

Dry eye disease (DED) is a common ocular condition with a reported prevalence ranging from 7.4% to 33.7%.1 DED can be grossly organized into two functional categories, aqueous-deficient and evaporative. To be effective, treatment should be aimed at the underlying etiology. Aqueous-deficient DED occurs when tear production is insufficient, whereas evaporative DED is due to instability of the tear film resulting from meibomian gland dysfunction (MGD), which can lead to abnormal lipid production.
Approximately 86% of patients diagnosed with DED have contributing MGD.2 For some of these individuals, conservative treatment such as the administration of lubricating drops and ointments, low-dose antiinflammatory topical medications, and warm compresses is insufficient to control the signs and symptoms of DED. In response, industry has developed a number of in-office technologies aimed at providing long-term relief from chronic DED. The use of these technologies often requires a significant upfront equipment commitment, and many of the procedures are not covered currently by Medicare or Medicaid.
This article discusses some of these treatment modalities and their use for managing MGD-related DED.
THERMAL TREATMENT
LipiFlow Thermal Pulsation System. LipiFlow (Johnson & Johnson Vision) is used to perform a 12-minute in-office procedure indicated for the treatment of MGD. A single-use, disposable eyepiece applies heat along the palpebral surface of the eyelids to liquify meibum while simultaneously delivering mechanical pulsation to the meibomian glands to express their contents. Patients who underwent one LipiFlow treatment showed a greater improvement in Ocular Surface Disease Index (OSDI) scores and meibomian gland secretion at 3 and 12 months than individuals treated conservatively with warm compresses and eyelid hygiene.3,4
iLux. Similar to the LipiFlow device, the iLux (Alcon) is used for an in-office procedure that delivers a combination of heat and mechanical pulsation to express meibum. The device heats the eyelids with light-emitting diodes via disposable silicone pads. A randomized controlled trial found an improvement in meibomian gland secretion, OSDI scores, and tear breakup time in patient groups treated with either the iLux or LipiFlow. No statistically significant differences between the two groups were observed.5
TearCare system. The TearCare system (Sight Sciences) also uses thermal energy to improve meibum flow. Heat is applied to the eyelids via a single-use customizable and flexible silicone eyepiece that is designed to fit different lid shapes. Unlike the LipiFlow and iLux, which deliver mechanical pulsation, the TearCare system requires manual expression of the meibomian glands after treatment.6
Intense pulsed light. Intense pulsed light (IPL) therapy uses thermal energy to coagulate blood vessels. IPL is widely used in dermatology to treat vascular lesions, acne, and rosacea. Current devices use broad-spectrum polychromatic light with wavelengths ranging from 500 to 1,200 nm. Thermal energy is selectively absorbed by intradermal melanin and hemoglobin, leading to coagulation.
Patients with rosacea who underwent IPL reported an improvement in dry eye symptoms, prompting research into the use of IPL as an ocular treatment. Vora et al reported reduced OSDI scores, decreased lid margin edema and vascularity, and increased meibum quality scores after IPL treatment.7 DED signs and symptoms are likely improved by IPL through an inhibition of inflammatory mediators at the eyelid margin vessels and in the tear film.8
An improvement in the morphology and functionality of the meibomian glands9 and a significant increase in tear breakup time10 were found after IPL treatment using the OptiLight (Lumenis). The user enters the patient’s skin type into the device, and the software selects the appropriate treatment parameters.
Similarly, the dry eye mode on the Quadra Q4 Platinum Series Intense Pulsed Light Treatment System (DermaMed) allows treatment to be customized according to the patient’s Fitzpatrick skin type and pain tolerance. During IPL in general and with the DermaMed device in particular, patients wear opaque goggles to prevent damage to intraocular melanin-containing tissues. Preselected IPL settings are used to treat the lower eyelid and canthus areas. The procedure generally takes 5 minutes. During IPL, 10 to 15 spots are placed along the lower eyelid. The superior eyelid is avoided because of a risk of damage to intraocular structures. IPL is performed every 4 to 6 weeks for a total of four sessions, followed by maintenance therapy as needed. Caution is warranted in patients with a Fitzpatrick skin type greater than 4 given the risk of permanent depigmentation.7
ATHERMAL TREATMENT
Another option for MGD treatment is low-level light therapy (LLLT), which may be performed in a standalone fashion with the Equinox (Espansione Group) or in combination with IPL using the Epi-C Plus (Espansione Group).11
During LLLT, a mask is applied for 15 minutes. Photobiomodulation of the meibomian glands triggers endogenous heating of both eyelids by activating the cellular production of adenosine triphosphate. The meibomian glands can be expressed after treatment. In a prospective trial of LLLT combined with IPL, the average OSDI score decreased, and the ability to express the meibomian glands improved after treatment.11
IMAGING
The LipiView II Ocular Surface Interferometer and LipiScan Dynamic Meibomian Imager (both from Johnson & Johnson Vision) are noninvasive imaging devices designed to identify appropriate candidates for treatment with the LipiFlow system or similar devices.
