CASE PRESENTATION
A 64-year-old woman presents for a cataract evaluation. The patient has a history of cataract surgery on the right eye and received an AcrySof IQ PanOptix Toric IOL (Alcon). She has not undergone cataract surgery on the left eye.
The patient complains of hazy vision in the right eye that is not clear at any distance. She has a cataract in the left eye. Her BCVA is -0.50 +1.50 x 175º = 20/20 OD and -4.00 +2.25 x 105º = 20/30 OS.
A slit-lamp examination reveals a clear cornea and no abnormalities in either eye. The toric IOL in the right eye is well centered on the 95º axis (Figure 1). A 2+ nuclear sclerotic cataract with 1+ cortical changes and an early posterior subcapsular cataract are present in the left eye. Topographic and epithelial thickness maps and biometry measurements are shown in Figures 2, 3, and 4, respectively.
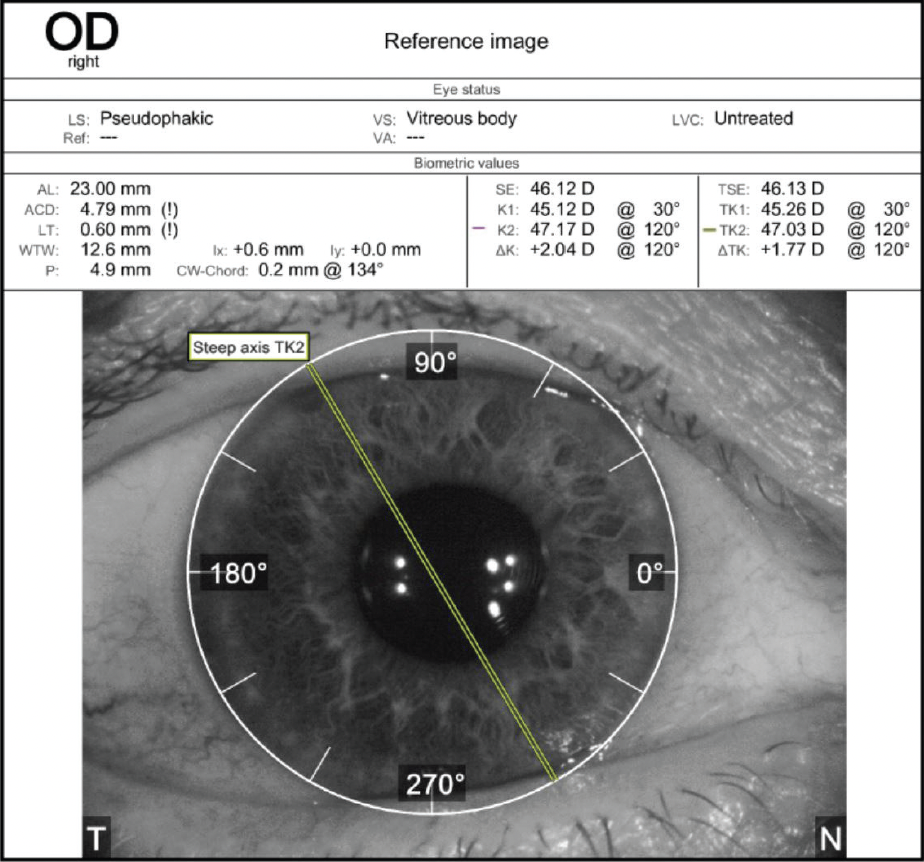
Figure 1. Orientation of the toric IOL in the right eye.

