
As new refractive surgical technologies and techniques become available, selecting the most appropriate solution for each patient becomes increasingly challenging. There are now eight types of refractive surgical procedures: LASIK, PRK, small-incision lenticule extraction (SMILE), corneal inlays, phakic IOLs, refractive lens exchange, refractive cataract surgery, and CXL (Figure). Each procedure has particular advantages and disadvantages for a given patient. The decision process for patients around age 50, who are now entering the second stage of dysfunctional lens syndrome (DLS), is particularly complex, as there is significant overlap among available options in this age group (see Is Dysfunctional Lens Replacement LASIK for Baby Boomers?). This cohort includes many natural plano presbyopes or post-LASIK patients who have enjoyed excellent distance vision for many years, so they often have high expectations.
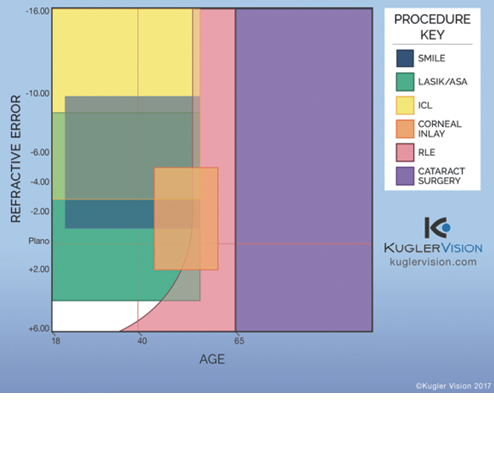
Figure. The refractive surgery procedure spectrum. (Abbreviations: SMILE, small-incision lenticule extraction; ASA, advanced surface ablation; RLE, refractive lens exchange.)
Refractive lens exchange (RLE) has gained acceptance in recent years as advances in IOL design, ocular biometry, and the ability to enhance postoperative refractive error have led to LASIK-like precision and the ability to correct near and intermediate vision in addition to distance. RLE has significant benefits over corneal refractive surgery, including a larger range of treatable refractive errors, elimination of age-based crystalline lens scatter and future cataracts, the ability to provide bilateral near vision, and less disruption of the corneal surface.
With proper patient selection, these advantages can lead to excellent outcomes and extremely satisfied patients. This article details the decision-making process we use in our practice to determine an individual’s suitability for refractive surgery, and specifically for RLE.
PATIENT SELECTION
Needs evaluation. As with any refractive surgery patient, the process of determining whether the patient is an appropriate candidate for RLE begins with a thorough assessment of his or her visual needs, including occupational requirements, leisure activities, hobbies, and any anticipated changes to those needs in the near and distant future. Understanding the patient’s expectations of what the surgery will provide to him or her is also crucial.
Objective testing and examination. In our practice, we divide the patient evaluation process into two phases. In phase 1, we determine visual acuities, eye dominance, and manifest refraction, and we perform topography/tomography (»Pentacam, Oculus Optikgeräte), endothelial spectroscopy, optical scatter index (OSI) measurement with the HD Analyzer (Visiometrics), meibomian gland imaging, tear-film aberrometry, and anterior segment slit-lamp examination.
Pertinent findings in phase 1 include magnitude of refractive error, BCVA, keratometry measurement, axis and magnitude of astigmatism, topographic regularity, corneal pachymetry, dry eye status, lens clarity, OSI measurement, and presence of corneal disease. With this information, we can determine whether the patient is a candidate for a corneal refractive procedure such as LASIK, a lens-based procedure such as RLE, both, or neither. An in-depth discussion and review of testing takes place with the patient, and a decision is made as to which procedure and technique will most likely be used.
In phase 2, we obtain Placido-disc topography and wavefront aberrometry (»iTrace, Tracey Technologies), macular OCT, optical biometry with the »Lenstar (Haag-Streit) or »IOLMaster (Carl Zeiss Meditec), and any specific testing that may be needed based on phase 1 findings. IOP is checked with an Icare tonometer (Icare USA), and the eyes are dilated. After cycloplegic refraction is obtained, slit-lamp and posterior segment examinations are performed.
Pertinent findings in phase 2 include lens clarity, higher-order aberrations arising from the corneal surface and internal optics, macular health, peripheral retinal health, and axial length. Armed with this additional information, the surgical plan is refined.
