Compromised Corneal Endothelium
Sara Bozorg, MD

Once you decide an IOL exchange is the best next option for a patient, it is time to make a surgical plan. Careful consideration of the patient’s history is required, including prior surgical procedures and comorbid ocular disease.
SEVEN PEARLS
This article shares seven pearls for performing an IOL exchange in an eye with compromised corneal endothelium.
No. 1: Counsel the patient. Obtain an endothelial cell count if possible and counsel the patient that an IOL exchange could lead to further endothelial cell loss and the possible need for an endothelial keratoplasty (EK). If the patient already has a corneal transplant, an IOL exchange may cause the current graft to fail, necessitating a repeat EK. When available, alternatives to IOL exchange merit strong consideration.
No. 2: Develop a preoperative plan and backup plans. Acquire as much information as possible on the lens being explanted, including make and model, characteristics, material, and design (ie, one or three pieces). This information can help you decide if the lens is amenable to folding, cutting, or twisting and promote precision during explantation, minimizing trauma to the endothelium. Formulate a step-by-step plan that lists all potentially necessary instruments (Figure 1).1

Figure 1. Preoperative planning sheets can help the surgeon and scrub/circulating nurses in the OR prepare.
No. 3: Be gentle. Follow your plan and be even more gentle than you usually are during surgery. Be generous in your use of a dispersive OVD2 because it is cheaper than endothelial cells. Always maintain awareness of your instruments, especially when they are entering and exiting the eye.
No. 4: Do not be afraid to enlarge the incision. Minimize the chances that a haptic will contact the endothelium during lens explantation. Whichever technique you use to remove the IOL, a second instrument can be used to protect the endothelium as you maneuver the lens (Figure 2). If necessary, enlarge the wound slightly to prevent the lens from putting stress on the incision or contacting the endothelium on the way out.
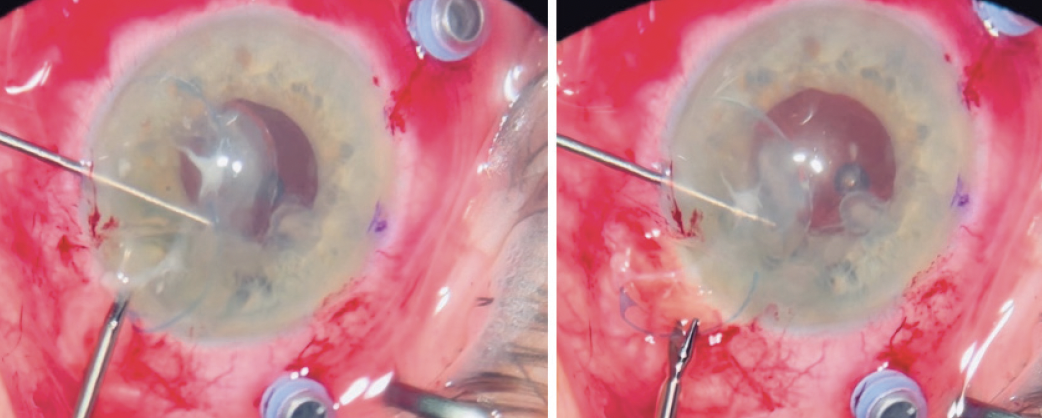
Figure 2. A second instrument in the nondominant hand is used to protect the endothelium as the IOL is gently maneuvered out of the main incision.
Figures 1 and 2 courtesy of Sara Bozorg, MD
No. 5: Avoid leaving vitreous in the anterior chamber. This tip may seem obvious. The goal is to avoid additional surgery and reasons to return to the OR. If you are exchanging an IOL in the setting of an open posterior capsule, instilling triamcinolone acetonide at the end of the case can help ensure that all vitreous has been removed from the anterior chamber. No vitreous should be left in contact with the endothelium.
No. 6: Select the new IOL carefully. Avoid hydrophilic acrylic IOLs in patients who may need an EK in the future because the incidence of IOL calcification is greater in the setting of gas and air.3 This well-known phenomenon can affect the visual outcome and may necessitate another IOL exchange in the future.
No. 7: Be ready for the unexpected. Surgical surprises are not uncommon in these eyes. Have ready all the instruments you may need for plans A through D.
Plan A: The goldilocks plan. The bag and zonules are intact. The original lens is explanted, and a new lens is placed.
