Kamra
By R. Luke Rebenitsch, MD

The concept of corneal inlays is nothing new: José Barraquer, MD, has been credited with the original idea as early as the 1940s.1 The benefits of these implants are numerous—reversibility, ease of repositioning, and possible combination with previous and future refractive correction. Early designs were associated with difficulties such as vascularization, keratolysis, decentration, and poor biocompatibility.2-7 Only recently have technological advances overcome these concerns. Approved by the FDA in April, the Kamra (AcuFocus) is the first inlay available in the United States.8,9
AT A GLANCE
• According to Dr. Chu, emmetropic presbyopes may be some of cataract and refractive surgeons’ more challenging patients, because they never had vision problems before becoming presbyopic.
• Dr. Rebenitsch has found the Kamra to greatly improve presbyopic patients’ vision and to achieve high satisfaction in this age group. He typically recommends the inlay to patients under the age of 55 and refractive lens exchange to those who are older.
• The Kamra and Raindrop Near Vision Inlay are approved in the United States, and the Presbia Flexivue Microlens is in FDA clinical trials.
• The transparent Raindrop Near Vision Inlay changes the shape of the cornea by increasing its central curvature, creating greater depth of focus.
• The Presbia Flexivue Microlens can change the way the cornea refracts light without altering its anterior corneal shape. This clear implant causes a mild myopic shift, typically leaving the eye about 1.00 D myopic postoperatively, says Dr. Maloney.
Like most corneal inlays, the Kamra is implanted in the patient’s nondominant eye. This device is placed at least 200 µm from the corneal surface, where it blocks unfocused peripheral rays of light in order to increase depth of focus. The inlay is composed of polyvinylidine difluoride. It is 6 µm thick, has a diameter of 3.8 mm, and features a central annulus of 1.6 mm. There are 8,400 microperforations arranged in a pseudorandom pattern to allow nutrient flow.
VISUAL RESULTS
The US investigational device exemption trial was a 3-year, international, multicenter study consisting of 507 patients who were 45 to 60 years of age and had a manifest refraction spherical equivalent (MRSE) of -0.75 to +0.50 D.10 Subjects’ distance BCVA was at least 20/20 in both eyes. At 3 years, patients achieved an average improvement in their near vision of J8 to J2 while maintaining a distance UCVA averaging 20/20. Binocular contrast sensitivity and visual fields were clinically unaffected.
Further analysis of the study determined other important factors for future surgical planning and patient selection. First, patients with an MRSE of -0.75 D experienced the greatest improvement in near vision while maintaining uncompromised distance vision. Second, a 6 × 6 spot-line separation delivered the greatest improvement in near vision, no lost lines of distance BCVA, and the greatest refractive stability compared with larger spot-line separations. Finally, unlike with monovision, there was no change in stereopsis.11 It should also be noted that most diagnostic equipment could still be used after the Kamra’s implantation.
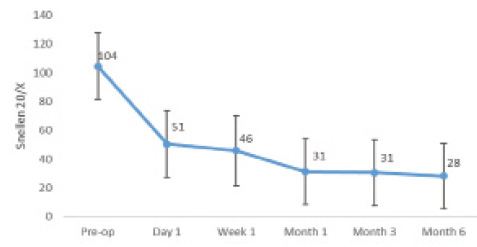
Figure. Monocular mean near UCVA (converted from LogMar) of the author’s first 22 patients.
Numerous studies in the United States and abroad have corroborated these results.12-15 In my experience with more than 100 Kamra inlays, I have had similar, if not better, results.
CONSIDERATIONS
Comparison to Other Presbyopic Solutions
The Kamra has been shown to improve near vision across the presbyopic age group, although those under the age of 50 experienced the greatest improvement.16 Results with the inlay and IOLs were comparable, but there were some advantages with the latter modality for certain intermediate and near demands.16 I now typically recommend the inlay to patients under the age of 55 and refractive lens exchange to those who are older than 55 years of age. There can be much overlap, depending on the measured scatter within a patient’s visual pathway as well as his or her preference.
