As we surgeons continue to improve our outcomes, our enhancement rates continue to decline. As a result, unfortunately, when patients require an enhancement, they consider it a failure of the procedure, because they expect that we should never have to intervene a second time. When we do bring a patient back, therefore, we must thoroughly understand what it is that got us here in the first place.
—Karl G. Stonecipher, MD, section editor
CASE PRESENTATION
A 46-year-old woman returns after an uneventful topography-guided LASIK procedure (Figures 1-3). The patient complains of blurred vision, problems when driving at night, and challenges on cloudy days. Her UCVA measures 20/30 OD, and her BCVA is 20/20 OD. The UCVA of her left eye is 20/15. The current manifest refraction is plano +0.75 × 018, which is consistent with the WaveLight Vario Topolyzer (Alcon) image taken postoperatively (Figure 4). The examination is otherwise normal. Pachymetry readings are 519 µm at the thinnest point, and the residual stromal bed is 412 µm in the right eye.
How would you enhance this result?
—Case prepared by Karl G. Stonecipher, MD.
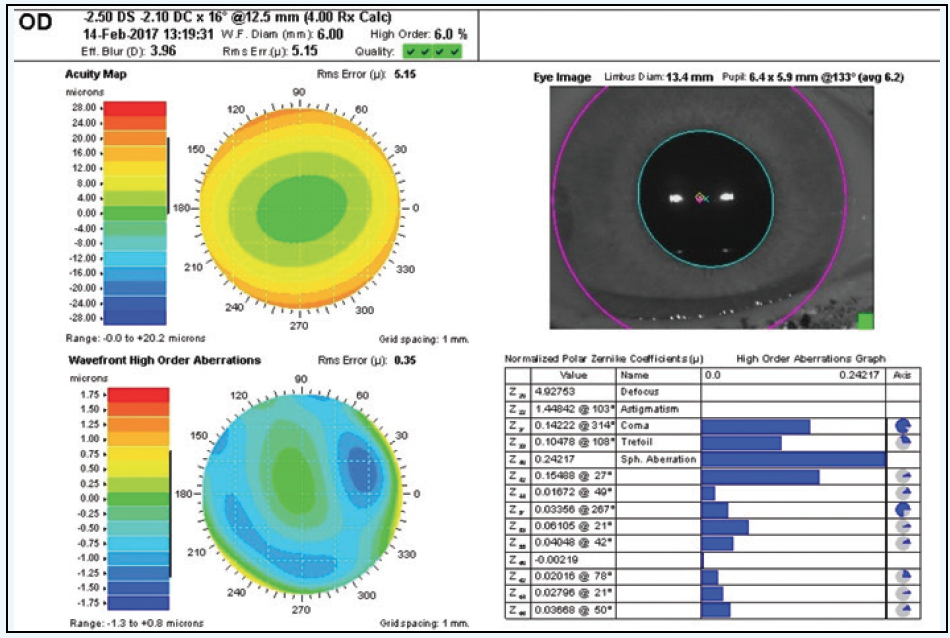
Figure 1. Preoperative examination prior to LASIK.
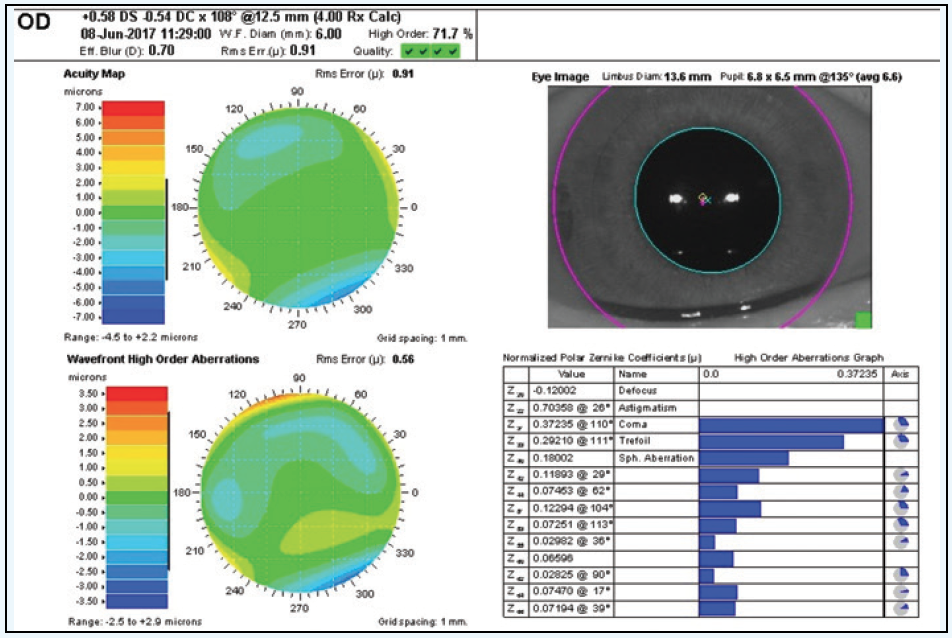
Figure 2. Examination after topography-guided LASIK.
