Case Presentation
In 2001, a 71-year-old man had been experiencing a decrease in vision for the past few years and requested a consultation for management. The patient inquired whether he was a candidate for laser vision correction.
Four years before this patient’s presentation to our center, his BCVA was 20/30. One year before presentation, his BCVA was 20/40 with a manifest refraction of -7.50 +6.00 x 145º OD and 20/25 with a manifest refraction of -1.50 +2.00 x 048º OS. Early cataracts were detected, and keratoconus was diagnosed based on topography.
The patient grew increasingly unhappy with his inability to see and tried a gas permeable contact lens fitting in an effort to improve vision. Unfortunately, this effort was unsuccessful.
On presentation at our center, the patient reported nighttime glare that limited his ability to drive. He stated that his eyesight was deteriorating and that he could not see well enough to recognize familiar faces. Both eyes had 2+ nuclear sclerosis and dense cortical changes, and a posterior subcapsular cataract was evident in the right eye. His BCVA was 20/70 OD and 20/60 OS (Figures 1 and 2).
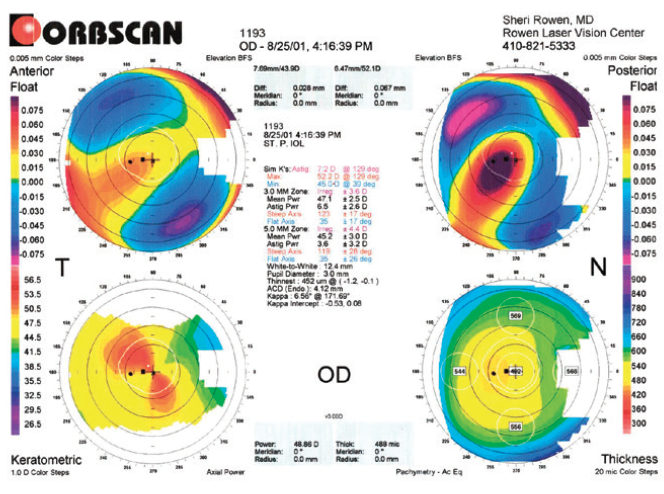
Figure 1. Topography of the right eye obtained in 2001.
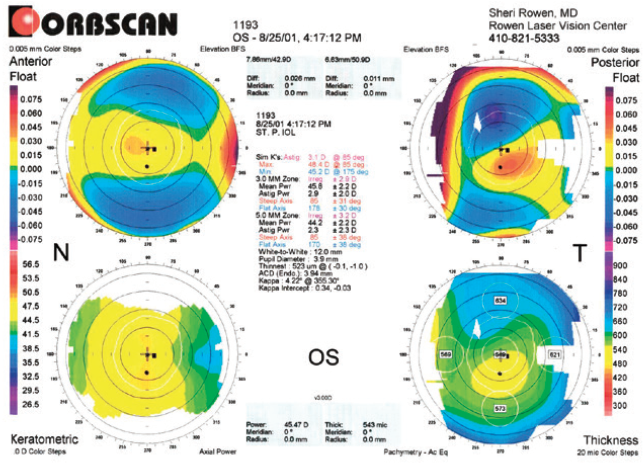
Figure 2. Topography of the left eye obtained in 2001.
Figures 1 and 2 courtesy of Sheri Rowen, MD, FACS
How would you proceed? How much of the vision loss is attributable to cataracts versus keratoconus? How reliable do you expect the A-scan to be? Which IOL would you select?
—Case prepared by Sheri Rowen, MD, FACS
WHAT I DID IN and my thoughts from 2001: SHERI ROWEN, MD, FACS
Cataract surgery was performed, and a STAAR Toric IOL (STAAR Surgical) was implanted in each eye to alleviate the excessive astigmatism caused by keratoconus. In the right eye, four A-scan measurements (Sonomed) revealed four completely different outcomes because of irregular keratometry (K) readings. Toric IOLs predicted to approach a target of emmetropia ranged in power from 13.50 D + 3.50 D to 16.00 D + 3.50 D. It was impossible to rely on any of the measurements because of the extreme variation.