Figure 2. Topography of the right (A) and left (B) eyes.
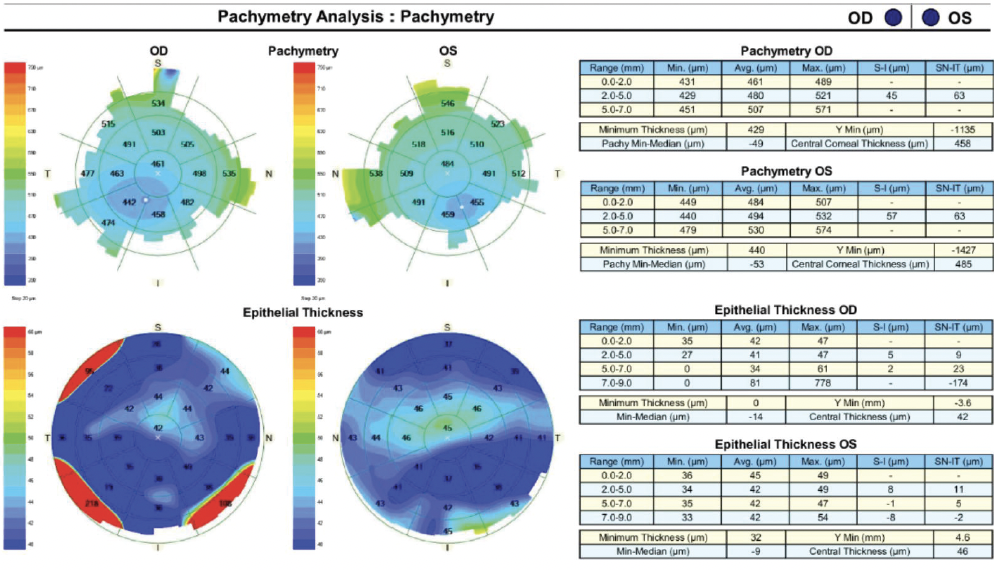
Figure 3. Pachymetry and epithelial thickness mapping of both eyes.
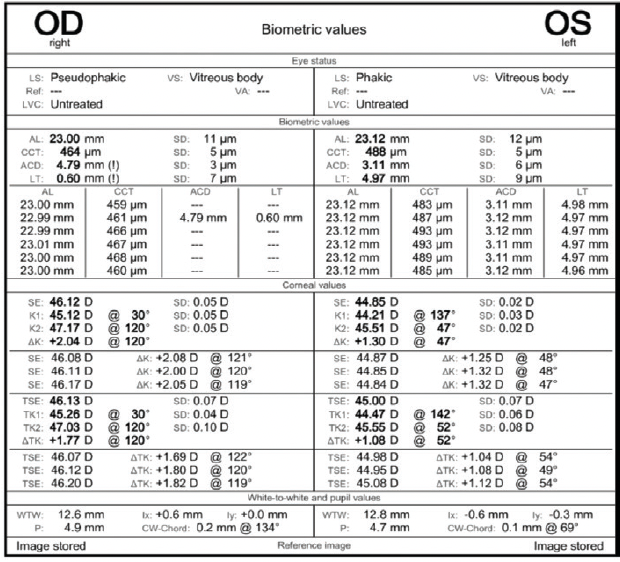
Figure 4. Biometry measurements of both eyes.
How would you proceed? If you would perform an IOL exchange in the right eye, which IOL would you consider implanting? If you favor a toric IOL, on which axis would you place it? Considering that this patient underwent cataract surgery by another ophthalmologist who did not check preoperative tomography or topography, how would you explain the patient’s condition to her?
—Case prepared by Audrey R. Talley Rostov, MD

NEDA NIKPOOR, MD
This patient has undetected keratoconus, which is the likely cause of her poor vision after cataract surgery and the implantation of a trifocal IOL. This is a tricky situation, and communicating the diagnosis requires educating the patient without blaming her surgeon. I would explain to the patient that she has mild keratoconus that could easily have been missed if topography had not been performed. I would use the Pentacam (Oculus Optikgeräte) as a visual aid when educating her about keratoconus and explaining that the corneal shape and resultant astigmatism are asymmetrical, leading to poor vision.
It is important to assess the patient’s refraction qualitatively and quantitatively. If a refraction can produce crisp, clear 20/20 VA and a quality of vision that is acceptable to her, it would be worth considering an IOL rotation or exchange. Otherwise, a trial with a rigid gas permeable (RGP) contact lens should help to determine whether the cornea or the lens is causing the hazy vision. I find it helpful to use the iTrace (Tracey Technologies) and the website astigmatismfix.com when deciding whether to rotate an IOL.
Because her BCVA is poor, I would perform an IOL exchange. A monofocal IOL would be the safest option, followed by a monofocal toric. I would, however, consider implanting an extended depth of focus IOL. If the patient wants the highest chance of a satisfactory outcome and does not mind wearing an RGP lens, I would recommend the implantation of a monofocal IOL followed by a contact lens fitting.