Combination With Laser Vision Correction
The strong recommendation is to place the Kamra in a corneal pocket, but the device has been implanted under a LASIK flap as a combined procedure with equal or lesser success.12,16-18 The inlay’s placement in a deep pocket facilitates centration and causes less inflammation and remodeling because of the location and density of keratocytes.19
Given the improved visual results with an MRSE of -0.75 D in the nondominant eye and plano in the dominant eye, I perform laser vision correction (LVC) in nearly all patients receiving the Kamra. I target a plano result in the dominant eye and -0.75 to -1.00 D in the nondominant eye; then, I place the inlay in a pocket of approximately 250 µm. I recommend initially performing LVC and implantation of the inlay on different days.
Pupillary Size
The size of the pupil has been shown to have no effect on near vision and only a minimal effect on distance vision.20 This has been my experience as well.
Pseudophakia and Cataract
Although the Kamra can provide stable vision for years, lenticular opacities and cataracts will develop. I inform patients of this eventuality and explain that successful cataract surgery with monofocal IOL implantation can be performed while the inlay remains in place.21
Speed of Visual Recovery and Long-Term Stability
The speed of visual recovery after implantation of the Kamra is slower than after other forms of LVC. In my experience, most patients are satisfied with the procedure within 1 to 4 weeks, although their vision continues to improve for months (Figure).
Patients must be monitored closely during the postoperative period, because fluctuations—especially from ocular dryness—are normal. I counsel patients that it can take up to 6 months for full stability. I also inform them that hyperopic shifts, which are typically the result of inflammation and remodeling, can happen 3 to 6 months after the procedure. I find that a course of topical steroids typically resolves this problem.
CONCLUSION
The Kamra is a safe and effective treatment for presbyopia. Visual recovery is slower than after other forms of vision correction, and as with all refractive surgery, patient selection and education are important. I have found the inlay to greatly improve presbyopic patients’ vision and to achieve high satisfaction in this age group.
1. Barraquer JI. Modification of refraction by means of intracorneal inclusions. Int Ophthalmol Clin. 1966;6(1):53-78.
2. Deg JK, Binder PS. Histopathology and clinical behavior of polysulfone intracorneal implants in the baboon model. Polysulfone lens implants. Ophthalmology.1988;95:506-515.
3. Alió JL, Mulet ME, Zapata LF, et al. Intracorneal inlay complicated by intrastromal epithelial opacification. Arch Ophthalmol. 2004;122:1441-1446.
4. Choyce P. The present status of intracameral and intracorneal implants. Can J Ophthalmol. 1968;3:295-311.
5. Dohlman CH, Refojo MF, Rose J. Synthetic polymers in corneal surgery: glyceryl methacylate. Arch Ophthalmol. 1967;177:52-58.
6. Sweeney DF, Vannas A, Hughes TC, et al. Synthetic corneal inlays. Clin Exp Optom. 2008;91:56-66.
7. Laroche G, Marois Y, Guidoin R, et al. Polyvinylidene fluoride (PVDF) as a biomaterial: from polymeric raw material to monofilament vascular suture. J Biomed Mater Res.1995;29:1525-1536.
8. Santhiago MR, Barbosa FL, Agrawal V, et al. Short-term cell death and inflammation after intracorneal inlay implantation in rabbits. J Refract Surg. 2012;28(2):144-149.
9. Kamra Inlay – P120023. US Food and Drug Administration website. http://1.usa.gov/28J6zQH. Updated April 29, 2015. Accessed June 20, 2016.
10. Durrie D. The effect of different monovision contact lens powers on the visual function of emmetropic presbyopia patients (an American Ophthalmological Society Thesis). Trans Am Ophthalmol Soc. 2006;104:366-401.
11. Jalali S, Aus der Au W, Shaarawy T. AcuFocus corneal inlay to correct presbyopia using femto-LASIK. One year results of a prospective cohort study. Klin Monbl Augenheilkd. 2016;233(4):360-364.
