Refractive Misses
Thomas Meirick, MD, and Andrew Chen, MD


One of the most frustrating complications of modern cataract surgery is a postoperative refractive surprise. Although surgical complication rates in cataract surgery remain low—approximately 2% in large trials—refractive surprises are not uncommon.1,2 Approximately 25% of patients have a postoperative refractive surprise if the stringent criterion of ±0.50 D is used.3
PREOPERATIVE WORKUP
A careful ocular history and a comprehensive examination are key parts of predicting the risk of these complications and preventing them. Refractive surprises are more common in patients with glaucoma, regardless of their preoperative BCVA.4
A history of refractive surgery puts patients at increased risk of refractive misses.3 Optimization of the ocular surface, sufficient time out of contact lenses before surgery, and good control of chronic systemic diseases such as diabetes help ensure accurate biometry and proper lens selection.5-8 Immersion ultrasonography is ideal in eyes requiring ultrasonography, such as those with dense cataracts or media opacities; contact ultrasonography can artificially shorten axial length.9,10
Corneal topography should be used to confirm keratometry readings, especially when a toric lens is being considered. The results should be carefully reviewed to rule out irregular astigmatism that may be not apparent on examination (Figure 1).
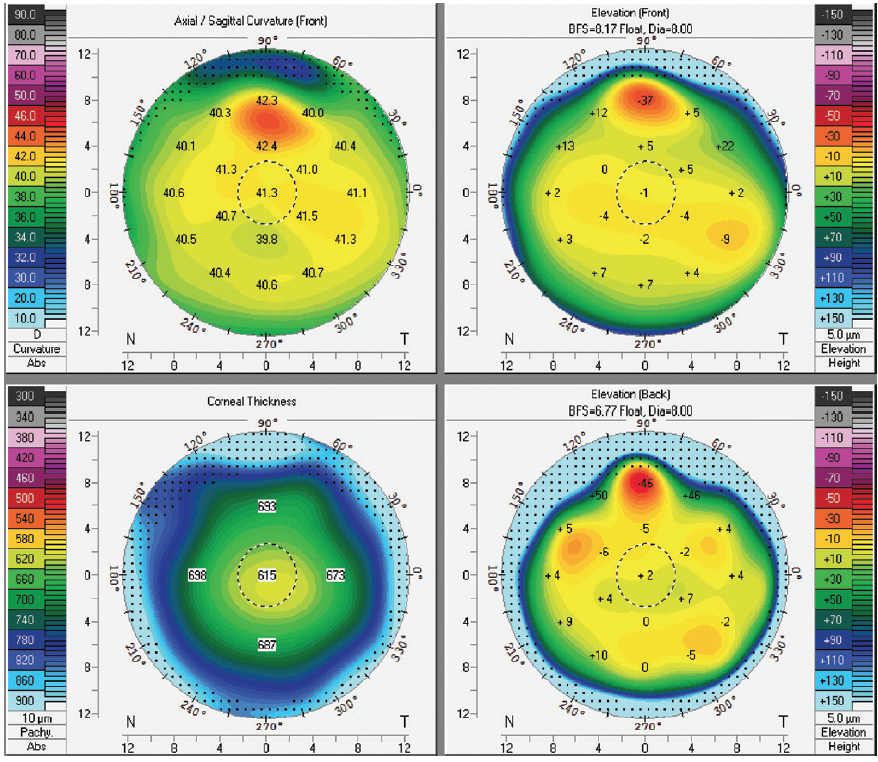
Figure 1. Scheimpflug topography of a patient with forme fruste keratoconus, otherwise undetected on examination.
