The treatment of presbyopia has been wide and varied. This article describes what is available and what may be coming down the pike.
HERE AND NOW
Monovision
Monovision with both contact lenses and IOLs is the most established strategy for addressing presbyopia. It is also the easiest and cheapest by far, provided that the patient accepts some loss of stereopsis as well as distance vision in one eye. The ideal amount of residual myopia for correction remains a subject of debate and can vary from -1.25 to -2.00 D.
Accommodating IOLs
The only approved accommodating IOL in the United States is the Crystalens (Bausch + Lomb), which provides good distance and intermediate vision in addition to variable amounts of near vision. Potential drawbacks to this technology include fibrosis (Z syndrome) and a more frequent need for refractive touchups relative to multifocal and monofocal IOLs. An advantage of this IOL is that it may be used for patients who have mild age-related macular degeneration, glaucoma, or a history of radial keratotomy or laser vision correction. A toric version of the lens (Trulign; Bausch + Lomb) is also available. (For more information on the Crystalens and Trulign IOLs, visit beye.com/1146 and beye.com/1135, respectively.)
Multifocal IOLs
The recently released low-add versions of the AcrySof IQ ReStor lenses (Alcon) and Tecnis Multifocal IOL (Abbott Medical Optics) provide patients with a better-accepted coverage of near, intermediate, and distance vision than the original high-powered models. Glare, halos, and problems related to contrast sensitivity are generally less noticeable if these lenses are not implanted in patients with high aberration values. Centration of the IOLs can be an issue, and hitting the refractive target of less than 0.75 D of cylinder is important for the highest quality of vision. Pupillary size can have a strong effect on both distance and near vision. (For more information on AcrySof IQ Restor and Tecnis Multifocal IOLs, visit bit.ly/dellCRST716.)
Corneal Inlays
The Kamra (AcuFocus) is the first FDA-approved corneal inlay. The procedure is performed in a deep corneal pocket, and centration is critical. The inlay provides good near vision with some compromise of distance vision in the nondominant implanted eye. Downsides of the technology include the expense of and need for access to a femtosecond laser that makes pockets and an instrument to help center the inlay. Contrast sensitivity is poor in the operated eye, but binocular contrast sensitivity can function well. Problems with fibrosis of the inlay and cosmesis can be corrected by removal of the inlay.1 (For more information on this corneal inlay, please see Dr. Rebenitsch’s article and visit beye.com/840.)
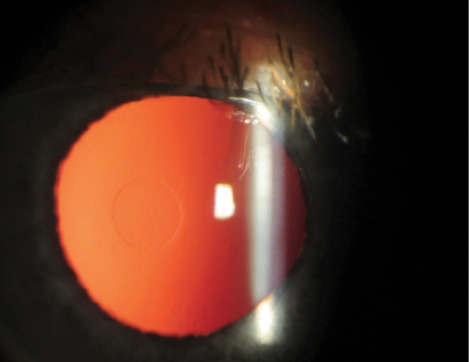
Figure 1. Retroillumination of an eye in which the Raindrop Near Vision Inlay has been implanted.
The FDA approved the Raindrop Near Vision Inlay (ReVision Optics; Figure 1) on June 29. The device is indicated to improve near vision in presbyopic patients who have emmetropic refractions (+1.00 to -0.50 D). This transparent hydrogel implant is 2 mm wide and 30 µm thick. It can be placed either under a LASIK flap or in a 150-µm corneal pocket. No special instrumentation is needed to place or center the inlay. Fibrosis has led to the removal of the device in a small percentage of cases, and an average loss of 1 line of distance vision in the treated nondominant eye is typical.2 (For more information on this implant, please see Dr. Chu’s article and visit beye.com/841.)
THE FUTURE
Topical Drops
Presbyopia Therapies is developing a pharmacological approach to presbyopic correction that expands depth of focus. PRX-100 provides miosis without stimulating accommodation. According to the company, the drops provide a pupillary size of 1.6 mm for about 8 hours. (For more information on this technology, visit bit.ly/dell716.)
Accommodating IOLs

Figure 2. The FineVision lens is a trifocal design.