12. Dexl AK, Jell G, Strohmaier C, et al. Long-term outcomes after monocular corneal inlay implantation for the surgical compensation of presbyopia. J Cataract Refract Surg. 2015;41(3):566-575.
13. Yilmaz OF, Alagoz N, Pekel G, et al. Intracorneal inlay to correct presbyopia: long-term results. J Cataract Refract Surg. 2011;37(7):1275-1281.
14. Waring GO 4th. Correction of presbyopia with a small aperture corneal inlay. J Refract Surg. 2011;27(11):842-845.
15. Tomita M, Waring GO 4th. One-year results of simultaneous laser in situ keratomileusis and small-aperture corneal inlay implantation for hyperopic presbyopia: comparison by age. J Cataract Refract Surg. 2015;41(1):152-161.
16. Vilupuru S, Lin L, Pepose JS. Comparison of contrast sensitivity and through focus in small-aperture inlay, accommodating intraocular lens, or multifocal intraocular lens subjects. Am J Ophthalmol. 2015;160(1):150-62.e1.
17. Tomita M, Kanamori T, Waring GO 4th, et al. Simultaneous corneal inlay implantation and laser in situ keratomileusis for presbyopia in patients with hyperopia, myopia, or emmetropia: six-month results. J Cataract Refract Surg. 2012;38(3):495-506.
18. Patel S, McLaren J, Hodge D, et al. Normal human keratocyte density and corneal thickness measurement by using confocal microscopy in vivo. Invest Ophthalmol Vis Sci. 2001;42(2):333-339.
19. Tomita M, Kanamori T, Waring GO 4th, et al. Retrospective evaluation of the influence of pupil size on visual acuity after Kamra inlay implantation. J Refract Surg. 2014;30(7):448-453.
20. Tan TE, Mehta JS. Cataract surgery following Kamra presbyopic implant. Clin Ophthalmol. 2013;7:1899-1903.
21. Juseynova T, Kanamori T, Waring GO 4th, et al. Outcomes of small aperture corneal inlay implantation in patients with pseudophakia. J Refract Surg. 2014;30(2):110-116.
Raindrop Near Vision Inlay
By Y. Ralph Chu, MD

Emmetropic presbyopes may be some of cataract and refractive surgeons’ more challenging patients, because they never had vision problems before becoming presbyopic. Compared with other candidates for refractive correction, these patients may need more counseling upfront about their options, and some studies suggest that this group of individuals has limited tolerance for monovision correction (somewhere around 1.50 D, which is less than needed for best near function).1 Corneal inlays can benefit these patients, and several of these devices are available in Europe. I have been a part of the investigator study group evaluating the Raindrop Near Vision Inlay (ReVision Optics), which received FDA approval on June 29. The implant is indicated to improve near vision in presbyopic patients who have emmetropic refractions (+1.00 to -0.50 D).
DETAILS
The Raindrop is the first and only inlay that changes the shape of the cornea by increasing its central curvature, creating greater depth of focus. The device is manufactured from a hydrogel material with a water content and refractive index similar to that of the cornea. The diameter of the implant measures 2 mm, and it has a thickness of 30 µm. The Raindrop Near Vision Inlay is implanted under a LASIK corneal flap created with a femtosecond laser at 30% of the patient’s central corneal thickness. Because the inlay is not refractive and is completely transparent, it does not obstruct diagnostic and surgical views of internal ocular structures.
Published data are now available from ReVision Optics’ pivotal investigational device exemption study, which enrolled 343 patients at 11 different sites. At the 12-month follow-up visit, the average near UCVA improved by 5 lines in the treated eye, with no loss of binocular distance vision; 93% of patients achieved a near UCVA of 20/25 or better. Intermediate UCVA improved by 2.5 lines, and distance UCVA decreased by 1.2 lines. The severity of visual symptoms was rated either absent or mild in 95.9% of patients, with blurred vision (1%), glare (2%), and halo (4%) the most common complaints.2,3 Patients’ satisfaction was high: 92% were somewhat, very, or completely satisfied with their inlay vision.