Courtesy of Andrew Chen, MD
IOL POWER CALCULATION
In a retrospective review, the Barrett II formula was associated with the lowest overall average prediction error and the highest percentage of eyes within ±0.50 D of their goal. These results remained true in short eyes (< 22.5 mm). For long eyes (> 25.5 mm), although the Barrett II formula was slightly less accurate, it was the second most accurate of the formulas evaluated in this study.11
Smaller studies have confirmed the Barrett II formula’s accuracy in several subgroups, including patients with steep corneas, elderly patients, and patients from multiple ethnic backgrounds.12-15 Newer formulas such as the Kane, Olsen (4-factor), and Emmetropia Verifying Optical compare favorably to the Barrett II.16 When possible, A-constants should be optimized to increase refractive accuracy.17 Finally, in patients with a history of refractive surgery, the ASCRS maintains a postrefractive surgery calculator that offers multiple calculation methods.
MANAGEMENT
Refractive misses may not require intervention. A patient may be content with a small myopic surprise, for example. If the patient is unhappy, the least invasive choice is to trial corrective spectacles or contact lenses. If the patient is averse to wearing corrective lenses, laser vision correction, an IOL exchange, and a piggyback IOL are alternatives.
The options available for managing refractive surprises are increasing. The Light Adjustable Lens (RxSight), for example, allows a postoperative adjustment to the refractive result. Early data suggest that the refractive outcomes achieved with this lens are stable for several years, and no significant effect of the UV light upon the cornea or macula has been shown.18 These lenses can be especially useful when the patient has a history of refractive surgery.19
1. Chancellor J, Soliman MK, Shoults CC, et al; Pseudophakic Macular Study Group. Intraoperative complications and visual outcomes of cataract surgery in diabetes mellitus: a multicenter database study. Am J Ophthalmol. 2021;225:47-56.
2. Narendran N, Jaycock P, Johnston RL, et al. The Cataract National Dataset electronic multicentre audit of 55,567 operations: risk stratification for posterior capsule rupture and vitreous loss. Eye (Lond). 2009;23(1):31-37.
3. Lundström M, Dickman M, Henry Y, et al. Risk factors for refractive error after cataract surgery: analysis of 282 811 cataract extractions reported to the European Registry of Quality Outcomes for cataract and refractive surgery. J Cataract Refract Surg. 2018;44(4):447-452.
4. Manoharan N, Patnaik JL, Bonnell LN, et al. Refractive outcomes of phacoemulsification cataract surgery in glaucoma patients. J Cataract Refract Surg. 2018;44(3):348-354.
5. Hovanesian J, Epitropoulos A, Donnenfeld ED, Holladay JT. The effect of lifitegrast on refractive accuracy and symptoms in dry eye patients undergoing cataract surgery. Clin Ophthalmol. 2020;14:2709-2716.
6. Ng LT, Lee EM, Nguyen AL. Preoperative assessment of corneal and refractive stability in soft contact lens wearing photorefractive candidates. Optom Vis Sci. 2007;84(5):401-409.
7. Goudie C, Tatham A, Davies R, Sifton A, Wright M. The effect of the timing of the cessation of contact lens use on the results of biometry. Eye (Lond). 2018;32(6):1048-1054.
8. Rayess Y, Arej N, Abdel Massih Y, Antoun J, Waked N. Influence of soft contact lens material on corneal warpage: prevalence and time to resolution. Can J Ophthalmol. 2018;53(2):135-138.
9. Trivedi RH, Wilson ME. Axial length measurements by contact and immersion techniques in pediatric eyes with cataract. Ophthalmology. 2011;118(3):498-502.
10. Yang QH, Chen B, Peng GH, Li ZH, Huang YF. Accuracy of axial length measurements from immersion B-scan ultrasonography in highly myopic eyes. Int J Ophthalmol. 2014;7(3):441-445.
11. Melles RB, Holladay JT, Chang WJ. Accuracy of Intraocular Lens Calculation Formulas. Ophthalmology. 2018;125(2):169-178.
12. Zhang C, Dai G, Pazo EE, et al. Accuracy of intraocular lens calculation formulas in cataract patients with steep corneal curvature. PLoS One. 2020;15(11):e0241630.
13. Sella R, Chou L, Schuster AK, Gali HE, Weinreb RN, Afshari NA. Accuracy of IOL power calculations in the very elderly. Eye. 2020;34(10):1848-1855.