LipiView uses white light interferometry to measure the lipid layer thickness of the tear film. Blackie et al found that 74% of patients with severe DED had a lipid layer thickness of less than 60 nm, whereas 72% of patients without DED had a lipid layer thickness that was greater than 75 nm.12 Finis et al found lower lipid layer thickness to be associated with a decreased ability to express the meibomian glands, suggesting that lipid layer thickness can be used as a screening tool for MGD.13
LipiScan noninvasively assesses meibomian gland structure; it uses dynamic illumination to minimize reflections and adaptive transillumination to provide equal lighting across the surface of the eyelids. Normal acini appear as discrete linear groups of light gray circular structures, whereas meibomian gland atrophy is seen as a darker gray absence of those structures. Serial images can be used to track the progress of therapy and improve patient compliance. Once meibomian glands atrophy, their function decreases and their ability to regenerate is lost. Widespread destruction of glands seen on LipiScan images could therefore identify patients who may be less responsive to LipiFlow treatment (Figures 1–4).14
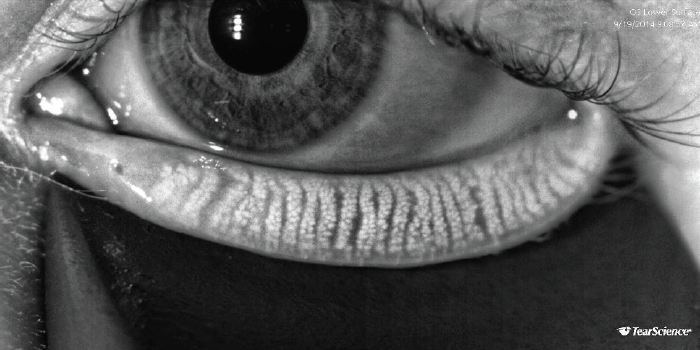
Figure 1. Dynamic illumination with the LipiScan shows normal meibomian gland acini without dropout.
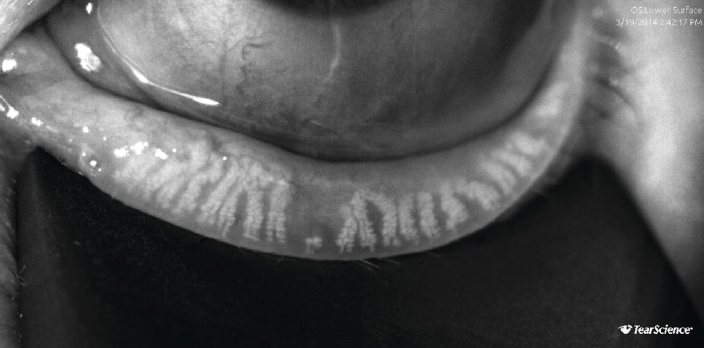
Figure 2. LipiScan image showing meibomian gland atrophy and a loss of acini.
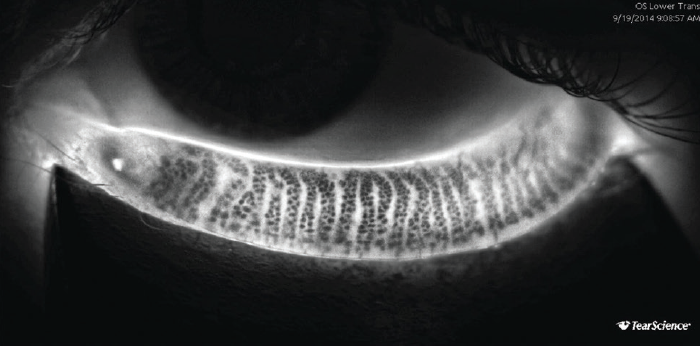
Figure 3. Adaptive transillumination with the LipiScan shows normal meibomian gland structure without dropout.
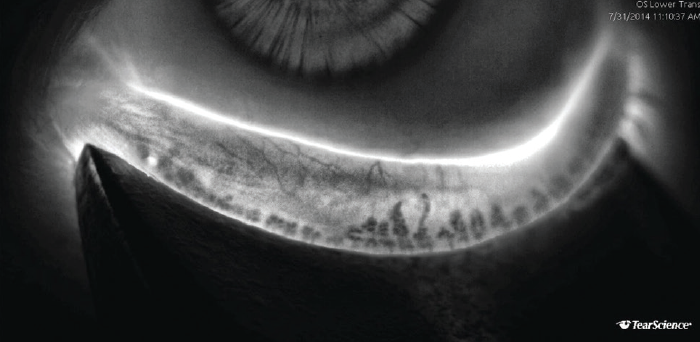
Figure 4. Adaptive transillumination with the LipiScan shows severe meibomian gland atrophy that may render treatment with the LipiFlow device less effective than it would be in a patient with less meibomian gland dropout.
