CASE PRESENTATION
A 67-year-old woman experiences trauma to the right eye from high-pressure water. Her optometrist notes a dislocated IOL in the eye and urgently refers the patient for an eye examination. Her ocular history includes bilateral cataract surgery with a multifocal IOL in 2016 followed later that year by an IOL exchange in the right eye for a different multifocal lens.
On presentation, the patient reports redness and photophobia in the right eye. She describes a shadow in her inferior peripheral vision but says her central vision is unchanged from baseline. Her UCVA is 20/25-2 OD and 20/40+2 OS. The IOP is 20 mm Hg OU.
Upon examination, the lens appears to be well centered. The inferonasal haptic, however, has extruded from the capsular bag, and there may be zonular dialysis in the area (Figures 1 and 2; here). Retinal hemorrhages are visible in the periphery, but no retinal breaks or detachment is evident. No anterior chamber reaction is observed, and the rest of the examination is unremarkable.
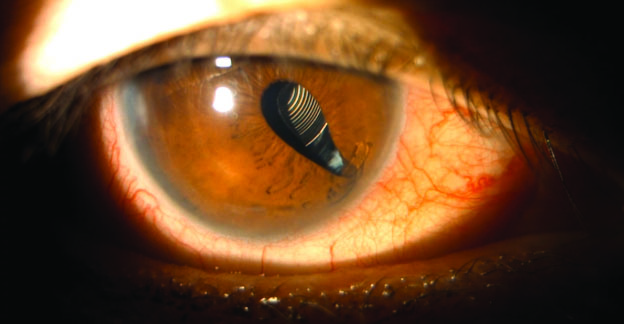
Figure 1. External photograph of the right eye. Inferonasal extrusion of the IOL haptic is evident.
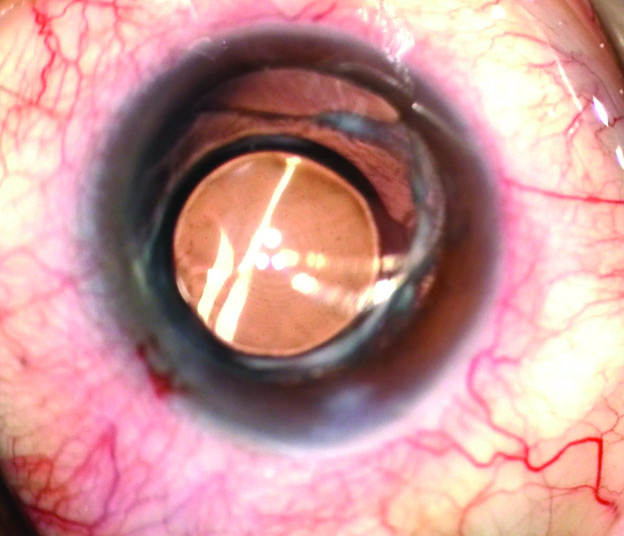
Figure 2. A haptic extrudes through the capsular bag, and 2 to 3 clock hours of zonular dialysis are visible.
How would you proceed? Would you reposition the IOL, exchange it, or perform scleral fixation?
— Case prepared by Neda Nikpoor, MD

CATHLEEN MCCABE, MD
I have had several patients with similar pathology after blunt trauma. Because only one haptic is anterior to the iris and the lens appears to be well centered, one option would be to check whether the anterior capsule is intact in the area of the affected haptic, inflate the bag with an OVD, and tuck the haptic back into the bag.
Alternatively, because the IOL is not a toric model, it might be possible to free the lens within the bag and reorient it by 90º to take advantage of stronger intact zonules and the capsule. Figures 1 and 2 appear to show some fibrosis of the bag, and fully mobilizing the IOL without further damaging the zonules could be difficult in areas of fibrosis. The lens appears to be a one-piece hydrophobic acrylic design, possibly one of the Tecnis lenses (Johnson & Johnson Vision). In my experience, this family of lenses typically develops fibrosis near the haptic-optic junction adjacent to the region where the haptic is widest. This area is where the greatest care should be exercised to avoid further trauma. It is also possible that the damage to the zonules and capsular stability would hinder centration and stabilization of the IOL, even after its rotation.