Plan B: The posterior capsule is open or the zonules are weak. It may be possible to perform sulcus fixation of the lens and posterior optic capture. If the zonules are compromised but the bag is otherwise intact, one option is to place the IOL in the sulcus and lightly suture it to the iris. A recent study showed no clear difference in final endothelial cell count between iris- and scleral-sutured IOLs.4
Plan C: The bag cannot be saved. There are generally three options for lens placement in this situation: an anterior chamber IOL, an iris-fixated IOL, or a sulcus-fixated IOL. The last can be a sutured Monoflex (model CZ70BD, Alcon)5 or Akreos or enVista (both from Bausch + Lomb), a variation on the glued IOL technique first described by Amar Agarwal, MS, FRCS, FRCOphth,6 or the Yamane technique for intrascleral haptic fixation.7
Suturing a Monoflex IOL can be a useful option in this situation because of its 7-mm optic, which may provide a larger diaphragm to support gas/air if needed during a future EK and less potential space for a thin graft to slip behind during insertion.
A small-incision secondary IOL with the Yamane technique is also a good option because it minimizes manipulation of the eye, the risk of hypotony, and potentially the opportunity for endothelial cell loss.
In general, the best plan C lens is the one with which you are the most comfortable.
Plan D: Surgery is aborted. Halting surgery and either coming back another day or referring the patient to a colleague is always an option and sometimes the best course of action.
CONCLUSION
IOL exchange in the setting of a corneal graft or dystrophy can be challenging. With appropriate patient counseling and preoperative planning, surgery can achieve an excellent outcome.
1. Ahmed IIK, Campos-Möller X, Shah M, Sheybani A. Illustrated Advanced Anterior Segment Surgery: A Step-by-Step Guide for Challenging Cases. SLACK; 2021.
2. Park SSE, Wilkinson SW, Ungricht EL, et al. Corneal endothelium protection provided by ophthalmic viscosurgical devices during phacoemulsification: experimental study in rabbit eyes. J Cataract Refract Surg. 2022;48(12):1440-1445.
3. Balendiran V, MacLean K, Mamalis N, Tetz M, Werner L. Localized calcification of hydrophilic acrylic intraocular lenses after posterior segment procedures. J Cataract Refract Surg. 2019;45(12):1801-1807.
4. Lau THA, Garg A, Popovic MM, Kertes PJ, Muni RH. Scleral-fixated and iris-fixated intraocular lens implantation or fixation: meta-analysis. J Cataract Refract Surg. 2022;48(12):1462-1468.
5. Snyder ME, Perez MA. Tiltless and centration adjustable scleral-sutured posterior chamber intraocular lens. J Cataract Refract Surg. 2014;40(10):1579-1583.
6. Agarwal A, Kumar DA, Jacob S, et al. Fibrin glue-assisted sutureless posterior chamber intraocular lens implantation in eyes with deficient posterior capsules. J Cataract Refract Surg. 2008;34(9):1433-1438.
7. Yamane S, Sato S, Maruyama-Inoue M, Kadonosono K. Flanged intrascleral intraocular lens fixation with double-needle technique. Ophthalmology. 2017;124(8):1136-1142.
Anterior Capsular Phimosis
Ben LaHood, MBChB(DIST), PGDipOphTH(DIST), PhD, FRANZCO

Exchanging an IOL can be stressful, especially in the presence of anterior capsular phimosis, when the shrunken anterior capsulorhexis, zonular stress, and likelihood of the IOL’s adhering to the capsule make lens removal more challenging. The timing and method of treatment are determined based on the severity of the condition and the equipment available. I establish a primary plan and IOL choice and multiple backup options that can be used depending on the intraoperative findings.
PREVENTION
Capsular phimosis is associated with a small capsulorhexis, zonular pathologies such as pseudoexfoliation syndrome, and systemic conditions such as diabetes. It is unclear whether overlap of the edge of the capsulorhexis onto the IOL optic is beneficial or whether leaving the optic uncovered facilitates adhesion between the anterior and posterior leaflets.
Implanting a capsular tension ring in eyes with zonular weakness or loss can help reduce the risk of phimosis. Additionally, leaving the anterior capsular leaflet unpolished can facilitate capsular adhesion, help stabilize the IOL, and reduce the risk of posterior capsular opacification. It can also, however, make IOL explantation more challenging. I therefore polish the anterior capsule whenever I believe there is a high risk of phimosis and IOL exchange and leave it unpolished if the risk is low.
I prefer to implant a hydrophilic IOL in eyes with preexisting risk of phimosis because this type of implant is easier to exchange than a hydrophobic IOL should that procedure be necessary (Figure 3). For mild and stable phimosis, I do not touch the capsule.