I decided to insert a 16.00 +3.50 x 130º IOL in the right eye based on reasonably consistent axial length readings, even though the keratometry and axial length components were actually hypotheses. The K readings ranged from 43.47 D @ 37º to 50.50 D @ 131º, and axial length measurements ranged from 23.75 to 24.75 mm. In the left eye, the K readings ranged from 43.62 D @ 178º to 46.12 D @ 88º, and the axial length measurement was 23.81 mm. An 18.50 +3.50 x 88º toric IOL was inserted in the left eye.
The patient’s UCVA was 20/40 OD 1 day after surgery, and UCVA was 20/40 OS 1 day after surgery. The patient was able to read 20/30 OU without correction. His BCVA was 20/30-2 with a manifest refraction of -2.00 +2.00 x 125º OD and 20/25 with a manifest refraction of -2.75 +1.50 x 48º OS. The patient was ecstatic with the results.
I believe toric lens technology will allow ophthalmologists to treat patients who have complex keratoconus without any ill effects on the cornea.
This patient’s axis of astigmatism had remained the same for 10 years. Even if the diopters of cylinder increase, he will still benefit greatly from having at least 2.50 D corrected by the IOL. In this case, the improvement was more than 5.00 D, reducing the measured cylinder by more than 7.00 D to 2.00 D—an unexpected but positive result.
Toric IOL technology is slowly gaining popularity and, if used correctly, will be a great adjunct to all cataract and intraocular refractive surgery. The lens is always oriented along the steep axis of astigmatism (ie, the highest number using the plus cylinder). It is never based on the refraction of the patient because the lens can contribute to that number, skewing the effect of correcting corneal astigmatism only. It is difficult to ascertain exactly how 5.00 D of astigmatism was eliminated in this patient’s right eye, but the crystalline lens must have contributed to his extreme astigmatism, which meant that the preoperative K readings might not have been precise. I am truly overwhelmed by these results in a patient with keratoconus.
WHAT I WOULD DO IN 2021
Sheri Rowen, MD, FACS

It is quite interesting to revisit a case from 20 years ago and to think about the technologies that were available then. There were far fewer options for vision correction. In 2001, the STAAR Toric IOL was the only available toric lens. The AcrySof Toric IOL (Alcon) was not introduced until 2005, and general acceptance of toric IOLs has remained slow. The few of us surgeons who implanted toric lenses in the early 2000s did so altruistically for patients, despite the extra work involved, because we recognized the visual benefits.
I began using STAAR lenses in 1992 and STAAR Toric lenses in 1998. The Orbscan (Bausch + Lomb) was introduced in 1995 and was considered state of the art for topography at the time of this case. The IOLMaster (Carl Zeiss Meditec) was introduced in 2000, so I must have had access only to Sonomed A-scan technology just before purchasing my first IOLMaster.
This patient’s large amount of astigmatism required correction. After reviewing the topography and large span of measurements, I remain pleased with the outcome of surgery. The axis appears to be reasonably regular and well identified centrally, so I would still place a toric IOL, albeit a one-piece acrylic model, on the steep axis.
In current practice, it is worth considering CXL whenever a patient presents with keratoconus. Because the cornea tends to be stable by the time a person reaches 71 years of age, I would strive to obtain accurate preoperative measurements, perform cataract surgery, and then monitor this patient for changes. If corneal changes were to be observed, CXL would be indicated. I would perform CXL before cataract surgery if this patient were younger. Intrastromal corneal ring segment implantation can also be considered to offset corneal changes and normalize the shape of the cornea.