To my mind, this is a favorable scenario for scleral fixation of the IOL using the belt loop technique. The traumatic damage appears to be localized to the haptic that has been displaced in front of the pupil, there is visible fibrosis of the capsular bag, and the lens optic remains centered. A belt loop fashioned from a 6-0 polypropylene suture would be passed through the capsular bag and around the haptic at the optic-haptic junction. The suture would then be anchored in the superficial layers of the sclera to reposition the haptic securely and ensure that the IOL remains centered and well supported (see The Belt Loop Technique for Scleral Fixation). An anterior vitrectomy would likely be unnecessary, and the risk of further damage to the zonules and capsular bag would be minimal.
The Belt Loop Technique for Scleral Fixation
By Cathleen McCabe, MD
- Step No. 1: Mark the conjunctiva 2 mm posterior to the limbus in the axis of the optic-haptic junction.
- Step No. 2: Cut both ends of a segment of a 6-0 polypropylene suture at a bevel.
- Step No. 3: Using a hemostat, bend two 30-gauge needles at a 90º angle.
- Step No. 4: Test the suture by passing it into the 30-gauge needle outside the eye.
- Step No. 5: Pass the needle through conjunctiva and sclera just posterior to the previously placed mark on the conjunctiva. Keep the angle of insertion parallel to the iris to create a long tunnel.
- Step No. 6: Place the tip of the needle behind the capsule. Pierce the capsule at the optic-haptic junction and then pierce the anterior capsule.
- Step No. 7: Feed the cut 6-0 polypropylene suture into the needle lumen in one of three ways. The first option is to thread the needle inside the eye using microforceps. The second is to preplace the suture in the lumen of the needle and allow extrusion of the material (without bending the needle). The third is to externalize the needle through a paracentesis incision and use tying forceps to insert the suture into the lumen.
- Step No. 8: Push the trailing end of the suture toward the eye as you withdraw the needle from the sclera and conjunctiva.
- Step No. 9: Grasp the exposed suture end and make a large safety flange with low-temperature handheld cautery.
- Step No. 10: Repeat the previous steps and place a second needle anterior to the mark on the conjunctiva, through conjunctiva and sclera, and into the sulcus.
- Step No. 11: Place the trailing end of the same piece of cut suture into the needle lumen, withdraw it from the eye, and create a large flange.
- Step No. 12: Slowly externalize more of the suture from each exposed end to adjust the tension so that the IOL remains planar and centered.
- Step No. 13: Hold the suture such that, with proper tension, it slightly indents the sclera.
- Step No. 14: Cut the suture 1 to 1.5 mm from the ocular surface and use low-temperature cautery to make a small flange.
- Step No. 15: Bury the flange into superficial layers of sclera.
Click here to watch a demonstration.
In my experience, the belt loop technique preserves the benefits of a premium lens, carries a low risk of further traumatizing the zonules and capsular bag, and can provide long-term stability and centration of an existing IOL.

YURI MCKEE, MD
The square-edged haptic of an acrylic IOL is likely to irritate and damage the iris and ciliary body. Corneal damage is also a risk in this situation. The dislocated haptic would therefore be addressed without delay.
The choice of surgical approach depends on the stability of the capsule. Scarring of the capsule—common after cataract surgery—might make rotating the IOL difficult. Three clock hours of zonular dialysis are present, and the capsule has likely been compromised by the dislocated haptic.
Ideally, the haptic could be reposited in the capsule, and the IOL could be rotated away from the damage in the anterior capsule. This strategy might be wishful thinking, so I would be ready to place capsular support segments as needed to stabilize the capsule. My preference would be to secure the segments to the sclera using the Canabrava flanged suture technique1 but with the suture flanges buried in the sclera to reduce the risk of erosion and late endophthalmitis. If vitreous has prolapsed through the zonular dialysis, removal via a pars plana approach might be indicated. An injection of triamcinolone acetonide or the use of an intraoperative light pipe could help to identify vitreous prolapse, if present.
If, during surgery, it becomes evident that the capsule is too damaged to save, a complete vitrectomy would be performed, the IOL would be removed, the visual axis of the residual capsule would be cleared, and a monofocal IOL would be secured using intrascleral haptic fixation.
In cases of trauma like this one, I hope for the best but prepare multiple contingency plans. Informed consent includes the various possible outcomes of surgery, and I remind patients that surgery will be rehabilitative rather than refractive.

