Meibomian gland dysfunction (MGD) is characterized by abnormal composition and/or abnormal secretion of meibum. Patients with the disease exhibit various degrees of inflammation and often experience dry eye symptoms such as redness, burning, crusting, irritation, and fluctuating vision. Although MGD is often present early in life, the prevalence of dry eye increases with age. A recent study by Lemp et al showed that MGD occurred in 86% of patients presenting for dry eye care.1 MGD and dry eye disease (DED) are very much interrelated, both disrupting and destabilizing the ocular surface.
CATARACT SURGERY
Cataract surgery is one of the most commonly performed procedures by ophthalmologists today. Often, when patients present for a cataract evaluation, their baseline complaints can overlap with MGD and DED; that is, both groups complain of blurry vision. Patients presenting for cataract surgery frequently do not realize they have ocular surface disease and may even be asymptomatic. Patients' expectations of precise visual outcomes are at an all-time high, and a healthy ocular surface is pivotal to obtaining these results. Nor can clinicians take full advantage of advanced technologies if the ocular surface is unhealthy. It therefore behooves surgeons to address underlying MGD and DED preoperatively. In my experience, a poor tear film can lead to diagnostic errors such as altered biometry, abnormal topography, and refractive surprises—especially in patients receiving toric IOLs.
DRY EYE DISEASE
The estimates of DED's prevalence may be falsely low. The Prospective Health Assessment of Cataract Patient Ocular Surface (PHACO) Study evaluating patients presenting for cataract surgery found that over 60% of these individuals had abnormal tear breakup time (TBUT) and that even more had some degree of corneal fluorescein staining.2 Postoperatively, corneal sensitivity decreases,3 which may explain an increase in ocular surface symptoms. An abnormal tear film reduces retinal image quality by 20% to 40%, and image quality degradation and higher-order aberrations occur more often in eyes with DED compared with healthy eyes.4-6 This impairment in vision quality may explain why patients with MGD and DED are unhappy after cataract surgery, especially after receiving multifocal IOLs.
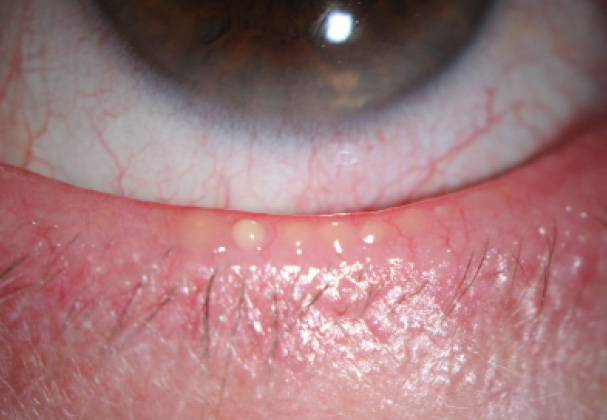
Figure. Presurgical patient with MGD illustrated by thick meibum expressed with gentle pressure on the eyelid. This patient also has eyelid margin telangiectasias and mild conjunctival injection.
MAKING IT SIMPLE
Today, clinicians have access to many tools that measure surrogate markers for DED and MGD, including the thickness of the lipid layer (LipiView; TearScience), noncontact TBUT (Oculus Keratograph; Oculus), tear osmolarity (TearLab Osmolarity System; TearLab), and matrix metalloproteinase 9 levels (InflammaDry; Rapid Pathogen Screening). These technologies add to traditional approaches such as the Schirmer test and TBUT using fluorescein.
In my practice, I prefer to use point-of-care testing for my presurgical patients, because it simplifies the screening process and allows me to easily identify DED and MGD in this patient population. My technicians collect the data, and I interpret the test results to determine whether or not MGD or DED treatment is needed prior to considering cataract surgery.
CLINICAL EXAMINATION
Diagnostic tools are only part of the picture. Another important step in treating patients with MGD is the clinical examination. I have found that a simple inspection of the meibomian glands and the corneal epithelium provides excellent information, and the examination does not take long to perform. I specifically look for an alteration in the meibomian glands' position or architecture and the presence of telangiectasias. Most importantly, I apply gentle pressure on the glands in order to assess the quality of the oil expressed (Figure). This step can be accomplished using a thumb or a cotton swab or more formally with a meibomian gland evaluator (TearScience). The simple maneuver can provide a great deal of information about the lipid layer and an asymptomatic patient's propensity for developing DED postoperatively.
WHEN AND HOW TO TREAT MGD
I believe that it is critical to screen all patients undergoing cataract surgery for DED and MGD. If either or both conditions are present, I always discuss the diagnosis with my patients so that they are aware of these disease processes as a potential barrier to achieving their desired outcomes after cataract surgery.
TREATMENT CHOICES
There are three types of intervention for MGD: (1) over-the-counter (OTC) therapies, (2) anti-inflammatories, and (3) disease-modifying procedures. OTC therapies are primarily supportive in my opinion. Artificial tears, warm compresses, lid hygiene/massage, and lubricating ointments can help but are often limited in that patients are noncompliant, and these treatments are insufficient for addressing the underlying cause of advanced disease. I offer anti-inflammatory treatments such as topical corticosteroids or cyclosporine to patients who have an elevated tear osmolarity and/or matrix metalloproteinase 9.
Although steroids tend to improve inflammation relatively quickly, these agents are not suitable for long-term use. My preferred approaches to treating MGD are LipiFlow (TearScience) and intense pulsed-light therapy (off-label use of an FDA-approved device). In my experience, these procedures help to restore gland function and tear film health better than traditional OTC therapies. LipiFlow is frequently my primary intervention for presurgical patients, because it is well tolerated and, unlike intense pulsed-light therapy, can often produce results in 4 to 6 weeks, which allows patients to undergo surgery sooner.
CONCLUSION
Addressing MGD and DED before cataract surgery is important to achieving the best visual outcomes. Diagnostic and treatment decisions can be simplified and protocoled to allow rapid diagnosis and intervention. Excellent counseling is also vital to setting reasonable expectations for patients prior to a surgical procedure. n
1. Lemp MA, Crews LA, Bron AJ, et al. Distribution of aqueous-deficient and evaporative dry eye in a clinical-based patient cohort: a retrospective study. Cornea. 2012;31(5):472-428.
2. Trattler WB, Reilly CD, Goldberg DF, et al. Cataract and Dry Eye: Prospective Health Assessment of Cataract Patient Ocular Surface (PHACO) Study. Paper presented at: ASCRS Symposium and Congress; May 25-29, 2011; San Diego, CA.
3. Khanal S, Tomlinson A, Esakowitz L, et al. Changes in corneal sensitivity and tear physiology after phacoemulsification. Ophthalmic Physiol Opt. 2008;28(2):127-134.
4. Tutt R, Bradley A, Begley C, Thibos LN. Optical and visual impact of tear break-up in human eyes. Invest Ophthalmol Vis Sci. 2000;41:4117-4123.
5. Goto E, Yagi Y, Masumoto Y, Tsubota K. Impaired functional visual acuity of dry eye patients. Am J Ophthalmol. 2002;133:181-186.
6. Montés-Micó R. Role of the tear film in the optical quality of the human eye. J Cataract Refract Surg. 2007;33:1631-1635.
Preeya K. Gupta, MD
• assistant professor of ophthalmology at the Duke University Eye Center in the Division of Cornea and Refractive Surgery
• clinical director of the Duke Eye Center at Page Road in Durham, North Carolina
• (919) 660-5071; preeya.gupta@duke.edu
• consultant to Allergan, Biotissue, Shire, and TearScience