14. Liu J, Wang L, Chai F, et al. Comparison of intraocular lens power calculation formulas in Chinese eyes with axial myopia. J Cataract Refract Surg. 2019;45(6):725-731.
15. Kuthirummal N, Vanathi M, Mukhija R, et al. Evaluation of Barrett universal II formula for intraocular lens power calculation in Asian Indian population. Indian J Ophthalmol. 2020;68(1):59-64.
16. Melles RB, Kane JX, Olsen T, Chang WJ. Update on intraocular lens calculation formulas. Ophthalmology. 2019;126(9):1334-1335.
17. Aristodemou P, Knox Cartwright NE, Sparrow JM, Johnston RL. Intraocular lens formula constant optimization and partial coherence interferometry biometry: refractive outcomes in 8108 eyes after cataract surgery. J Cataract Refract Surg. 2011;37(1):50-62.
18. Schojai M, Schultz T, Schulze K, Hengerer FH, Dick HB. Long-term follow-up and clinical evaluation of the light-adjustable intraocular lens implanted after cataract removal: 7-year results. J Cataract Refract Surg. 2020;46(1):8-13.
19. Moshirfar M, Duong AA, Shmunes KM, Castillo-Ronquillo YS, Hoopes PC. Light adjustable intraocular lens for cataract surgery after radial keratotomy. J Refract Surg. 2020;36(12):852-854.
Preventing and Managing Pseudophakic Cystoid Macular Edema
Mitra Nejad, MD

Complications during cataract surgery are rare, and the majority of patients have improved vision immediately postoperatively. Complications, however, can develop weeks after routine, uncomplicated surgery. The most common of these is cystoid macular edema (CME), which occurs after 1% to 2% of all cataract surgeries.1
The peak incidence of pseudophakic CME is 6 to 8 weeks after surgery. The gold standard for its diagnosis is fluorescein angiography. Most surgeons, however, rely on OCT for both diagnosis and monitoring. Treatment ranges from topical eye drops to intraocular injections to vitreoretinal surgery.
PREVENTION
Perhaps the most important pearl for CME prevention is the preoperative identification of patients who are at increased risk of developing this complication. These include patients who developed CME in the contralateral eye, those with a history of CME in the operative eye, and those with a leaky blood-retinal barrier.2 The last is often seen in patients with diabetes, retinal vein occlusion, macular degeneration, retinal dystrophies, radiation retinopathy, or epiretinal membranes and those with a history of uveitis or prostaglandin use (Figures 2 and 3). In one study, patients with a preoperative central macular thickness greater than 260.5 µm were at nine times greater risk of postoperative intraretinal cysts after uncomplicated cataract surgery than patients with a normal macular thickness.3
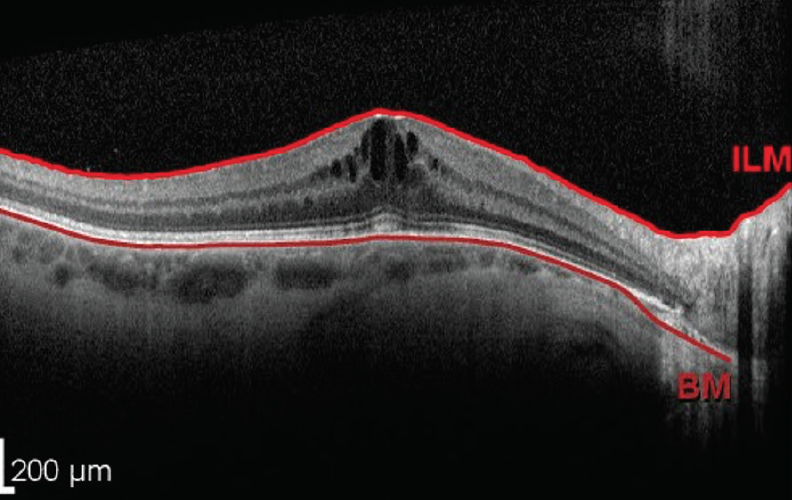
Figure 2. Postoperative pseudophakic CME in a patient with a history of chronic anterior uveitis.