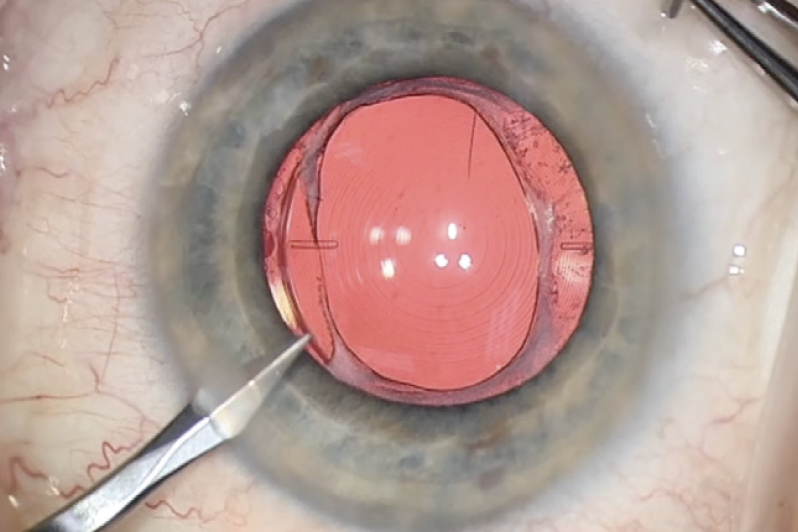
Figure 3. The eye has mild anterior capsular phimosis and a well-centered hydrophilic IOL. No capsule intervention is required for this IOL exchange.
INTRAOPERATIVE TREATMENT STRATEGIES
My treatment strategy varies depending on the severity and stability of the phimosis.
Preoperative radial relaxing incisions. This is my preferred technique. It is the most controlled strategy in my hands and provides the most information on the eye before entry. Oftentimes, it is possible for me to determine whether there is adherence between the IOL and capsule. During treatment, the anterior capsule is cut in four to eight radial directions with an Nd:YAG laser to release the contracting pressure symmetrically (Figure 4). The IOL can become pitted easily, so care is taken to remove any posterior offset from the Nd:YAG settings.
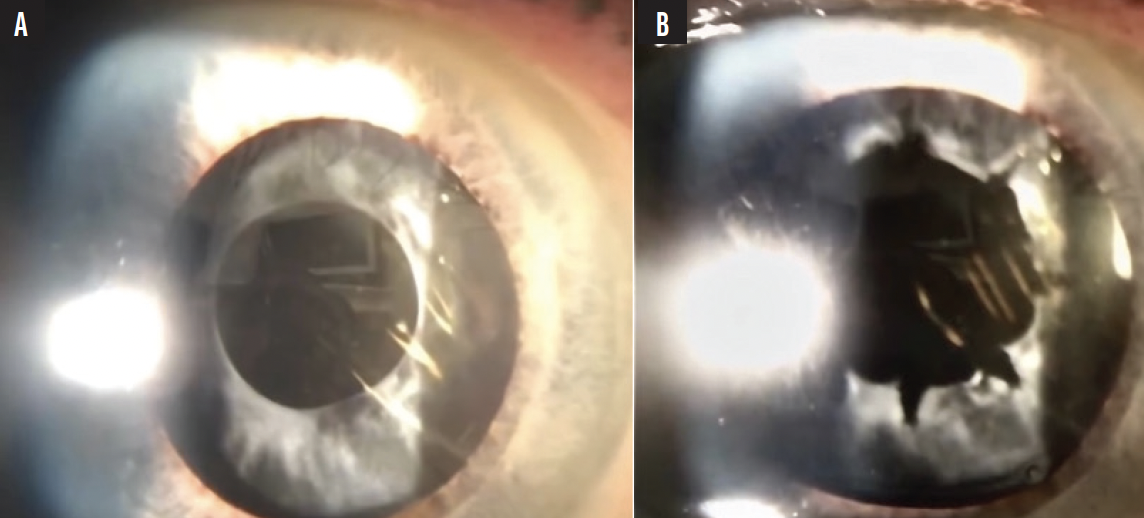
Figure 4. Anterior capsular phimosis before (A) and after (B) relaxing incisions are created in the anterior capsule with an Nd:YAG laser.