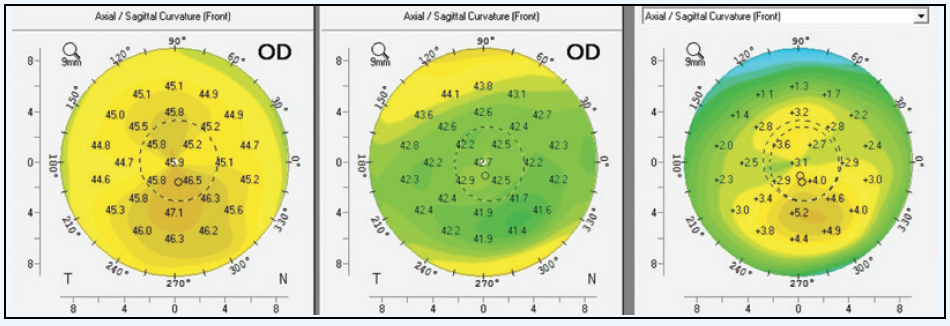
Figure 3. Pre- and postoperative Pentacam (Oculus Optikgeräte) measurements with difference map after topography-guided LASIK.
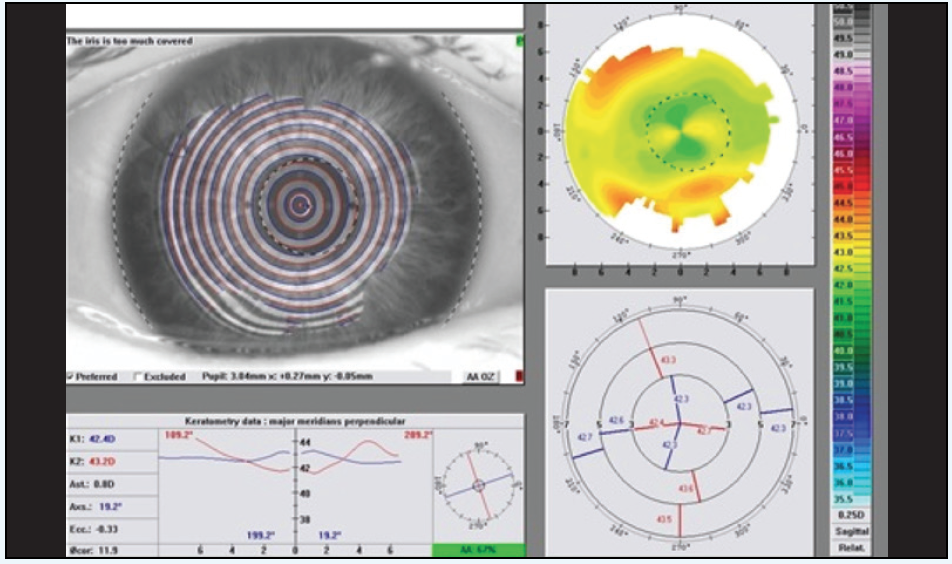
Figure 4. Postoperative measurements with the WaveLight Vario Topolyzer.

JERRY TAN, MBBS(S’pore), FRCS(Edin), FRCOphth, FAMS
This is an interesting case. The preoperative examination shows asymmetrical astigmatism, which can be thought of as a combination of symmetrical with-the-rule (WTR) astigmatism and vertical coma (Figure 5).