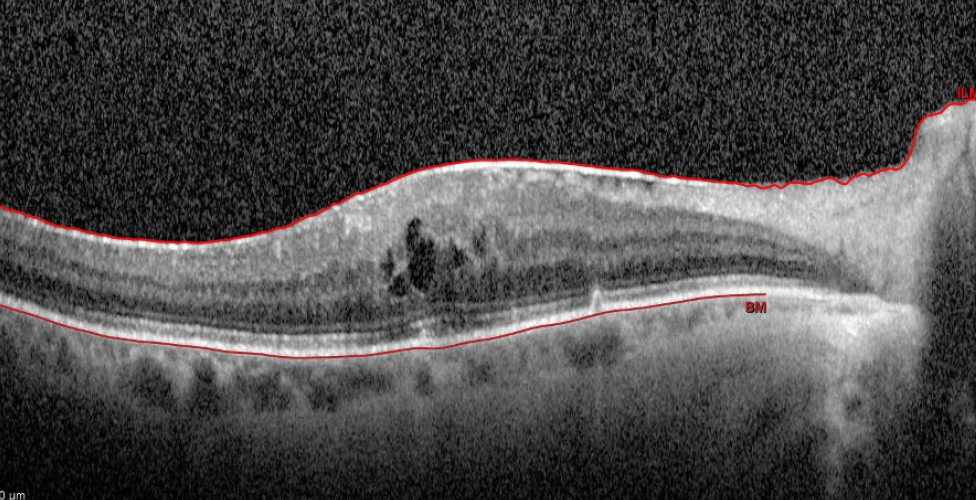
Figure 3. Postoperative pseudophakic CME in a patient with a preoperative epiretinal membrane.
Courtesy of Mitra Nejad, MD
Identifying high-risk patients is helpful for the following reasons:
- Patients can be educated to monitor their vision for changes that may occur 1 to 2 months after surgery.
- Surgeons can individualize the perioperative drop regimen. Those who do not routinely use a nonsteroidal topical therapy postoperatively may consider incorporating one for higher-risk patients.
Intraoperatively, avoiding complications such as posterior capsular tears and retained lens fragments decreases the risk of postoperative CME. Another helpful surgical tactic is to increase the efficiency of nuclear removal. Studies suggest that total cumulative dissipated energy and phaco time in uncomplicated cataract surgery directly correlate with CME development.4 A recently published retrospective case series suggests that the intraoperative use of NSAIDs can reduce the risk for postoperative CME.5
MANAGEMENT
The first step of management is to perform a thorough evaluation to determine the cause of CME. In the event an anterior vitrectomy was performed, vitreous strands that adhere to the wounds can cause traction, inducing CME. Vitreolysis with an Nd:YAG laser can be performed to relieve this traction. A careful examination, including gonioscopy, for retained lens fragments is indicated for patients with CME and those with prolonged anterior chamber reactions and/or corneal edema. Removal of a retained fragment can quickly resolve CME.
Pseudophakic CME that has been present for months is generally more resistant to treatment than recently formed cysts. Early detection and treatment are therefore advantageous. Topical steroids and topical NSAIDs, either alone or in combination, are commonly used as first-line treatment. In the United States, topical NSAID use is an on-label indication for the prevention of inflammation and pain for 14 days after cataract surgery. Their use for CME treatment, however, is considered off-label.
Different NSAID formulations have been shown to be effective for the prevention and treatment of CME. At least one study has suggested that nepafenac 0.1% and bromfenac 0.09% may be more effective than diclofenac 0.1% and ketorolac 0.4% for the management of chronic pseudophakic edema.6 More studies are needed to confirm the superiority of these newer-generation NSAIDs.