Figures 3 and 4 courtesy of Ben LaHood, MBChB(dist), PGDIPOPHTH(dist), PhD, FRANZCO
The central rim of the phimosis is usually the thickest and toughest to cut. It is possible to cut a complete ring of contracted capsule to provide a wider aperture. If it does not fall away as planned, however, further intervention is required.1 Another tip for preoperative radial relaxing incisions is to use the Nd:YAG laser to create additional holes in the anterior capsule. These can later be used as ports for the injection of an OVD to help dissect the IOL.
Intraoperative relaxing incisions. This strategy is a good option for particularly thick fibrotic capsules because they would require high power with an Nd:YAG laser and pose a higher risk of lens pitting. At the time of IOL exchange surgery, the anterior capsule is viscodissected away from the lens. Intraocular scissors are used to make at least two opposing radial incisions to relieve tension. Placement is crucial to avoid IOL decentration from any subsequent contraction of the capsule.
Excision of the fibrosed capsule. Severe phimosis can leave minimal room for the passage of instruments. An Nd:YAG laser treatment is not advisable because precise, central placement is crucial. Some think that pitting the IOL should not matter if it is to be exchanged, but sometimes the exchange surgery cannot be completed. It is therefore best to do as little damage as possible.
The anterior vitrector can be used to cut bites out of the anterior capsule. The fibrotic phimosis can then be removed and the capsulorhexis enlarged. This strategy is particularly good in the long term for cases of progressive anterior capsular phimosis. The clean bites from the vitrector also decrease the risk of radial extensions of the tear during IOL exchange surgery.2
Femtosecond laser capsulorhexis recutting. Another useful strategy for severe phimosis that leaves a small or no opening in the capsule is to create a new capsulorhexis with a femtosecond laser.3 This is the least common of the techniques.
Exchanging the IOL for a scleral- or iris-fixated IOL. Removing both the pathologic capsule and an unwanted IOL can be an elegant solution. I choose this technique if the IOL is significantly decentered or tilted and the capsular pathology appears to be progressive. If the capsule can be preserved, I exchange the IOL and secure the new IOL within the capsule. In eyes with pseudoexfoliation and significant pseudophakodonesis, I prefer to remove the IOL and the capsular bag together. A scleral tunnel is used to avoid inducing more astigmatism. There are many techniques for scleral and iris fixation. In my hands, the Yamane technique is the simplest and requires the least amount of operating time, reducing the loss of corneal endothelial cells.
CONCLUSION
Anterior capsular phimosis can make IOL exchange more challenging. The techniques outlined here can help improve a surgeon’s chance of success. Safely enlarging the capsular opening before or during surgery is required in all but the most mild cases. For severe cases with compromised zonules, removing the existing IOL and capsular bag together can be considered.
1. Wilde C, Ross A, Awad M, Chen HC, Dua HS. Management of anterior capsular contraction syndrome: pitfall of circular capsulotomy technique with the neodymium YAG laser. Eye. 2018;32(9):1546-1548.
2. Yeh PC, Goins KM, Lai WW. Managing anterior capsule contraction by mechanical widening with vitrector-cut capsulotomy. J Cataract Refract Surgery. 2002;28(2):217-220.
3. Gerten G, Schultz M, Oberheide U. Treating capsule contraction syndrome with a femtosecond laser. J Cataract Refract Surg. 2016;42(9):1255-1261.
The Presence of Zonulopathy
Brian Shafer, MD

There are two components of a successful IOL exchange in the setting of zonulopathy: the explant and the implant.
THE EXPLANT
The goal is to ensure that surgery does not worsen the status of the zonules. I rely on eight pearls to accomplish this.
No. 1: Confirm that the IOL has not fully dislocated before starting the case. You do not want to administer a retrobulbar block, complete prepping and draping, and make the first incision only to realize that the IOL is sitting on the retina and your retina partner is busy in clinic and unable to help. (Ask me how I know.)
No. 2: Administer a retrobulbar block or have the patient under general anesthesia. Surgery can go differently than expected. Proper analgesia and patient comfort are crucial.
No. 3: Place a trocar for posterior infusion. This can help maintain the IOP and minimize the risk of posterior dislocation of lens material or the IOL. If a capsulotomy has been performed, a pars plana anterior vitrectomy can be used to minimize the risk of vitreous prolapse during the exchange.
No. 4: Dissect the anterior capsule away from the IOL by injecting a dispersive OVD from a bevel-down 26-gauge needle. The sharp needle can enter the plane easily, minimizing the amount of force required to separate the capsule from the IOL.
No. 5: Place capsular hooks early. When zonular support is poor, it is easy to create more zonulopathy. Capsular hooks can help minimize iatrogenic zonular dehiscence.
