOL–capsular bag complex in a patient with a prior trabeculectomy. Cut segments of a 6-0 polypropylene suture are passed around the haptic and anchored to the sclera by creating flanges with low-temperature cautery after externalizing the suture through the sclera and conjunctiva.
Exchange. In some cases, an IOL exchange is required:
- The IOL has been damaged (perhaps multiple central pits were created in the lens during an Nd:YAG laser capsulotomy);
- The lens design is not compatible with ocular health (eg, the patient developed retinal disease after receiving a multifocal lens); and
- An incorrectly powered IOL was implanted.
An IOL exchange almost always requires an anterior vitrectomy, especially if the IOL–capsular bag complex has subluxated. After IOL removal and a thorough anterior vitrectomy, a secondary IOL, either an anterior chamber lens or a fixated posterior chamber lens, is inserted. Many of the techniques are the same as those described earlier but with the advantage of choosing the lens power, design, and fixation technique most suitable to the patient’s ocular anatomy and health as well as for the surgeon’s level of experience and comfort level. (For more on IOL exchange, see “Indications for IOL Exchange,” and “When to Explant a Presbyopia-Correcting IOL and What to Put in Its Place,".)
After the IOL is securely in place and centered, my preference is to instill triamcinolone acetonide to check for vitreous, to constrict the pupil with an intracameral miotic, and to perform an intracameral injection of an antibiotic (moxifloxacin or cefuroxime). Because these patients have usually received a retrobulbar block, a topical antibiotic/steroid ointment, a patch, and a shield are placed.
I also like to administer a topical IOP-lowering medication such as a fixed combination of brimonidine and timolol. If I am concerned about an IOP spike due to a retained OVD, I instruct the patient to administer a sustained-release acetazolamide tablet at bedtime. Postoperatively, I monitor the IOP and prescribe topical antibiotic, steroid, and NSAID medications.
CONCLUSION
Subluxated IOLs are a surgical challenge. These cases require unique skills, a creative mindset, and a variety of tools and tricks to achieve a successful result. It is therefore worthwhile for surgeons to practice and develop a variety of techniques in order to be well prepared. I find it immensely rewarding to develop solutions tailored to the specific situation that restore the patient’s vision.
1. Dayani PN, Grand MG. Maintenance of warfarin anticoagulation for patients undergoing vitreoretinal surgery. Trans Am Ophthalmol Soc. 2006;104:149-160.
2. Grand MG, Walia HS. Hemorrhagic risk of vitreoretinal surgery in patients maintained on novel oral anticoagulant therapy. Retina. 2016;36(2):299-304.
3. Yamane S, Sato S, Maruyama-Inoue M, Kadonosono K. Flanged intrascleral intraocular lens fixation with double-needle technique. Ophthalmology. 2017;124(8):1136-1142.
4. Video: speaker discusses the Hoffman pocket technique. Ocular Surgery News. November 4, 2015. Accessed April 12, 2021. https://www.healio.com/news/ophthalmology/20151104/video-speaker-discusses-the-hoffman-pocket-technique
5. Hoffman RS, Fine IH, Packer M. Scleral fixation without conjunctival dissection. J Cataract Refract Surg. 2006;32(11):1907-1912.
6. Chee SP, Chan NS. Suture snare technique for scleral fixation of intraocular lenses and capsular tension devices. Br J Ophthalmol. 2018;102(10):1317-1319.
7. Canabrava S, Canêdo Domingos Lima AC, Ribeiro G. Four-flanged intrascleral intraocular lens fixation technique: no flaps, no knots, no glue. Cornea. 2020;39(4):527-528.