After topical therapy is initiated, cataract surgeons may begin to assess patients for an improvement in CME after about 1 month. If there is no significant improvement, referral to a retina specialist may be advisable. Patients with postoperative CME are often disappointed and may feel reassured by the referral to a specialist. Intravitreal injections of corticosteroids and intravitreal dexamethasone implants can be effective. Anti-VEGF agents may provide more targeted therapy for those with retinal vasculopathies predisposing them to the development of CME.7 In eyes with chronic CME and vitreomacular traction, pars plana vitrectomy may also be considered.
CONCLUSION
Most cataract surgeons encounter CME a few times per year. It is important to identify which patients are at increased risk of this complication and to take steps toward its prevention. When CME does occur, prompt diagnosis and the rapid initiation of topical treatments lead to the resolution of most cases of pseudophakic CME. Referral to a retina colleague may be warranted to prevent irreversible vision loss in patients experiencing chronic CME.
1. Henderson BA, Pineda R, Chen SH. Essentials of Cataract Surgery. Slack; 2014.
2. McCafferty S, Harris A, Kew C, et al. Pseudophakic cystoid macular edema prevention and risk factors; prospective study with adjunctive once daily topical nepafenac 0.3% versus placebo. BMC Ophthalmol. 2017;17(1):16.
3. Doncel-Fernandez CJ, Alferez-Asenio ML, Quereda-Castaneda A, Castro-Luna G. Preoperative central macular thickness as a risk factor for pseudophakic macular edema. Graefes Arch Clin Exp Ophthalmol. 2021;259(1):37-43.
4. Anastasilakis K, Mourgela A, Symeonidis C, Dimitrakos SA, Ekonomidis P, Tsinopoulos I. Macular edema after uncomplicated cataract surgery: a role for phacoemulsification energy and vitreoretinal interface status? Eur J Ophthalmol. 2015;25(3):192-197.
5. Walter K, Kauffman L, Hess J. Rate of pseudophakic cystoid macular edema using intraoperative and topical nonsteroidal antiinflammatory drugs alone without steroids. J Cataract Refract Surg. 2020;46(3):350-354.
6. Warren KA, Bahrani H, Fox JE. NSAIDs in combination therapy for the treatment of chronic pseudophakic cystoid macular edema. Retina. 2010;30(2):260-266.
7. Guo S, Patel S, Baumrind B, et al. Management of pseudophakic cystoid macular edema. Surv Ophthalmol. 2015; 60(2):123-137.
High IOP
Mohamed S. Sayed, MD

Every cataract surgeon occasionally encounters a patient with elevated IOP after cataract surgery, even after uneventful phacoemulsification. Measures can be taken to minimize the incidence of this complication. Identifying higher-risk patients such as those with advanced glaucomatous optic neuropathy is important because even a brief period of significant IOP elevation may result in permanent damage.
The cataract procedure itself may reduce IOP by an average of 2 to 4 mm Hg.1 This is likely brought about by the widening of the anterior chamber (AC) angle and aqueous outflow pathway that occurs when the thick, cataractous crystalline lens is replaced with a significantly thinner IOL. This widening is particularly pronounced in eyes with a preexisting narrow angle, such as those with significant hyperopia. Further, cataract surgery is an increasingly popular alternative to laser peripheral iridotomy in eyes with primary angle closure and anatomically narrow angles (Figure 4).2
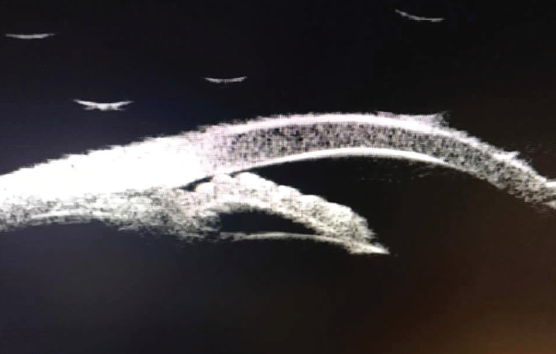
Figure 4. Ultrasound biomicroscopy of an eye with a small AC and narrow angle. Cataract surgery may be a better alternative to laser peripheral iridotomy to prevent episodes of intermittent or acute angle closure.