NEDA SHAMIE, MD
Corneal topography and anterior segment imaging in this case clearly demonstrate significant irregular astigmatism and findings consistent with moderate keratoconus. If the keratoconus had been detected at the time of the cataract procedure, it is unlikely that a trifocal IOL would have been recommended because the higher-order aberrations of an irregular keratoconic cornea can have a significantly negative impact on visual outcomes with a multifocal lens.
Because a trifocal IOL has been implanted, the goal here is to salvage the results if possible. The blurry vision that the patient described may be the result of the residual refractive error. I would determine whether her vision and blur improve when her refractive error is corrected with trial frames in the office. If correcting the refractive error sufficiently improves vision such that the patient’s expectations are met, I would consider rotating the toric IOL. The problem is that corneal refractive surgery is not an option for this patient because of her corneal status. She therefore might need to consider wearing glasses or contact lenses. If correcting the refractive error does not address the patient’s visual complaints, my next step would be to rule out comorbidities such as dry eye disease and tear dysfunction, posterior capsular opacification, and maculopathy before viewing the irregular astigmatism as the only culprit. An RGP overrefraction can help to determine if the cornea is the cause of the patient’s hazy vision.
In my opinion, the best option for the patient would be to exchange the trifocal IOL for a small-aperture IOL, but the latter is not yet available in the United States, where I practice. If she is willing to wait until this lens becomes available, I would encourage her to do so. Otherwise, I would explant the trifocal IOL and implant a monofocal IOL with the expectation that the best quality of vision will likely be achieved with an RGP or scleral lens postoperatively.
To address the patient’s questions, I would explain that a preexisting corneal condition is the reason for her suboptimal outcome and that this condition is not easily detected by the basic workup done for cataract surgery. I would further explain that it is not unusual for previously undetected eye conditions to be discovered only after further evaluation to determine the cause of a suboptimal outcome after uneventful cataract surgery.

ROGER ZALDIVAR, MD, MBA
There are two points I wish to emphasize. First, diffractive IOLs are for healthy eyes. Second, when the decisions made by an earlier surgeon were far from ideal, we must be careful about our comments and facial expressions.
The implantation of a diffractive trifocal IOL is generally an excellent strategy for achieving spectacle independence. Three things, however, have become apparent through collective surgical experience:
- No. 1: These lenses are not forgiving of residual refractive error or poor centration;
- No. 2: Enhancement surgery is required to address residual ametropia or a poorly positioned IOL; and
- No. 3: Tear film stability is important. With more than 10.00 D of superior/inferior asymmetry, the results of topography for both of this patient’s eyes are far from ideal.
I would not attempt to regularize the corneal surface because the patient’s BCVA is 20/20. Instead, I would perform an IOL exchange. A great option in situations like this one is to filter defocused light with the aid of small-aperture optics such as an IC-8 IOL (AcuFocus). It is worth considering the bilateral implantation of small-aperture IOLs in a patient with highly aberrated eyes.
I would carefully explain the patient’s clinical condition to her without burying the original surgeon. I would emphasize that, although the original IOL technology is excellent, a more forgiving IOL should provide greater comfort.

WHAT I DID: AUDREY R. TALLEY ROSTOV, MD
Epithelial mapping and tomography with the Pentacam demonstrated keratoconus. I elected to perform an IOL exchange and chose an enVista IOL (model MX60, Bausch + Lomb) for two reasons. First, this lens has zero spherical aberrations, which I thought would be advantageous in the presence of a keratoconic cornea. Second, multifocal IOLs are contraindicated for eyes that have a multifocal cornea.
The trifocal IOL was cut with scissors and removed with forceps. A -1.25 D postoperative refraction was targeted. I explained to the patient that she would need to wear glasses to see well at all distances. Her postoperative BCVA was 20/20 without the prior dysphotopsias, and she was pleased with her outcome.
I explained to the patient that keratoconus can be missed unless advanced testing is done.




