
In my pediatric referral practice, I routinely perform CXL on children who are 10 to 13 years of age. Most clinicians don’t think of patients in this age group as having keratoconus. Nor do clinicians generally consider CXL to be a viable treatment option for them. However, evidence suggests that keratoconus is much more prevalent in pediatric patients than previously thought and that we physicians aren’t looking closely enough for the disease, especially in the pediatric population.1,2 Historically, keratoconus was thought to affect about one in every 2,000 people. Recent studies have shown that the prevalence may be as high as one in every 375 people or, in certain populations, one in every 21 people.1,2
Keratoconus can reach an advanced stage in very young patients and leave them with 20/800 or count fingers BCVA. We should have a high level of concern about young people with even mild ectatic disease. A 10-year-old has little natural crosslinking of the collagen fibers. Evidence suggests that keratoconus presentation at a younger age indicates a more aggressive and rapidly progressing form of the disease (Figure).3 Because years will elapse before crosslinking naturally occurs in children’s corneas, they are at risk of a significant loss of visual function.
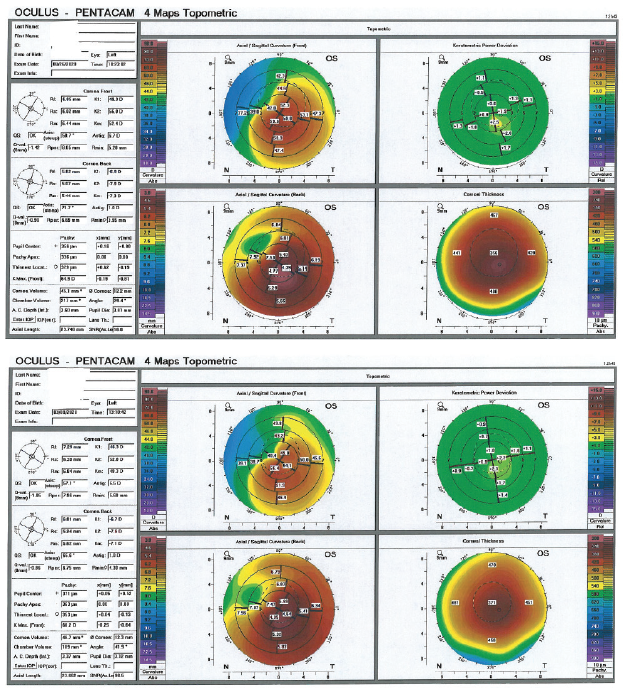
Figure. Anterior segment imaging shows significant keratoconus progression in the left eye of a 13-year-old patient from the time of the first examination (top) to the second examination 5 months later (bottom).
Screening Children
Refractive error changes throughout childhood and adolescence, but the level of astigmatism should be fairly stable. New or worsening astigmatism is a red flag for the presence of keratoconus. A myopic shift beyond 1.00 D per year can suggest an abnormal cornea.
Although it fell out of favor with the advent of new retinal imaging technologies, dilated retinoscopy is an excellent screening tool for keratoconus in children because retinoscopic changes typically occur much earlier than refractive changes or corneal signs of the condition. For young patients at increased risk of developing keratoconus (see Risk Factors for Pediatric Keratoconus), I strongly recommend performing pupillary dilation and retinoscopy as a first step. Topography is warranted in a patient with visible warpage on retinoscopy to confirm the diagnosis.
RISK FACTORS FOR PEDIATRIC KERATOCONUS
- Change in astigmatism
- Rapidly changing refractive error
- 20/20 BCVA cannot be achieved, and no other explanation is available (amblyopia is a diagnosis of exclusion)
- Family history of keratoconus or corneal transplantation
- Personal history of eye rubbing (atopic)
- Down syndrome
Keratoconus is associated with both genetic and environmental factors.4 I have heightened concern about a patient with Down syndrome, known atopic conditions with significant eye rubbing, or a family history of keratoconus. Some clinicians consider genetic testing for pediatric patients with a family history of the disease. There is no single genetic locus for keratoconus; however, researchers have identified gene variants associated with the disease. The first commercially available genetic test for keratoconus (AvaGen, Avellino Labs) uses a cheek swab and can identify more than 1,000 variants across 75 genes associated with keratoconus and more than 70 mutations of the transforming growth factor beta–induced gene for corneal dystrophies. Data on the sensitivity and specificity of this test are limited.
