


The cornea is the primary optical element, and it acts as a barrier to protect the internal components of the eye against mechanical damage, UV light, and infection. The cornea consists of the epithelium, stroma, endothelium, Bowman layer, and Descemet membrane (Figure 1).1 Corneal blindness affects many people worldwide, and the main limiting factor in corneal transplantation is a lack of donor tissue. Keratoprosthesis implantation involves removing the diseased cornea and replacing it with an artificial cornea, and serious complications such as corneal melting, glaucoma, prosthesis extrusion, and membrane formation can occur.2
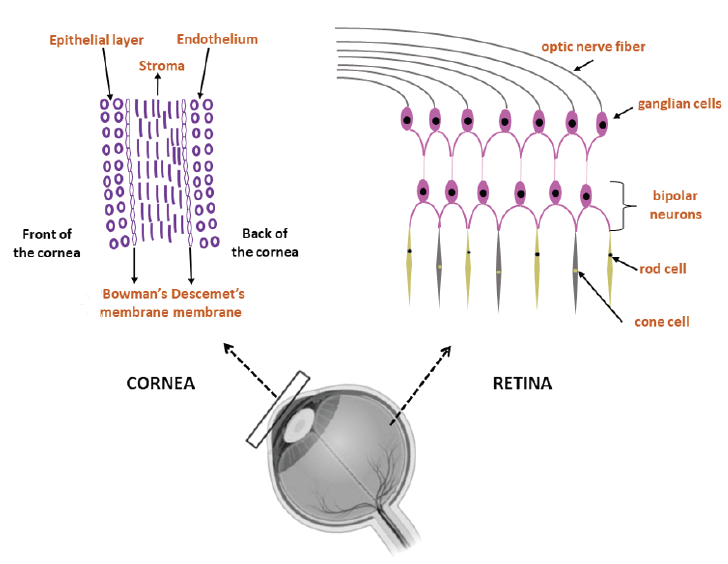
Figure 1. The schematic structures of the cornea and retina.
The retina collects light before transmitting information to the brain to provide images. In human retinas, cells with specific functions are embedded in the extracellular matrix (ECM), which consist of hyaluronic acid, a negatively charged polysaccharide. Photoreceptor (PR) cells located in the intermediate layers of the retina are responsible for light detection. Retinal disease that impedes the function of PR cells can cause blindness. Regenerating healthy PR cells in the eye is a promising strategy for treating retinal disease, but it is difficult to culture PR cells isolated in vitro because they undergo morphological changes and deep apoptosis without the support of ECM and other cell types.3
3D PRINTING: CATEGORIES AND TECHNIQUES
Researchers are investigating 3D printing for corneal and retinal tissue engineering. Approaches fall into three categories: polymer, cell, and hydrogel (Figure 2). The four techniques, described next, are depicted in Figure 3.
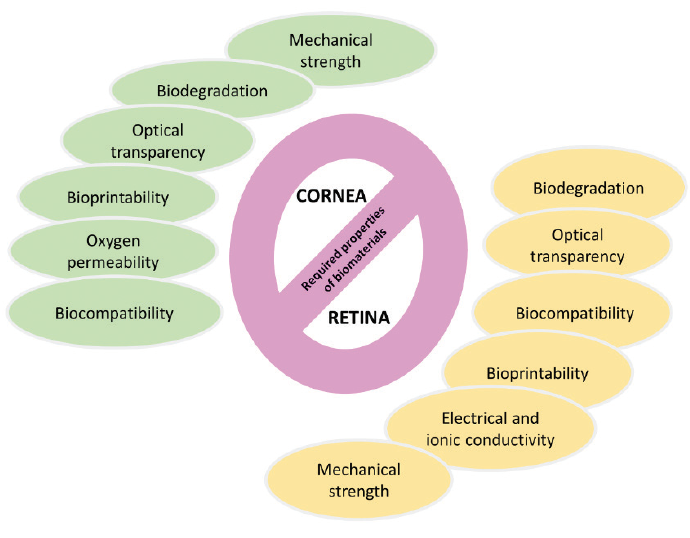
Figure 2. The required properties of the biomaterials for corneal and retinal tissue engineering.