The acrylic FluidVision lens (PowerVision) features anterior and posterior optics with a central cavity between them. The compressible haptics contain a silicone oil-based fluid. The two large haptics are connected to the central fluid cavity, and they work together with the eye’s natural processes to aid accommodation. When accommodation occurs, the zonules release tension on the capsule, compressing the haptics. This pushes the silicone fluid between the two optics and creates an accommodative effect. At present, a 4-mm incision is required for the IOL’s implantation, but future injectors may decrease the size needed. Alcon recently got involved with support of this technology. (For more information on this IOL, visit beye.com/1147.)
The Sapphire AutoFocal IOL (Elenza) features an electroactive liquid crystal optic encapsulated in an aspheric monofocal lens. The lens combines nanotechnology, artificial intelligence (neural networks-based memory), and advanced electronics to seamlessly and automatically focus an optic from far to near without movement. The IOL does not rely on precise contact with ciliary muscles to move and accommodate properly. Instead, it reportedly senses the pupil’s size and acts accordingly. The technology is still in development. (For more information on this lens, visit bit.ly/donnenfeld716.)
Trifocal IOLs
The FineVision (PhysIOL; Figure 2) and AT LISA Tri (Carl Zeiss Meditec) use the optical principle of constructive interference to capture and redistribute incoming light that would otherwise be lost to diffractive effects. More light can be directed to intermediate vision with this design than with a true multifocal lens. Trifocal IOLs also reportedly have fewer issues with glare and halos than a typical multifocal lens. (For more information on these IOLs, visit bit.ly/mertens716 and beye.com/1157.)
Extended Depth-of-Focus IOLs
Watch it Now
Howard Gimbel, MD, shares an edited-down version of the Kamra inlay procedure.
James Katz, MD, and Mark Kontos, MD, discuss scleral implant technology for presbyopic correction.
The Tecnis Symfony (Abbott Medical Optics) has rings but is not a true multifocal IOL, so patients will have fewer problems with glare and halos. This technology increases depth of focus with diffractive optics to expand the field of vision and decrease chromatic aberration. The compromise lies in fine near vision, but again, a real plus is that halos and glare can be comparable to that associated with monofocal lenses. (For more information on this IOL, visit bit.ly/dellCRST716.)
Adjustable IOLs
The Light Adjustable Lens (Calhoun Vision) has been available in Europe since 2008 but is in clinical studies in the United States. The lens is composed of macromers. Exposing these biocompatible photosensitive materials to a certain profile of ultraviolet light causes photopolymerization, which changes the shape and thus the power of the IOL. Light adjustment can induce asphericity centrally in the nondominant eye to provide an increased depth of field. This can reduce distance visual acuity in the treated eye. (For more information on this lens, visit beye.com/1158.)
Corneal Inlays
The Flexivue Microlens (Presbia) also has the CE Mark. Available with three add powers, the hydrophobic acrylic implant has a diameter of 3.2 mm, a central hole of 0.5 mm, and an edge that is 15 µm thick. This clear lens is placed in a corneal pocket and provides a refractive add power of between +1.50 and +3.50 D. (For more information on this device, please see Dr. Maloney’s article and visit beye.com/842.)
Scleral Expansion
VisAbility scleral implants (Refocus Group) are placed in the oblique quadrants of the sclera to strengthen the ciliary accommodative reaction. The procedure has become more automated in recent years, but no submission has yet been made to the FDA. (For more information, visit refocus-group.com.) /p>
1. FDA approves first-of-its-kind corneal implant to improve near vision in certain patients. US Food and Drug Administration website. April 17, 2015. http://1.usa.gov/28JWLs1. Accessed June 20, 2016.
2. Whitman J, Dougherty PJ, Parkhurst GD, et al. Treatment of presbyopia in emmetropes using a shape-changing corneal inlay: one-year clinical outcomes. Ophthalmology. 2016;123(3):466-475.
Jeffrey Whitman, MD
• president and chief surgeon, Key-Whitman Eye Center, Dallas
• (214) 754-0000
• financial disclosure: consultant to Alcon, Bausch + Lomb, Refocus Group, and ReVision Optics