Considerations for Treatment
No disease is known to mimic keratoconus in pediatric patients. If topographic and retinoscopic evidence of the disease is present in a patient who is younger than 14 years of age, I believe that child should undergo CXL as soon as possible. The only system approved for CXL in the United States is the iLink corneal remodeling platform (Glaukos), which uses the company’s KXL System and either its riboflavin 5’-phosphate in 20% dextran ophthalmic solution (Photrexa Viscous) or riboflavin 5’-phosphate ophthalmic solution (Photrexa). The iLink was not tested in patients younger than 14 years of age in the clinical trials, so its use in this population is off-label. (Editor’s note: For more on off-label use of a product, see “Understanding On-Label, Off-Label, and Unapproved Products.”)
When speaking to parents about the procedure, I disclose that this is an off-label indication. I have recently had little trouble getting insurers’ approval for pediatric CXL, even without documentation of disease progression. Significant loss of BCVA can occur in these children in as little as 6 months, so it is counterproductive to wait. I also talk with parents about the pros and cons of same-day bilateral treatment versus sequential unilateral treatment. The latter approach requires deciding whether to treat the more impaired eye first to prevent further disease progression or the better-seeing eye in an attempt to restore function more quickly.
Performing CXL with the patient under general anesthesia is much more common in pediatric patients than in adults, and it is safe and effective in a pediatric population.5 My youngest patients (9–10 years old) can rarely sit still with a speculum in their eye long enough for CXL. Those with anxiety disorders or underlying developmental disorders may also benefit from general anesthesia in a hospital setting. Also, I consider whether I want to follow the standard practice of placing a bandage contact lens after CXL. If I don’t think I will be able to remove the lens while the child is awake, I opt instead for a collagen shield that dissolves after 72 hours.
Outcomes
Results of CXL in pediatric patients have been excellent,3 with no statistically significant differences in efficacy observed between those who undergo treatment before or after the age of 17.6 Young patients are more likely than adults to have an aggressive wound-healing response to CXL, leading to the development of scar tissue, but there is no evidence to suggest that children’s corneas absorb riboflavin or UV light differently from adults’.
One CXL procedure is enough to stop ectatic progression in most adults. Research suggests that about 7% of all patients treated with CXL experience ectatic progression and may need retreatment. In pediatric populations, progression rates of 20% to 25% have been reported after CXL.3,6 Children who have undergone CXL should therefore be monitored closely for signs of ectatic progression.
A recent study found epithelium-off CXL to be safe and effective in children who were 13 to 18 years of age. These patients experienced 2-line improvements in UCVA and BCVA, a 2.20 D reduction in topographic astigmatism, and a 3.72 D decrease in maximum keratometry values. The rate of ectatic progression over 3 years was 13% in this study.7
Conclusion
I am aggressive about performing CXL on children with advanced keratoconus, even at the same stage for which I would recommend a corneal transplant in an adult. Transplants require long-term steroid therapy, suture removal, and lots of doctor visits; they carry the risks of rejection, aggressive wound healing, and repeat surgeries; and they restrict patients from playing sports. For these reasons, even when I think a child will eventually need a transplant, I talk to the parents about delaying the procedure for as long as possible. I have been amazed by the visual recovery—from very poor initial vision to 20/30 or 20/40—that can be achieved with scleral lenses after CXL in children.
Now that CXL is more widely available, I strongly encourage my colleagues who see pediatric patients for routine eye care to screen them more closely for ectatic disease so that they can receive treatment before they lose BCVA.
1. Godefrooij DA, de Wit GA, Uiterwaal CS, et al. Age-specific incidence and prevalence of keratoconus: a nationwide registration study. Am J Ophthalmol. 2017;175:169-172.
2. Torres Netto EA, Al-Otaibi WM, Hafezi NL, et al. Prevalence of keratoconus in paediatric patients in Riyadh, Saudi Arabia. Br J Ophthalmol. 2018;102(10):1436-1441.
3. Buzzonetti L, Bohringer D, Liskova P, Lang S, Valente P. Keratoconus in children: a literature review. Cornea. Published online July 2, 2020. doi: 10.1097/ICO.0000000000002420
4. Gordon-Shaag A, Millodot M, Shneor E, Liu Y. The genetic and environmental factors for keratoconus. Biomed Res Int. 2015;2015:795738.
5. Ahmad TR, Pasricha ND, Rose-Nussbaumer J, et al. Corneal collagen cross-linking under general anesthesia for pediatric patients with keratoconus and developmental delay. Cornea. 2020;39:546-551.
6. Barbisan PRT, Pinto RDP, Gusmao CG, et al. Corneal collagen cross-linking in young patients for progressive keratoconus. Cornea. 2020;39:186-191.
7. Shetty G, Behshad S. Corneal crosslinking outcomes in children with progressive keratoconus. Paper presented at: ASCRS Virtual Meeting; May 16-17, 2020.