Courtesy of Mohamed S. Sayed, MD
This article shares tips that may help to manage elevations in IOP on the first postoperative day and in patients with preexisting glaucoma.
ELEVATED PRESSURE ON the FIRST POSTOPERATIVE DAY
When IOP is elevated on the first postoperative day, the patient’s vision can be blurry from the associated corneal microcystic edema.
In the immediate postoperative period, an increase in IOP is most commonly related to a retained dispersive OVD in the AC angle. In this scenario, all of the OVD should be removed from the eye using high flow, vacuum, and I/A settings. If visualization is difficult, it can be helpful to sweep the angle. First, balanced salt solution is injected through the paracentesis incision to mobilize the OVD retained in the angle. It is then burped out of the sideport or main incision.
If, on the postoperative day 1 examination, the elevation in IOP is presumably the result of a retained OVD, several rounds of topical aqueous suppressants may be administered 10 to 15 minutes apart. Additionally, oral carbonic anhydrase inhibitors such as acetazolamide can be administered in the office and prescribed for the first few postoperative days if this class of medication is not contraindicated. When IOP reaches a relatively high level, however, an AC tap can be performed in the office after administering a topical anesthetic and 5% betadine solution with a 27-gauge needle on a 1-mL syringe (after removing its plunger) through the preexisting paracentesis incision. This maneuver can produce an immediate lowering of the pressure. An IOP check should be performed about 30 to 45 minutes later; if the IOP rises once again, the procedure may be repeated several times until an acceptable IOP is achieved and sustained.
An AC tap has inherent risks and should be avoided if other measures can successfully lower the IOP.
Another far less common cause of elevated IOP on the first postoperative day is malignant glaucoma, also known as aqueous misdirection. Hyperopic eyes with a short axial length are at risk for developing this complication, particularly if a peripheral iridotomy or glaucoma-filtering surgery was performed concurrently with or before cataract surgery. Timely recognition and treatment of this complication are of paramount importance. On examination, IOP can be elevated with significant microcystic edema, and the AC is uniformly shallow with iridocorneal and even IOL-corneal touch.
Ultrasound biomicroscopy can help confirm the diagnosis. It typically reveals anterior rotation of the ciliary body processes. Topical atropine 1% drops are often effective in aborting the episode. If the response is suboptimal, creating a posterior capsulotomy through a preexisting peripheral iridotomy may be of benefit. If the aforementioned measures fail, taking the patient back to the OR and performing a zonulohyaloidotomy with limited core vitrectomy may be necessary.
ELEVATED IOP IN A PATIENT WITH GLAUCOMA
Patients with glaucoma are particularly vulnerable to damage from even brief periods of IOP elevation. A thorough washout of all OVD from the AC at the conclusion of cataract surgery must therefore be attempted. Patients who are already on a regimen of topical antiglaucoma therapy should be instructed to administer their drops on the day of surgery. The postoperative use of oral carbonic anhydrase inhibitors may also be considered in select patients.
Many patients with glaucoma experience a postoperative IOP spike from steroid use, typically during the second postoperative week. A steroid response can be effectively managed by a rapid steroid taper, switching to a weaker steroid, or substituting a topical NSAID.
The approach to cataract surgery in patients with a history of glaucoma surgery such as trabeculectomy or glaucoma drainage device implantation differs from routine cataract surgery. Aqueous drainage and the degree of IOP control must be assessed before cataract surgery because inflammation, scarring, and other complications caused by the cataract procedure can compromise a functioning bleb.3,4 This is a particular concern when the interval between glaucoma and cataract surgery is short. The risk of bleb failure decreases as the time between the two procedures increases. Delaying cataract surgery for at least 2 years after trabeculectomy, if possible, may therefore reduce the risk of bleb failure. Moreover, the higher the IOP before cataract surgery, the higher the risks of bleb failure and postoperatively elevated IOP because blebs with borderline functionality may be more susceptible to the inflammation and scarring that cataract surgery can induce.