PERSONAL EXPERIENCE
As part of the US investigational device exemption study investigators’ group, I evaluated visual outcomes, topography, and refractive changes in my personal cohort of 25 patients who received the Raindrop Near Vision Inlay. At 18 months, I had outcomes for 23 patients. I evaluated their visual acuity with Early Treatment Diabetic Retinopathy Study (ETDRS) charts at near (40 cm), intermediate (80 cm), and distance (6 m). I used the iTrace (Tracey Technologies) to measure topographical and refractive changes along with manifest refraction. Figure 1 shows a typical topographic change after the device’s implantation—a central steepening at 18 months.
The largest increase was in central keratometry power, with a mean change of approximately 4.00 D from a preoperative value of 43.85 D to 47.81 D 18 months after surgery. Mean keratometry power also changed but to a lesser degree, from 43.79 D preoperatively to 45.52 D at 18 months, a steepening change of about 1.70 D.
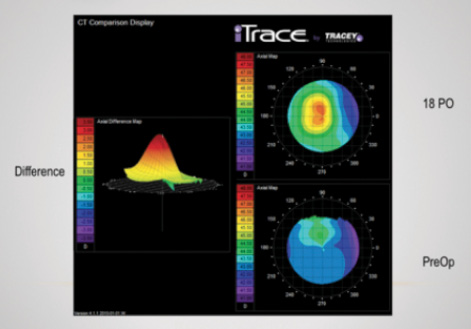
Figure 1. Topographic changes 18 months after implantation of the Raindrop Near Vision Inlay.
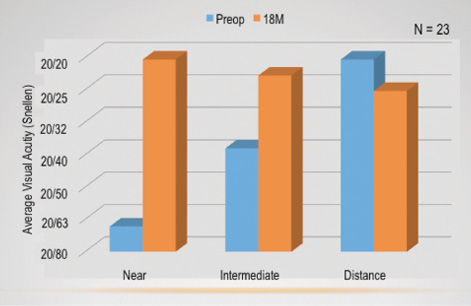
Figure 2. Average monocular visual acuity preoperatively and 18 months postoperatively.
In my subset of patients, there was a typical myopic shift of approximately 1.00 D (manifest refraction spherical equivalent) that stabilized over time. Preoperatively, mean manifest refraction spherical equivalent was +0.25 D, which shifted to -0.75 D at 6 months and stabilized out to 18 months.
I observed very little induced astigmatism in my patients; preoperatively, the mean cylinder was -0.25 D, and at 18 months, it was -0.30 D. Refractive cylinder remained stable after surgery.
On average, all of my patients saw about 20/20 at all distances binocularly. UCVA in the treated eye improved at near and intermediate distance and diminished slightly at distance.
Figure 2 shows the average monocular visual acuities from before surgery to 18 months postoperatively (when my patients saw about 20/20). Near and intermediate vision improved from preoperative levels, whereas distance vision decreased slightly. Figure 3 shows the average binocular visual acuities in this subset, and all patients saw approximately 20/20 across all three distances.
Patients completed an NEI RQL-42 quality-of-life (QOL) questionnaire preoperatively and 18 months postoperatively. The questions were measured on a scale of 0 to 100, with a higher score representing a better outcome. Although distance vision changed little from preoperatively to 18 months postoperatively, patients had increased scores in near vision, satisfaction with their correction, and satisfaction with their appearance. Figure 4 shows the mean QOL scores at the preoperative and 18-month time points.
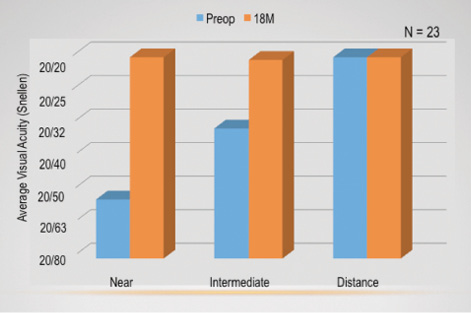
Figure 3. Average binocular visual acuity preoperatively and 18 months postoperatively.