Topography-guided treatment will correct both corneal astigmatism and corneal coma, but the refractive power of the cylinder obtained by the optometrist or surgeon will be overestimated on manifest as well as cycloplegic refraction. The reason is that, in the trial lens set, we only have cylinder, and there are no coma-correcting lenses! By overcorrecting cylinder, the refractionist is compensating for the coma by using cylinder. The patient also accepts this, because there is no other way to correct for the coma using a trial lens set. For example, if we have 2.00 D WTR cylinder on refraction, the coma will contribute some of the WTR astigmatism in an eye that has asymmetrical astigmatism.
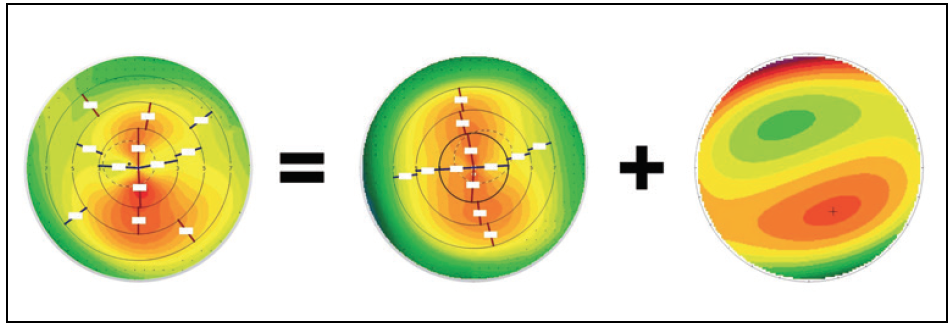
(Courtesy of Jerry Tan, MBBS(S’pore), FRCS(Edin), FRCOphth, FAMS.)
Figure 5. Asymmetrical astigmatism can be thought of as symmetrical astigmatism plus coma.If the surgeon enters the manifest or cycloplegic refraction cylinder, the patient’s topography-guided treatment will overcorrect the cylinder, which will rotate the axis of the astigmatism 90º from the original axis. This is what happened to this patient.
To prevent this problem, we must correlate the following parameters:
1. anterior corneal astigmatism within the central 3 mm of the cornea
2. the posterior corneal astigmatism within the central 3-mm zone
3. the magnitude of the coma in diopters (some topography/excimer laser software converts the measurement of coma in microns to diopters, which makes the dioptric calculation of cylinder easier [see Summary of Aberrations])
For example, if the manifest refraction shows -2.00 D cylinder at 180º and the corneal topography shows 2.00 D of WTR cylinder in the central 3-mm zone, the posterior corneal astigmatism is 0.50 D against the rule, and there is 0.50 D vertical coma, then the treatment entered into the laser should be -2.00 D (anterior corneal astigmatism) + (+0.50 D) (posterior corneal astigmatism) + 0.50 D (vertical coma in diopters) = -1.00 D (input of correction of astigmatism programmed into the laser).
Summary of Aberrations
The Schwind Amaris aberration summary (Schwind Eye-tech-solutions) converts coma microns into diopters.
- Summary of aberrations @ OZ = 7.00 mm
- Coma: 0.52 μm/0.48 D @ 234º
- Trefoil: 0.36 μm/0.11 D @ 33º
- SphAb: 0.58 μm/0.42 D
Abbreviations: OZ, optical zone; SphAb, spherical aberration.
If we do not compensate for the coma correction, then we will get an overcorrection and change the axis of the cylinder by 90º. The coma is now gone (eliminated by the topography-guided treatment), so what is left behind is an overcorrected cylinder.
This overcorrected cylinder can easily be corrected with a wavefront-optimized program, because there is no more coma in any direction after a topography-guided treatment. We can still use another topography-guided treatment, but it is not really necessary. In this example, we could therefore perform a wavefront-optimized correction of the patient’s residual astigmatism. If we use a topography-guided treatment, then we should set the Q value at zero for T-CAT treatments (Alcon WaveLight) or filter out spherical aberration correction in a Schwind Amaris treatment (Schwind Eye-tech-solutions), which is the same as a target Q value of zero.

