CASE PRESENTATION
A 52-year-old man has a history of a traumatic cataract in his left eye. His right eye is normal upon examination. The patient’s visual acuity measures 20/20 OD with a minimal refraction. The visual acuity of his left eye is hand motions, and there is no improvement with refraction.
The slit-lamp examination reveals normal anterior and posterior segments in the patient’s right eye. The left eye has a clear cornea, a quiet anterior chamber, and a dense 4+ brunescent cataract with questionable phacodonesis (Figure). There is no view of the posterior segment. The B-scan ultrasound is normal.
The patient desires spectacle independence. How would you prepare for cataract surgery, and how would you counsel this patient? Which IOL(s) would you choose?
—Case prepared by Audrey R. Talley Rostov, MD.
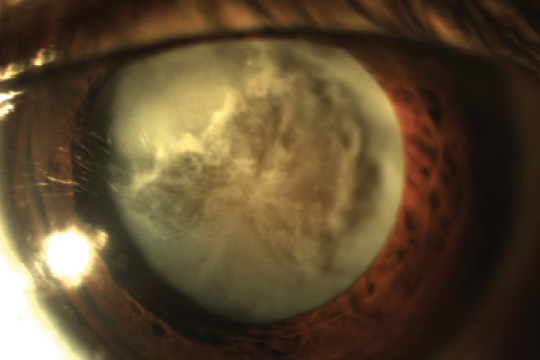
Figure. Traumatic cataract.

JOSEPH GIRA, MD
With the patient’s UCVA so good in the contralateral eye, he would be a great candidate for any of the presbyopia-correcting lenses. Depending on the degree of phacodonesis and whether a Crystalens (Bausch + Lomb) is placed, a capsular tension ring may be inserted. A low-add multifocal IOL is another option that could provide excellent spectacle independence. Discussing with the patient his goals and lifestyle (hobbies, work, etc.) would determine which technology would suit him best.
In regard to the surgery, I would stain the capsule with trypan blue dye to assist visualization. I would use a dispersive ophthalmic viscosurgical device (OVD) to prevent the capsule from trailing outward during formation of the capsulotomy. With excellent pupillary dilation, a femtosecond laser could easily be used to make a perfect capsulotomy.
As with any traumatic case, it would be important to ensure that the patient has realistic expectations preoperatively. He should do well with surgery but needs to understand that circumstances may complicate the procedure or even prevent the placement of a presbyopia-correcting lens.

ANITA NEVYAS-WALLACE, MD
Although at least 180º of zonular fibers are damaged or absent and the nasal/inferonasal crystalline lens is tilting posteriorly, there is no obvious vitreous in the anterior chamber. I would therefore attempt to keep the hyaloid face intact by filling the anterior chamber with air, then a dispersive OVD; I would inject the OVD first over the area of missing zonular fibers for tamponade. Next, I would carefully “paint” the anterior lens capsule with trypan blue dye without allowing it to go posteriorly, because it would stain the posterior capsule, thereby diminishing the possibility of any red reflex.
To decompress the possibly intumescent lens, I would make the initial puncture into the anterior capsule with a 25-gauge needle on a 3-mL syringe half filled with balanced salt solution, immediately perform decompression, and then add more dispersive OVD.
I would start the capsulorhexis superotemporally so that initial tearing was toward, rather than away from, the area of zonular dehiscence. Once the tear reached about 11 o’clock, I would start placing iris hooks or capsule retractors (MicroSurgical Technology) to support the capsule so that creating the capsulorhexis did not further disinsert the zonule. Scarred areas of the anterior capsule that resisted tearing could be cut with scissors. I would center the capsulorhexis on the bag, not the pupil, and make it small enough to allow optic capture. Although a laser might make creating the capsulotomy easier, it would increase the risk of radial tears, particularly when capsule retractors or iris hooks were used to support the bag (I. Ahmed, written communication, January 2016).1-4
I would proceed with gentle hydrodissection and frequently decompress the bag without overly decompressing the anterior chamber. Aspirating cortical material might help to mobilize the nucleus; I would avoid forcible rotation. So as not to stress the zonule, I would use a vertical chop phaco technique. I would frequently re-inject a dispersive OVD to tamponade the zonular dehiscence and to refill the capsular bag.
Once there was room, I would insert an Ahmed Capsular Tension Segment (Morcher; distributed in the United States by FCI Ophthalmics). I would thread the eyelet (before insertion) with a 7–0 Gore-Tex suture (W.L. Gore & Associates; off-label usage), which would be brought out temporarily through a nasal sideport incision and might later be used for securing the bag to the sulcus.
After removing cortical material via repeated viscodissection and manual aspiration using a 25-gauge cannula, I would refill the bag with a cohesive OVD. I would insert a capsular tension ring (CTR). To avoid placing stress on the zonule, I would guide the leading loop with a second instrument: the tip of a Fenzl hook, “trigger” aimed skyward, engages the CTR’s leading loop to avoid contact between the CTR and bag until much of the ring has been injected. When the leading loop is released, the CTR reaches the bag’s equator. I would not release the inserter from the trailing loop of the CTR until I had rotated the ring so that the loops were in a temporal position, where the zonule is less damaged.
I would recommend a mildly myopic target (about -1.25 to -1.50 D) to the patient so as to reduce his spectacle dependence, given that the uninjured eye sees 20/20 with a minimal refraction. I would avoid IOLs that are sensitive to decentration, such as toric and multifocal lenses, or IOLs that require a healthy zonule in order to function such as the Crystalens. I would have available a three-piece IOL, which offers the option of bag or sulcus placement with or without optic capture; the EC-3 IOL (Aaren Scientific) also offers the option of intrascleral haptic gluing. Whichever IOL I inserted, I would instill triamcinolone acetonide after OVD removal to verify that no vitreous was present in the anterior chamber. Residual refractive error could be addressed later with astigmatic keratotomy or laser vision correction.