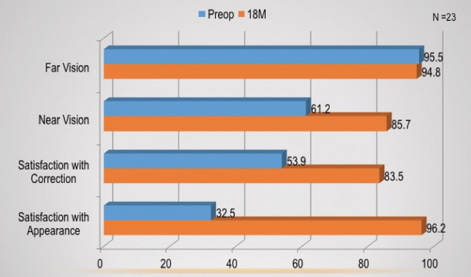
Figure 4. NEI RQL-42 QOL questionnaire mean values preoperatively and 18 months postoperatively.
WHAT THE FUTURE HOLDS
After receiving the Raindrop Near Vision Inlay, my patients exhibited good functional vision with average binocular acuities of approximately 20/20 at all measured distances. I am excited about the addition of this device to the options for treating presbyopia, because I think the inlay can provide great satisfaction to this growing and demanding group of patients.
1. Durrie DS. The effect of different monovision contact lens powers on the visual function of emmetropic presbyopic patients. Trans Am Ophthalmol Soc. 2006;104:366-401.
2. Whitman J, Dougherty PJ, Parkhurst GD, et al. Treatment of presbyopia in emmetropes using a shape-changing corneal inlay: one-year clinical outcomes. Ophthalmology. 2016;123:466-475.
3. Kilcoyne J. Treatment of presbyopia. Presented at: OIS @ AAO 2015; November 12, 2015; Las Vegas, NV. Available at: http://ois.net/ois-aao-2015/presentations. Accessed April 18, 2016.
Presbia Flexivue Microlens
By Robert K. Maloney, MD

The Presbia Flexivue Microlens (Presbia) is an intracorneal inlay designed to treat presbyopia by altering the refractive power of the cornea. This tiny multifocal lens, 3.2 mm in diameter, has a central 1.6-mm-diameter area focused for distance vision and a peripheral annulus for near vision, much like a multifocal contact lens (Figure 1). The implant is available in nine add powers that can be customized to the patient’s degree of presbyopia. Made of a hydrophilic acrylic polymer that has an index of refraction higher than that of the corneal stroma, the inlay can change the way the cornea refracts light without altering its anterior corneal shape. That feature allows the implant to be inserted into a corneal pocket made with a femtosecond laser in a simple, in-office procedure under topical anesthesia. The lens is crystal clear and invisible to the naked eye, a cosmetic advantage (Figure 2).
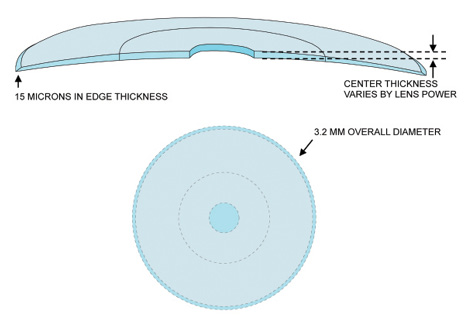
Figure 1. The Presbia Flexivue Microlens is 3.2 mm in diameter, with a central distance portion measuring 1.6 mm in diameter and an annular reading portion surrounding the distance zone.
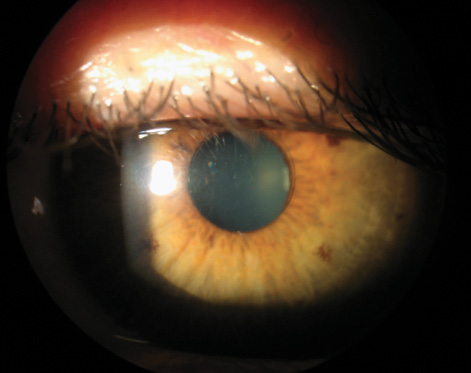
Figure 2. The implanted multifocal inlay is invisible to the naked eye and nearly invisible at the slit lamp.