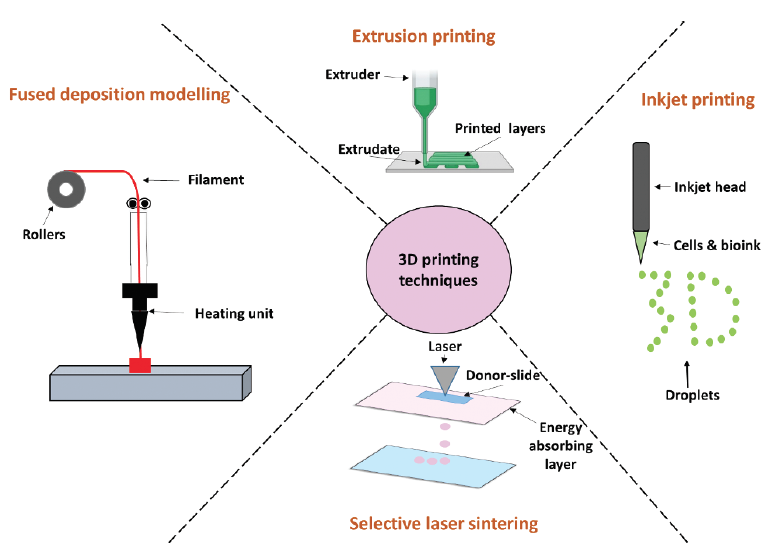
Figure 3. A schematic representation of 3D printing techniques.
Technique No. 1: Fused deposition modeling. A filament containing a thermoplastic polymer is heated until it reaches a semiliquid state. The materials are then extruded onto the panel to form layers.
Technique No. 2: Selective laser sintering. A laser is used to bind powder particles together instead of using sputtering, a process that ejects atoms from a source material to be deposited on a substrate. With selective laser sintering, laser energy etches a specific pattern onto the surface of the powder bed.
Technique No. 3: Inkjet printing. An inkjet head prints a liquid binder into fine layers of powder based on object profiles. Thermal and piezoelectric heads are used in inkjet systems. The application of electricity to the thermal head creates a vapor bubble over the resistor. In a piezoelectric system, the volumetric change in the fluid chamber is induced by applying voltage to the piezoelectric element.
Technique No. 4: Extrusion printing. Liquid resin is placed in a liquid reservoir. This approach is based on light curing, where a positionally programmed laser scans the resin surface to initiate photopolymerization. This hardens the resin and turns it from a liquid to a solid.4
3D BIOPRINTING
Several research teams have reported on their use of 3D printing technology for the creation of corneal and retinal tissue.
Corneal tissue. Let's review the results of seven studies.
No. 1. Sorkio et al fabricated corneal tissue using laser-assisted 3D bioprinting.5 These researchers bioprinted three types of corneal structures: corneal epithelium, lamellar corneal stromal layers with human adipose-derived stem cells, and a final structure with both a stromal and an epithelial part. Cells demonstrated good viability after printing.
No. 2. Bektas and Hasirci fabricated keratocyte-loaded, 3D-bioprinted gelatin methacrylate hydrogels to build corneal stroma.6 A live/dead viability assay demonstrated 98% cell viability on day 21.
No. 3. Kutlehria et al fabricated corneal stroma using 3D bioprinting.7 They combined sodium alginate, gelatin type B, and type 1 collagen solutions. A live/dead assay demonstrated that the corneal keratocytes maintained greater than 95% viability for 2 weeks.
No. 4. Campos et al combined collagen tip 1, agarose bioinks, and keratocytes to create corneal stroma.8 After 7 days of incubation, bioprinted keratocytes were viable and preserved their innate phenotypes.
No. 5. Our group created corneal stroma with a combination of polyvinyl alcohol and chitosan.9 Biocompatibility studies demonstrated that the constructs were compatible with human adipose-derived stem cells.
No. 6. Isaacson et al used sodium alginate, collagen type 1 bioinks, and corneal keratocytes to build corneal stroma.10 The keratocytes exhibited high cell viability (83%) at day 7.