The topographic measurements that could be obtained today would obviously be more informative. Currently available acrylic one-piece toric IOLs are more rotationally stable and can correct up to 4.00 D of astigmatism in the United States and more outside the United States. More advanced topography and anterior OCT scans, including epithelial thickness maps, would be obtained; numerous technologies are available for these purposes. The Atlas corneal topography system (Carl Zeiss Meditec) could help to obtain better central K readings at 0 to 3 mm. The IOLMaster, Lenstar (Haag-Streit), or Argos (Alcon) would be used for biometry measurements. Intraoperative aberrometry with the ORA system (Alcon) would be performed; if this instrument were able to capture data in this eye, the information could be used to obtain more accurate measurements of sphere, cylinder, and toric IOL orientation. I avoid making arcuate corneal incisions in keratoconic eyes. Without access to the original measurements, I cannot comment on the spherical powers of the IOLs I chose, but I certainly attempted to avoid a hyperopic outcome.
To optimize results, astigmatismfix.com would be consulted to determine if a rotational adjustment of either IOL is necessary postoperatively. This site was developed by John P. Berdahl, MD, and David R. Hardten, MD, FACS. If a refractive surprise were encountered after cataract surgery on the first eye, I would not hesitate to perform an IOL exchange. Because the STAAR Toric IOL corrected only 2.30 D of astigmatism, perhaps the original corneal measurements were not as accurate as they appeared, which could explain the excellent outcome achieved in the right eye. The measurements in the left eye clearly were slightly off as well, but perhaps rotation of the IOL could have improved the outcome. The patient’s postoperative UCVA was better than his visual acuity had ever been with spectacles, so I guess not too many changes are required other than the use of a more rotationally stable toric IOL that corrects a greater amount of cylinder!
Sumitra S. Khandelwal, MD

Patients who present for laser vision correction or cataract surgery often do not realize that irregular astigmatism is a cause of their vision issues. This patient is an excellent example, especially because topography is not routinely used to screen patients with decreased vision outside of laser vision correction and cataract surgery evaluations.
My first step would be to determine whether this patient has progressive keratoconus. If he does, I would perform CXL first. Determining if keratoconus is progressing can be challenging. The patient’s vision and refraction have been shifting, but is keratoconus or the cataract the cause? Prior topographic measurements for this case are not available; it is therefore impossible to be certain if keratoconus is progressing. Based on the patient’s age, I would not perform CXL but would counsel him that the procedure may become necessary in the future if disease progression is documented.
Next, I would determine if his vision issues are due to keratoconus or cataracts. The significant change in lens opacity points to the cataracts. If there is a question, say in a younger patient or someone with minimal lens opacity, I refer the individual for a rigid gas permeable or scleral lens overrefraction. In a few similar instances I have encountered, the problem was entirely corneal in nature, and correction with contact lenses provided excellent vision.
I would determine the goals of cataract surgery here. This patient does not wear contact lenses. I would counsel him that IOL options are limited owing to his irregular astigmatism. I would not choose a multifocal lens. A toric lens, however, could be a good option. When I look at the central 3 mm on topography, the astigmatism is fairly regular. I would obtain multiple corneal measurements (eg, one biometer and two topographers or two biometers and one topographer) to see if they align. That information plus habitual wear can help confirm the best axis and magnitude of astigmatism to correct. Higher-powered toric lenses are available today than were available in 2001 and would be excellent options here. I like to use the 3 mm on the Placido topography, which was not given on the Orbscan. If symmetric astigmatism is present in the central 3 mm of the cornea, I would use a toric IOL. Residual myopic astigmatism in some patients can provide a small amount of extended depth of focus, so the goal would be to undercorrect rather than overcorrect both the astigmatism and myopia. I also favor zero-aberration aspheric lenses for patients similar to this one.
Spherical power selection is important in this situation. Biometry is the standard for these cases. Mild myopia would be the target in the left eye because the K values are fairly normal. For the right eye, however, I would consider targeting as much as -1.50 D of myopia. This is based on studies showing that the steeper the cornea, the more hyperopic the outcome in patients with keratoconus.1 Additionally, I would use a modern IOL power calculation formula that has been shown to have the least residual refractive error such as the Barrett,2 the SRK/T, or the Kane keratoconus formula.3
Spectacles may be used to correct residual error, although it is fascinating how well some patients with similar presentations can still see at near. Retaining some with-the-rule myopic astigmatism is often key to these excellent outcomes.