All images courtesy of Johnson & Johnson Vision
DEMODEX BLEPHARITIS
Demodex folliculorum is a commensalistic mite found on cilia. In one epidemiologic study, these mites were isolated from the eyelashes of 28.7% of patients who had been diagnosed with blepharitis.15 The presence of Demodex can cause low-grade inflammation of the eyelids and block the meibomian glands. Conservative treatment consists of lid scrubs with tea tree oil, but this approach is not always effective.16
BlephEx (BlephEx) is a handheld instrument incorporating a disposable microbrush that spins rapidly to mechanically debride Demodex cuffs and scurf to provide open outlets for meibomian gland secretions. Treatment lasts several minutes, and patients experience a tickling sensation but no overt pain. In a randomized controlled interventional treatment study, BlephEx treatment improved OSDI scores and reduced the quantity of Demodex folliculorum compared to therapy with lid scrubs or tea tree oil, but the difference in efficacy did not reach statistical significance.17
Small studies have suggested that IPL18 and LLLT19 can also decrease the population of Demodex mites.
1. The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007;5(2):93-107.
2. Lemp MA, Crews LA, Bron AJ, Foulks GN, Sullivan BD. Distribution of aqueous-deficient and evaporative dry eye in a clinic-based patient cohort: a retrospective study. Cornea. 2012;31(5):472-478.
3. Blackie CA, Coleman CA, Holland EJ. The sustained effect (12 months) of a single-dose vectored thermal pulsation procedure for meibomian gland dysfunction and evaporative dry eye. Clin Ophthalmol. 2016;10:1385-1396.
4. Greiner JV. Long-term (12-month) improvement in meibomian gland function and reduced dry eye symptoms with a single thermal pulsation treatment. Clin Exp Ophthalmol. 2013;41(6):524-530.
5. Tauber J, Owen J, Bloomenstein M, Hovanesian J, Bullimore MA. Comparison of the iLux and the LipiFlow for the treatment of meibomian gland dysfunction and symptoms: a randomized clinical trial. Clin Ophthalmol. 2020;14:405-418.
6. Karpecki P, Wirta D, Osmanovic S, Dhamdhere K. A prospective, post-market, multicenter trial (CHEETAH) suggested TearCare system as a safe and effective blink-assisted eyelid device for the treatment of dry eye disease. Clin Ophthalmol. 2020;14:4551-4559.
7. Vora GK, Gupta PK. Intense pulsed light therapy for the treatment of evaporative dry eye disease. Curr Opin Ophthalmol. 2015;26(4):314-318.
8. Dell SJ. Intense pulsed light for evaporative dry eye disease. Clin Ophthalmol. 2017;11:1167-1173.
9. Yin Y, Liu N, Gong L, Song N. Changes in the meibomian gland after exposure to intense pulsed light in meibomian gland dysfunction (MGD) patients. Curr Eye Res. 2018;43(3):308-313.
10. Dell SJ, Gaster RN, Barbarino SC, Cunningham DN. Prospective evaluation of intense pulsed light and meibomian gland expression efficacy on relieving signs and symptoms of dry eye disease due to meibomian gland dysfunction. Clin Ophthalmol. 2017;11:817-827.
11. Stonecipher K, Abell TG, Chotiner B, Chotiner E, Potvin R. Combined low level light therapy and intense pulsed light therapy for the treatment of meibomian gland dysfunction. Clin Ophthalmol. 2019;13:993-999
12. Blackie CA, Solomon JD, Scaffidi RC, et al. The relationship between dry eye symptoms and lipid layer thickness. Cornea. 2009;28(7):789-794.
13. Finis D, Pischel N, Schrader S, Geerling G. Evaluation of lipid layer thickness measurement of the tear film as a diagnostic tool for meibomian gland dysfunction. Cornea. 2013;32(12):1549-1553.
14. Hura AS, Epitropoulos AT, Czyz CN, Rosenberg ED. Visible meibomian gland structure increases after vectored thermal pulsation treatment in dry eye disease patients with meibomian gland dysfunction. Clin Ophthalmol. 2020;14:4287-4296.
15. Kemal M, Sümer Z, Toker MI, Erdoğan H, Topalkara A, Akbulut M. The prevalence of Demodex folliculorum in blepharitis patients and the normal population. Ophthalmic Epidemiol. 2005;12(4):287-290.
16. Gao YY, Di Pascuale MA, Elizondo A, Tseng SC. Clinical treatment of ocular demodecosis by lid scrub with tea tree oil. Cornea. 2007;26(2):136-143.
17. Murphy O, O’Dwyer V, Lloyd-McKernan A. The efficacy of tea tree face wash, 1, 2-Octanediol and microblepharoexfoliation in treating Demodex folliculorum blepharitis. Cont Lens Anterior Eye. 2018;41(1):77-82.
18. Prieto VG, Sadick NS, Lloreta J, Nicholson J, Shea CR. Effects of intense pulsed light on sun-damaged human skin, routine, and ultrastructural analysis. Lasers Surg Med. 2002;30(2):82-85.
19. Pult H. Low level light in the treatment of meibomian gland dysfunction. Invest Ophthalmol Vis Sci. 2020;61(7):99.