P. DEE G. STEPHENSON, MD
Trauma to the eye is associated with cataractous changes in the crystalline lens. Direct injury to the lens can occur with blunt trauma or with penetrating injuries of the globe. Management can be complicated by the presence of things like zonular dialysis; a posterior capsular tear; trauma to the iris, angle, or cornea; or just poor visualization. Unfortunately, the eye can look and behave much differently in the OR than the examination room. The preoperative discussion with this patient needs to be open and honest, and no promises like spectacle independence should be made.
There are two ways to handle the cataract extraction, either with a laser or manually. The optical coherence tomography on some laser platforms (eg, the Lensar Laser System with Streamline [Lensar]) can identify the anatomy and provide information on what is and is not normal. A laser can make a perfect capsulotomy without exerting undue stress on the zonules, and with the new patterns for fragmentation based on nuclear density, the lens can be fragmented with highly controlled and directed power. If the surgeon prefers a manual approach, the checkpoint should be the easiness of the capsulorhexis; converting to extracapsular cataract extraction would not be the end of the world.
I would place a CTR whether or not cataract surgery included the use of a laser. I would choose an aspheric monofocal IOL like the Sofport LI61A0 (Bausch + Lomb). This aspheric, fourth-generation silicone lens carries equal power from its center to its edge, so mild dislocation or tilt will not affect the power or quality of vision. Moreover, this IOL can be placed in the sulcus or with optic capture without a change in power.
A key to success in this case is to underpromise and overdeliver.
1. Abell RG, Darian-Smith E, Kan JB, et al. Femtosecond laser-assisted cataract surgery versus standard phacoemulsification cataract surgery: outcomes and safety in more than 4000 cases at a single center. J Cataract Refract Surg. 2015;41(1):47-52.
2. Yeoh R. Femtosecond laser-assisted capsulorhexis: anterior capsule tags and tears. J Cataract Refract Surg. 2015;41(4):906-907.
3. Oakley CL, Vote BJ, Abell RG, et al. Reply. J Cataract Refract Surg. 2015;41(4):907-908.
4. Abell RG, Davies PE, Phelan D, et al. Anterior capsulotomy integrity after femtosecond laser-assisted cataract surgery. Ophthalmology. 2014;121(1):17-24.
Section Editor Lisa Brothers Arbisser, MD
• emeritus position at Eye Surgeons Associates, the Iowa and Illinois Quad Cities
• adjunct associate professor, John A. Moran Eye Center, University of Utah, Salt Lake City
Section Editor Audrey R. Talley Rostov, MD
• private practice with Northwest Eye Surgeons, Seattle
• SightLife global partner
• kwalter@wakehealth.edu
Joseph Gira, MD
• private practice, Ophthalmology Consultants, St. Louis
• (314) 909-0633;kwalter@wakehealth.edu
• inancial disclosure: paid consultant to Bausch + Lomb
Anita Nevyas-Wallace, MD
• private practice, Nevyas Eye Associates, Bala Cynwyd, Pennsylvania
• clinical assistant, University of Pennsylvania Department of Ophthalmology, Philadelphia
• (610) 668-2777;kwalter@wakehealth.edu
• financial interest: none acknowledged
P. Dee G. Stephenson, MD
• president, American College of Eye Surgeons
• private practice, Stephenson Eye Associates, Venice, Florida
• associate professor of ophthalmology, University of South Florida College of Medicine, Tampa
• (941) 468-6363;eyedrdee@aol.com;Twitter @deestep03136405
• financial disclosure: key opinion leader for Bausch + Lomb and Lensar