COMBINED PROCEDURES
The development of MIGS devices and procedures has shifted surgical intervention earlier in the course of glaucoma (scan the QR code now to watch cataract surgery combined with ab interno trabeculotomy using the OMNI Surgical System [Sight Sciences]). MIGS also simplifies combining cataract surgery with IOP-lowering procedures for general ophthalmologists and cataract surgeons. More severe or complex cases should be reserved for glaucoma surgeons.
In patients with a history of elevated IOP, combining cataract surgery with a MIGS procedure may prevent postoperative pressure spikes. For instance, endoscopic cyclophotocoagulation and a trabecular outflow MIGS procedure can be combined, if necessary, to achieve better IOP control.
CONCLUSION
High IOP after cataract surgery can be managed effectively in most instances. Care should be taken during the preoperative examination to identify higher-risk patients and avoid even brief periods of significant IOP elevation, which may result in permanent damage.
1. Friedman DS, Jampel HD, Lubomski LH, et al. Surgical strategies for coexisting glaucoma and cataract: an evidence-based update. Ophthalmology. 2002;109(10):1902-1913.
2. Azuara-Blanco A, Burr J, Ramsay C, et al. Effectiveness of early lens extraction for the treatment of primary angle-closure glaucoma (EAGLE): a randomised controlled trial. Lancet 2016;388(10052):1389-1397.
3. Husain R, Liang S, Foster PJ, et al. Cataract surgery after trabeculectomy: the effect on trabeculectomy function. Arch Ophthalmol. 2012;130(2):165-170.
4. Mathew RG, Murdoch IE. The silent enemy: a review of cataract in relation to glaucoma and trabeculectomy surgery. Br J Ophthalmol. 2011;95(10):1350-1354.
Pseudophakic Bullous Keratopathy
Michael J. Taravella, MD

Persistent corneal edema after cataract surgery can be distressing for both physicians and patients. Signs and symptoms of corneal edema include poor visual acuity, Descemet folds, and an obviously thickened cornea. Severe edema, referred to as pseudophakic bullous keratopathy (Figure 5), may be associated with corneal bullae.
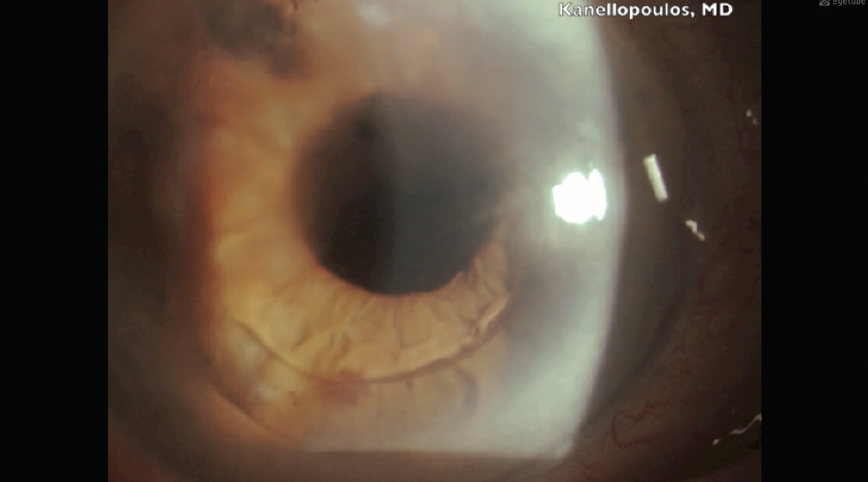
Figure 5. Obvious pseudophakic bullous keratopathy.
Courtesy of A. John Kanellopoulos, MD
The incidence of pseudophakic bullous keratopathy peaked in the mid-1980s and 1990s. Since that time, it has decreased steadily owing to advances in surgical technique and IOL design. Presently, pseudophakic bullous keratopathy is expected to occur after less than 0.5% to 1% of all cataract procedures.1 Efforts should be focused on prevention.