J. MORGAN MICHELETTI, MD, FACS
My first thought is to try to salvage the existing IOL if the eye’s visual potential remains excellent and the patient was happy with the IOL before the trauma. Before surgery, biometry and backup calculations for an IOL exchange would be performed.
This situation entails hoping for the best and planning for the worst. Refixation of the IOL is the hope, but a stepwise approach using multiple surgical plans and retaining the ability to pivot is mandatory.
The first step would be to confirm the anatomy and pathology. Sometimes, it is hard to tell in clinic whether a haptic is piercing the capsular bag or is simply anterior to the capsulorhexis (ie, it was never fully in the bag).
If the capsule is intact and the haptic is simply out of the bag, a dispersive OVD would be injected to reinflate the bag, and the haptic would be repositioned back into the capsular bag. Next, the capsular bag–IOL complex would be stabilized with a capsular tension ring (CTR) and/or a capsular tension segment (CTS). With 2 to 3 clock hours of zonular dialysis, the placement of a CTR alone might be enough to recenter the IOL, but the CTR should be gently placed using a two-handed technique with a Sinskey hook.
If the haptic has penetrated through the capsular bag, then the decision tree splits. If the defect is small and fibrotic and the rest of the capsule is stable, reinflation of the bag followed by 90º rotation of the IOL could be attempted to recapture the haptic fully within the capsule, followed by stabilization with a CTS. Haptic truncation would be another option, especially if fibrosis provides stability, followed by recentration of the bag with a CTS. The latter strategy, however, carries a greater risk of later IOL decentration within the bag if the superior haptic has not fibrosed in place.
If the capsular defect is larger or if a lack of fibrosis could allow it to extend, then the salvage options would be scleral fixation of the IOL using the belt loop technique or the horizontal punch and rescue technique. With either strategy, I would attempt to rotate and reposition the IOL back into the capsular bag to contain the haptics and prevent the development of uveitis-glaucoma-hyphema (UGH) syndrome. That said, successful refixation of a one-piece IOL outside the capsular bag with the belt loop technique or the punch and rescue technique has been reported.2 Nevertheless, if the IOL can be rotated 90º so that the haptic resides within the capsular bag, then the belt loop or punch and rescue strategy could be executed nasally to stabilize and recenter the IOL (click here to watch a demonstration).
Before proceeding to surgery, the patient must understand that the existing IOL might not be amenable to fixation and an IOL exchange might be necessary, which would mean a loss of multifocality.

WHAT I DID: NEDA NIKPOOR, MD
I explained to the patient that, during surgery, a decision would be made to reposition the haptic behind the iris, perform scleral fixation of the lens, or refer her for an IOL exchange. Intraoperatively, once the haptic had been repositioned behind the iris, the lens remained well centered. At that point, because the lens was quite stable despite a focal, small area of zonulopathy, the decision was made to stop to avoid further manipulation or decentration of the lens. As the adage goes, the enemy of good is better.
In this situation, simply repositioning the haptic behind the iris was the least invasive option to retain the lens and preserve the excellent vision and spectacle independence the patient had enjoyed. Scleral fixation would have been a viable option as well. Because the lens was already centered, however, repositioning the haptic was preferred to avoid manipulation that could have decentered the diffractive multifocal IOL. The risk of UGH syndrome with an exposed haptic was discussed with the patient both before and after surgery.
On postoperative day 1, the IOL was well centered, and her UCVA was 20/20 OD. At the most recent visit, 6 weeks after surgery, the lens remained well centered, her UCVA was 20/20 OD, and there was no evidence of inflammation or UGH syndrome.
The patient is happy with her visual acuity and spectacle independence but aware that repositioning or explantation of the lens may be required if it becomes dislocated or UGH syndrome develops. She is being monitored closely.
1. Canabrava S, Canêdo Domingos Lima AC, Ribeiro G. Four-flanged intrascleral intraocular lens fixation technique: no flaps, no knots, no glue. Cornea. 2020;39(4):527-528.
2. Micheletti JM, Weber N, McCauley MB, Doe EA, Coffee RE, Caplan MB. Punch and rescue technique for scleral fixation of dislocated single-piece intraocular lenses. J Cataract Refract Surg. 2022;48(2):247-250.