Abi Tenen, MBBS(Hons), FRANZCO

A major change since 2001 in terms of managing this case is that CXL has been established as a therapy to stabilize keratoconus. Even though he is 71 years old, this patient’s decreasing vision may be due in part to progressive corneal changes, so I would monitor him for a period of time and perform CXL before cataract surgery if corneal instability is observed. CXL may also discourage corneal shape shifting after the mechanical trauma of creating corneal incisions during cataract surgery.
It is standard for me to obtain imaging with the Pentacam (Oculus Optikgeräte), use the Barrett formula for IOL power calculations, and perform laser cataract surgery. The Kane formula, however, could be useful for the IOL power calculation for this patient’s right eye because of the steep K readings. I make a point of looking at the shape of the cone through the visual axis zone/central 3 mm. If the cone is particularly irregular, I may select a nontoric lens, but for this patient, I would prefer to undercorrect the cylinder rather than not to correct it. My preference for the right eye would be to implant an AcrySof IQ Toric IOL (Alcon), probably with half the toric value that the IOL formulas suggest. In contrast, the left eye could receive an AcrySof IQ Toric IOL with a low toric power or possibly an AcrySof IQ Vivity IOL (Alcon) because the cornea has undergone only relatively mild changes.
William B. Trattler, MD

This patient has keratoconus in the right eye with a steep cornea, a skewed radial axis, and an abnormal posterior float. The left eye is suspicious for keratoconus because of an abnormal posterior float and an inferiorly decentered thinnest location of the cornea. As noted in the presentation, he needs cataract surgery.
One question that typically arises is whether CXL should be performed before cataract surgery in patients with keratoconus. I typically recommend performing cataract surgery first in patients with documented nonprogressive keratoconus who have visually significant cataracts. Cataract surgery can significantly improve their vision and quality of life. Postoperatively, they can be monitored with annual topography or tomography. If keratoconus progresses, they can then undergo CXL. If, however, patients have progressive keratoconus and cataracts that are not visually significant or if they have developed ectasia after LASIK, I discuss performing CXL as the first step.
In this case, I would recommend cataract surgery first. The good news for this patient is that the axis of astigmatism in each eye appears to be symmetrical. He does not have significant irregular astigmatism. If his topography or tomography measurements and K values line up, he is likely eligible for a monofocal toric IOL. I use a femtosecond laser (Lensar Laser System, Lensar) that incorporates preoperative topography and uses iris registration to mark the axis of astigmatism; this allows me to easily align the toric IOL intraoperatively and to confirm its orientation during the postoperative period (Figure 3).

Figure 3. Capsular nubs created with the Lensar Laser System using preoperative topography and iris registration to help with the intraoperative alignment of a toric IOL.
Courtesy of William B. Trattler, MD
Cataract surgery with a monofocal toric IOL should greatly improve this patient’s vision. Compared to Dr. Rowen’s plan 20 years ago, the main differences are that monofocal toric IOLs are available that can correct higher levels of astigmatism and that current technology allows more accurate intraoperative IOL alignment. Additionally, monofocal toric IOLs are less likely to rotate postoperatively than the plate-haptic toric IOLs used in the original case.
1. Savini G, Abbate R, Hoffer KJet al. Intraocular lens power calculation in eyes with keratoconus. J Cataract Refract Surg. 2019;45(5):576-581.
2. Wang KM, Jun AS, Ladas JG, Siddiqui AA, Woreta F, Srikumaran D. Accuracy of intraocular lens formulas in eyes with keratoconus. Am J Ophthalmol. 2020;212:26-33.
3. Kane JX, Connell B, Yip H, et al. Accuracy of intraocular lens power formulas modified for patients with keratoconus. Ophthalmology. 2020;127(8):1037-1042.