No. 6: Reinflate the remaining capsular bag with an OVD. Ensure that an adequate amount of an OVD fills the bag out to the equator where the haptic tips will be freed.
No. 7: Free the haptics with a curved instrument such as a Sinskey hook. Make sure a capsular hook is positioned just anterior to the haptic tips so that additional stress is not placed on the zonules while you attempt to free them.
No. 8: Explant the IOL using the technique of your choice. I favor bisecting the optic and removing the two halves. Whatever technique you use, be gentle on the endothelium.
THE IMPLANT
Once the original IOL is out, it is time to determine how to ensure a stable configuration of the new IOL (Table). How to proceed depends on the answers to two questions: (1) Is the posterior capsule intact, and (2) how severe is the zonulopathy?
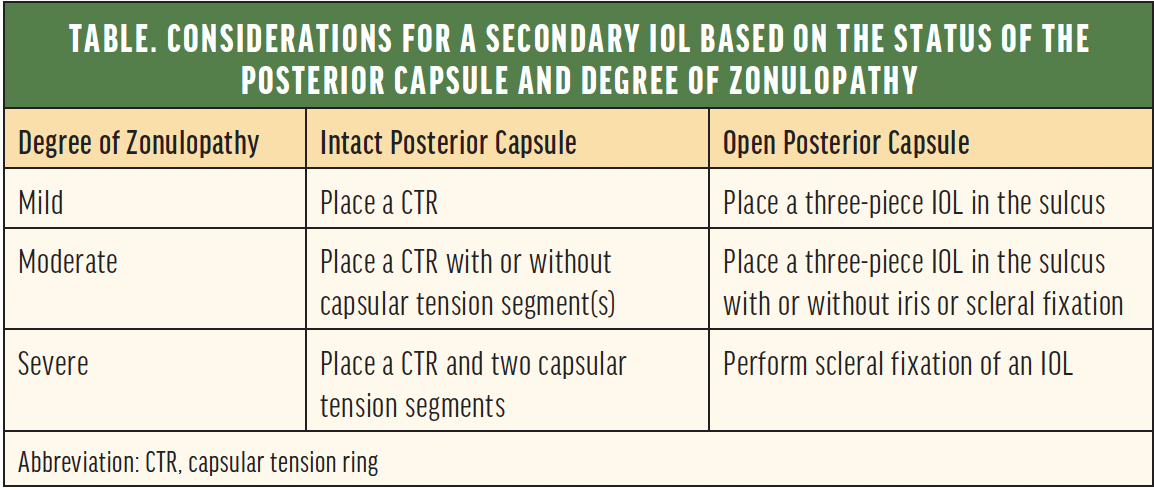
If the posterior capsule is intact, the options resemble those for primary cataract surgery in an eye with intraoperative zonular dehiscence. Depending on the degree of zonulopathy, plan to place a capsular tension ring with or without sutured capsular tension segments. At this point, any IOL may be implanted in the capsular bag. In most cases, there is a high chance of long-term stability.
More often than not, the posterior capsule has been opened with an Nd:YAG laser or iatrogenically during complicated cataract surgery. When the capsule is open, the surgical plan depends on the degree of zonulopathy (ie, mild, moderate, or severe). In all cases, consider implanting an IOL with polyvinylidene fluoride haptics to ensure that intrascleral haptic fixation can be performed in the future if necessary.
Mild zonulopathy. Place a three-piece IOL in the sulcus with or without optic capture. Ensure that the haptics are not positioned in an area of zonular dialysis to prevent tilt or sunsetting.
Moderate zonulopathy. Place a three-piece IOL in the sulcus with optic capture. If the lens is unstable, consider iris or intrascleral haptic fixation. Iris fixation minimizes the risk of lens tilt and decentration, but it increases the risk of uveitis-glaucoma-hyphema syndrome. Intrascleral haptic fixation minimizes the risk of the syndrome but carries the risks of tilt, decentration, and—over the long term—conjunctival erosion.
Severe zonulopathy. There is no utility to keeping the capsular bag in this situation. Remove it entirely and be careful to prevent posterior migration of the lens and capsular remnants. At this point, perform scleral fixation of the IOL of your choice.
CONCLUSION
Performing an IOL exchange in the setting of zonulopathy is challenging but obtainable with proper preparation. Because surgery is complex, postoperative healing time may be prolonged, and proper preoperative counseling is essential. Surgeries that start with a complication typically present unique obstacles. The reward of success is a happy patient.