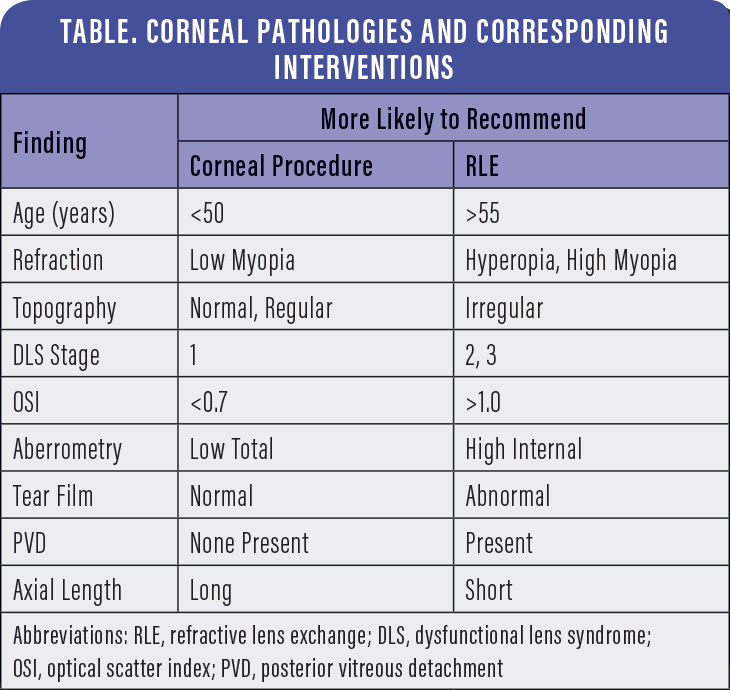
The ultimate decision as to which of the eight procedures will be used is complex and involves a multitude of subjective and objective considerations. However, certain objective considerations may help clarify the decision (Table).
For patients at increased risk for retinal detachment, such as those with long axial lengths and/or retinal conditions such as lattice degeneration, an evaluation by a retina subspecialist is recommended. This consultation may identify areas requiring prophylactic laser treatment, and it also helps patients understand the importance of retina health and attention to retinal symptoms.
SURGICAL PLAN
Once a definitive decision has been made to pursue RLE, the next step is to determine which technologies and techniques will be used to achieve the desired result. For patients with strong distance or night vision demands, bilateral monofocal or toric IOLs are excellent options. For patients with a desire for spectacle-free near and/or intermediate vision, extended depth of focus (EDOF) IOLs, multifocal IOLs, or a combination of the two may be used. Blended vision, or monovision, with monofocal or toric IOLs is also an excellent approach. It is well tolerated, particularly in patients with a history of using blended vision with contact lenses.
In all cases, existing astigmatism must be managed. There are many techniques available to manage astigmatism, including performing limbal relaxing incisions (LRIs) or on-axis paired corneal incisions, implanting toric IOLs, or combining the RLE procedure with a corneal procedure such as LASIK or PRK. (Editor’s note: For more information on managing astigmatism, see Toric IOLs for Astigmatism Correction, by Sneha Konda, BS; and Balamurali K. Ambati, MD, PhD, MBA, on pg 63.)
POSTSURGICAL PLAN
Perhaps just as important as the surgical plan is the postoperative plan. Despite tremendous advances in IOL calculation technology, including improved formulas, more accurate biometry, and the availability of intraoperative aberrometry, an enhancement rate of 10% or higher is reasonable and expected.
Patients undergoing RLE must be educated before surgery that there is a high likelihood that they will need an enhancement procedure of some sort following their RLE, and surgeons must be prepared to offer such procedures if needed, even if that means referring to a colleague for additional expertise.
Enhancements may include LRIs, LASIK, PRK, SMILE, piggyback IOL implantation, or IOL exchange. Patients should also be aware that there will be a nearly universal need for Nd:YAG capsulotomy at some time over the course of their lives. (Editor’s note: For more information on piggyback IOL implantation, see Piggyback IOL as an Enhancement Technique, by David A. Goldman, MD, on pg 69.)
SUMMARY
Refractive surgeons have at their disposal a complex array of procedures and techniques that they can employ to achieve their patients’ desired outcomes. Whether patients are in DLS stages 1, 2, or 3, the decision process begins by determining whether the outcome is best achieved with a cornea- or lens-based procedure. Astigmatism management and a postoperative enhancement plan are critical to success.