PREVENTION
Risk factors for pseudophakic bullous keratopathy include prior surgery, unrecognized (Fuchs) corneal dystrophy (Figure 6), toxic anterior segment syndrome, and a dense cataract that may require prolonged surgical time and a greater amount of phaco energy. A careful slit-lamp examination is required during the preoperative evaluation. If corneal guttata is found, specular microscopy and pachymetry are useful for documenting the extent of corneal involvement and mild corneal edema, if present. Eyes with cell counts below 1,000 cells/mm2 or extensive guttae are at risk of developing persistent edema. Corneal thickness greater than 600 µm indicates the possible presence of corneal edema preoperatively. For comparison, a normal cell count ranges from 2,000 to 3,000 cells/mm2. Corneal thickness is approximately 550 µm on average, although the normal range is broad.
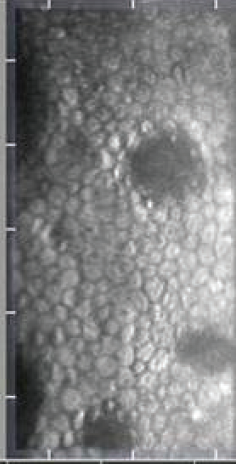
Figure 6. Fuchs corneal dystrophy.
Courtesy of Michael J. Taravella, MD
Patient counseling includes a discussion of the risk of corneal decompensation and a possible need for endothelial keratoplasty. If corneal edema is present preoperatively, cataract surgery can be combined with endothelial keratoplasty.2
Several strategies can help to limit the extent of endothelial damage and cell loss at the time of cataract surgery. These include the use of liberal amounts of dispersive OVD and balanced salt solution plus (the plus stands for glutathione, a free radical scavenger). A third measure is to minimize the use of potentially toxic intracameral medications. Strategies for decreasing phaco time and/or energy and potential endothelial trauma may also be adopted. For example, the use of a femtosecond laser for prechopping the nucleus may reduce the phaco time required for nuclear disassembly.3
MANAGEMENT
Moderate and severe corneal edema observed during the early postoperative period may respond to aggressive therapy. Inflammation after cataract surgery can be addressed with frequent applications of a potent topical steroid such as prednisolone acetate. This may be prescribed up to hourly in the first few days after surgery. Difluprednate may require less frequent application than other topical steroids.
Retained cortex and nuclear fragments can cause persistent inflammation. These must be addressed if present. Vitreous in the anterior chamber that touches the endothelium should be removed via vitrectomy. An IOP reduction may also be beneficial by reducing stress on the remaining endothelial cells. Brimonidine, timolol, and oral acetazolomide are preferred. In theory, a prostaglandin analogue may contribute to inflammation and probably should be avoided if possible.
Patients who do not respond to conservative methods may benefit from Descemet membrane or Descemet stripping automated endothelial keratoplasty.2 Excellent outcomes can be attained with endothelial keratoplasty. Generally, corneal surgery can be performed within a few months of the cataract procedure, preferably before corneal scarring becomes evident.
1. Taravella M. Postoperative corneal edema. Medscape. October 16, 2018. Accessed March 2, 2021. https://emedicine.medscape.com/article/1193218-overview#a5
2. Stuart AJ, Romano V, Virgili G, Shortt AJ. Descemet's membrane endothelial keratoplasty (DMEK) versus Descemet's stripping automated endothelial keratoplasty (DSAEK) for corneal endothelial failure. Cochrane Database Syst Rev. 2018;6(6):CD012097.
3. Wei Wei DY, Hui-Chen CC, Liang S, Ray M, Wee Tien AT. Comparing outcomes of phacoemulsification with femtosecond laser-assisted cataract surgery in patients with fuchs endothelial dystrophy. Am J Ophthalmol. 2018;196:173-180.