CLINICAL PARADIGM
The Presbia Flexivue Microlens is currently in FDA trials, although it is already approved for use in 42 countries. The ideal candidate is emmetropic or slightly hyperopic and suffering from presbyopia. The microlens is implanted in the nondominant eye only, similar to other intracorneal inlays.
The Presbia Flexivue Microlens causes a mild myopic shift, typically leaving the eye about 1.00 D myopic postoperatively. This slight refractive difference between the eyes is well tolerated. Because of the multifocal effect, reading vision is generally much better than a patient could achieve with 1.00 D of monovision. Outside the United States, surgeons may combine LASIK with the inlay. LASIK is performed first to create an emmetropic refraction in both eyes, and then the inlay is inserted into a corneal pocket under the LASIK flap to restore near vision. The device can stay in place permanently, or it can be removed if, for example, presbyopia advances and a stronger lens prescription is required.
CLINICAL RESULTS
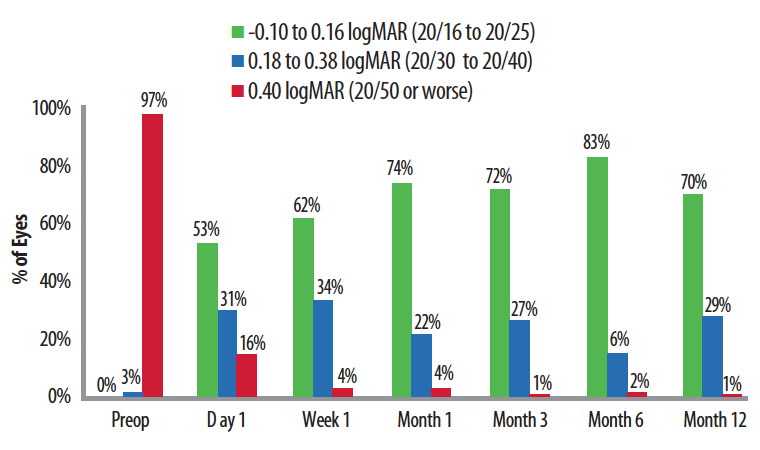
Figure 3. After the device’s implantation, approximately 75% of eyes achieved 20/25 or better near UCVA, equivalent to J1. Ninety-nine percent of eyes saw 20/40 or better at near.
Ioannis Pallikaris, MD, conducted the initial clinical trials in Europe, and Figure 3 summarizes his near vision results. In this study of 70 eyes, in each time interval, roughly three-quarters of eyes achieved logMAR vision of 20/25 or better; 20/25 near vision is equivalent to J1, consistent with excellent near acuity for almost all activities of daily living. Ninety-nine percent of eyes could see 20/40 or better at near, good enough for reading print of ordinary size such as on menus or a cell phone.
FDA CLINICAL TRIAL
The Presbia Flexivue Microlens is currently under investigation in the United States as part of a US investigational device exemption trial. Four hundred twenty-one patients have been enrolled in a 3-year clinical trial. All were close to emmetropic in both eyes and had the device inserted in their nondominant eye. Enrollment was completed in September 2015. The company anticipates the completion of follow-up in September 2018 and hopes to file the premarket approval application soon thereafter.
R. Luke Rebenitsch, MD
• refractive surgeon, ClearSight Center, Oklahoma City, Oklahoma
• (405) 733-2020; dr.luke@clearsight.com
• financial disclosure: has received speaking and educational honoraria from AcuFocus
Y. Ralph Chu, MD
• founder and medical director, Chu Vision Institute, Bloomington, Minnesota
• yrchu@chuvision.com
• financial disclosure: clinical investigator for ReVision Optics
Robert K. Maloney, MD
• director, Maloney Vision Institute, Los Angeles
• (310) 208-3937; info@maloneyvision.com
• financial disclosure: consultant to PresbiBio, a subsidiary of Presbia