No. 7. Mahdavi et al combined the gelatin methacrylate and stromal cells to produce corneal stroma.11 The constructs showed excellent substrate for stromal cell proliferation after 7 days in culture.
Retinal tissue. Let's review the results of three studies.
No. 1. Wang et al fabricated a multilayered tissue formed with hyaluronic acid-methacrylation by glycidyl-hydroxyl reaction to mimic native retinal tissue.12 Retinal pigment epithelium (RPE) was used in the culture to strengthen the differentiation. The investigators found that RPE provided the derivation of PRs from retinal progenitor cells.
No. 2. Shi et al built a retinal equivalent using a polycaprolactone ultrathin membrane, the cell monolayer ARPE-19, and a Y79 cell–laden alginate/pluronic bioink.13
No. 3. Masaeli et al fabricated a retina model composed of a layer of PR cells on top of a bioprinted RPE.14 Positioning the PR cells over the RPE did not alter the biological functions of the cells.
CONCLUSION
Research into the formation of transplantable 3D-printed cornea and retina holds great promise, but much engineering work remains to be done before this innovation in ophthalmology will truly disrupt patient care.
1. Sridhar MS. Anatomy of cornea and ocular surface. Indian J Ophthalmol. 2018;66(2):190-194.
2. Akpek EK, Alkharashi M, Hwang FS, Ng SM, Lindsley K. Artificial corneas versus donor corneas for repeat corneal transplants. Cochrane Database Syst Rev. 2014;11(11):CD009561.
3. Ma X, Qu X, Zhu W, et al. Deterministically patterned biomimetic human iPSC-derived hepatic model via rapid 3D bioprinting. Proc Natl Acad Sci U S A. 2016;113(8):2206-2211.
4. Ngoa TD, Kashania A, Imbalzanoa G, Nguyen KTQ, Hui D. Additive manufacturing (3D printing): a review of materials, methods, applications and challenges. Composites Part B: Engineering. 2018;143:172-196.
5. Sorkio A, Koch L, Koivusalo L, et al. Human stem cell based corneal tissue mimicking structures using laser-assisted 3D bioprinting and functional bioinks. Biomaterials. 2018;171:57-71.
6. Bektas CK, Hasirci V. Cell loaded GelMA: HEMA IPN hydrogels for corneal stroma engineering. J Mater Sci Mater Med. 2019;31(1):2.
7. Kutlehria S, Dinh TC, Bagde A, Patel N, Gebeyehu A, Singh M. High-throughput 3D bioprinting of corneal stromal equivalents. J Biomed Mater Res B Appl Biomater. 2020;108(7):2981-2994.
8. Duarte Campos DF, Rohde M, Ross M, et al. Corneal bioprinting utilizing collagen-based bioinks and primary human keratocytes. J Biomed Mater Res A. 2019;107(9):1945-1953.
9. Ulag S, Ilhan E, Sahin A, et al. 3D printed artificial cornea for corneal stromal transplantation. European Polymer Journal. 2020;133:109744.
10. Isaacson A, Swioklo S, Connon CJ. 3D bioprinting of a corneal stroma equivalent. Exp Eye Res. 2018;173:188-193.
11. Mahdavi SS, Abdekhodaie MJ, Kumar H, Mashayekhan S, Baradaran-Rafii A, Kim K. Stereolithography 3D bioprinting method for fabrication of human corneal stroma equivalent. Ann Biomed Eng. 2020;48(7):1955-1970.
12. Wang P, Li X, Zhu W, et al. 3D bioprinting of hydrogels for retina cell culturing. Bioprinting. 2018;11:e00029.
13. Shi P, Edgar TYS, Yeong WY, Laude A. Hybrid three-dimensional (3D) bioprinting of retina equivalent for ocular research. Int J Bioprint. 2017;3(2).
14. Masaeli E, Forster V, Picaud S, Karamali F, Nasr-Esfahani MH, Marquette C. Tissue engineering of retina through high resolution 3-dimensional inkjet bioprinting. Biofabrication. 2020;12(2):025006.




