Innovation in ophthalmology has brought forth new tools for treating an array of eye diseases. Historically, research into newer therapeutics for diseases affecting the front and back surfaces of the cornea has taken time, but that may begin to change in the next few years as current pipeline products progress through their development cycle.
An area of research attracting attention in corneal disease is the potential for creating therapies that target fibroblast growth factors (FGF), which are ubiquitous throughout the body and are involved in myriad biologic processes. Specifically, FGF1 has attracted interest due to its role in protecting and healing endothelial and epithelial cells of the cornea.
One company, Trefoil Therapeutics, has been developing an engineered form of FGF1 (TTHX1114) that has entered into in-human studies with an intracameral injection at the time of Descemet stripping only (DSO) procedures in patients with Fuchs endothelial cell dystrophy. In the STORM 2 trial, a phase 2 study, the control group received DSO alone. Group 2 underwent DSO after a priming dose of TTHX1114 that was delivered 3 days before the procedure, and then these patients received 4 weekly doses, given at the time of the procedure and for 3 weeks thereafter. Group 3 underwent DSO along with one high dose of TTHX1114 dosed at the time of the procedure. Group 4 received DSO plus a dose of TTHX1114 at the time of the procedure, then three subsequent weekly doses.
“The outcomes of the phase 2 STORM study (Figure 1) showed an accelerated improvement of vision and a resolution of corneal edema in response to the injection of the synthetic FGF1,” said Dr. Venkateswaran. She found it interesting that Group 3 had the most successful outcomes in the trial: gains in visual acuity at about 28 days post-injection and post-procedure, with continued improvement over the course of 2 months. This healing time was perhaps a little faster than with traditional DSO alone, but still longer than recovery times with DMEK.
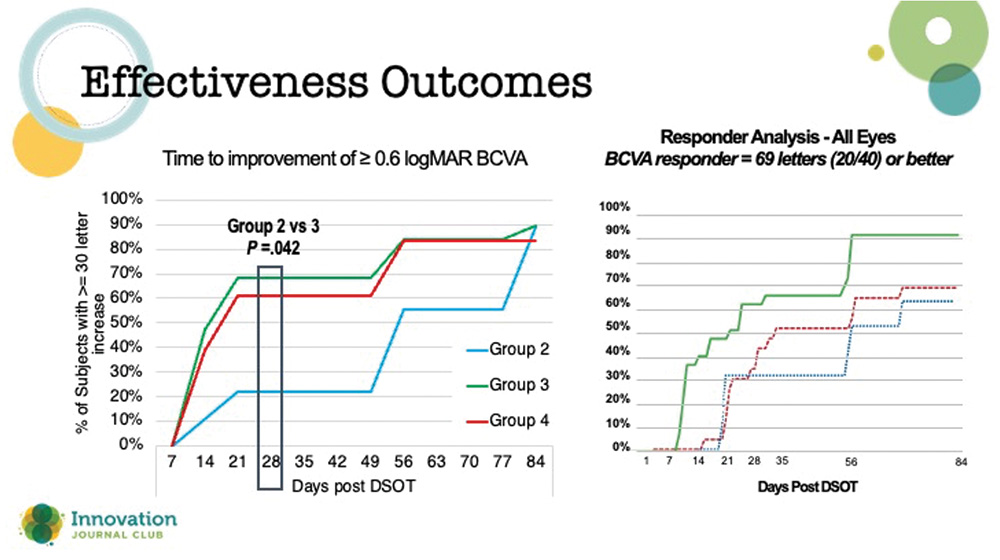
Figure 1. Effectiveness outcomes with TTHX1114 in the Storm 2 clinical trial.
Furthermore, 46% of the enrolled patients underwent cataract surgery concurrently with DSO, and the investigators found no difference in these patients’ recovery compared to those who received DSO only, and no differences in their pachymetry readings. Dr. Venkateswaran was encouraged by these findings. “Clinically, the potential of TTHX1114 is very relevant, because DSO is not always an isolated procedure in a pseudophakic patient,” she said. “Many of us are doing DSO at the time of cataract surgery, too. If we are able to accelerate healing time after cataract surgery in patients with endothelial disease such as Fuchs dystrophy, that's important.”
Dr. Venkateswaran stressed that the STORM studies are still ongoing, with data still needed on the long-term safety and efficacy profile of TTHX1114. Still, she sees its potential as an adjunctive therapy to traditional corneal surgeries. Although she prefers DMEK because of its expedited recovery for patients, there are some people who simply don’t do well with EK. “The compliance with drops, the positioning, the long-term effects of graft failure—graft longevity is limited, especially in eyes that have had multiple surgeries. If I can offer these patients DSO, plus start the process of endothelial recovery with a synthetic FGF1, maybe we can now have a more viable transplant-less option. It might open up that door for more patients.”