A. JOHN KANELLOPOULOS, MD
My colleagues and I have presented and reported some of the first topography-guided data,1 and over the years, I have found some points to be pivotal when using topography-guided ablations.
Point No. 1. Validating Topography Measurements. Current topography-guided treatment data available in the United States derive only from anterior corneal curvature mapping using the WaveLight Vario Topolyzer. (Outside the United States, Scheimpflug tomography total cornea-driven data are available as well [Oculyzer II; Alcon].) Anterior curvature-derived topography-guided data appear to be OK for most patients, because the posterior corneal surface has a very low power anyway (about -6.00 D), so slight changes of the posterior axis and amount are essentially negligible. In the rare case that there is a significant posterior corneal curvature deviation from the norm (such as an otherwise completely asymptomatic case of Descemet split from injury during forceps-assisted childbirth or a Descemet stripping automated endothelial keratoplasty case), posterior curvature data should be taken into account instead of using only anterior data. If Scheimpflug-driven (Oculyzer II) topography-guided correction with the Alcon WaveLight platform is not available (these also include posterior corneal data in the treatment calculation), I recommend comparing the proposed topography-guided data to wavefront-guided data. If they are significantly different, I would suggest a wavefront-optimized treatment or even a wavefront-guided treatment.2 If the patient had any visual complains postoperatively, these could be addressed with a customized enhancement (wavefront-guided, topography-guided, ray-tracing, etc.).
No. 2. Delivery of Treatment Data. Pupillary tracking and cyclorotation adjustment are pivotal to achieving the desired correction with an excimer laser.3 Once topography-guided treatment is engaged, the stakes are even higher, because the treatment is quite irregular, and in most cases, the inaccurate delivery of laser energy can increase instead of decrease coma and trefoil.
My impression is that this exact principle caused the problem in the case presented herein. By my rough estimate looking at the preoperative topography, the preoperative topographic cylindrical axis is just a little over 90º; the difference map on the same comparative topographic image series shows that the cylinder corrected (this is the cylinder axis of the difference map preoperatively minus postoperatively) was close to 115º (Figure 6). Considering that every 2º deviation deriving from cyclorotation reduces cylindrical correction by 8%, the deviation here appears to be huge.
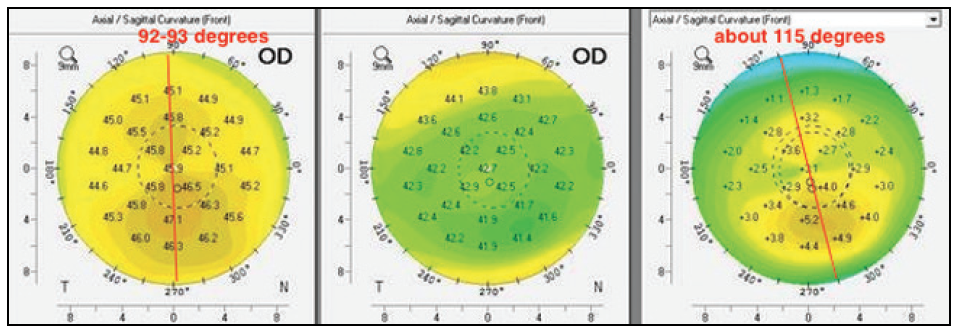
Figure 6. Dr. Kanellopoulos notes that the difference in axes between the pre- and postoperative maps suggests that cyclorotation was not compensated for in this case.
My colleagues and I have systematically studied and reported on this principle on two separate occasions: (1) when comparing topography-guided normalization of irregular corneas with and without cyclorotation compensation and (2) when comparing topography-guided LASIK with and without topographic manifest refraction adjustment4 (topography-modified refraction) and to small-incision lenticule extraction5 that, at the time, had no cyclorotation compensation available. The delta angle between the attempted to corrected was, in our opinion, the catalyst in the outcomes difference. If no cyclorotation data are available intraoperatively, perhaps abort a Contoura Vision (Alcon) and topographic manifest refraction treatment, and instead perform a wavefront-optimized procedure with the clinical refraction.
It seems that our work has changed surgeons’ understanding of refraction, as for experienced topography-guided users, even “normal” astigmatism in the presence of angle kappa over 100 µm (which seems to be common not just in hyperopes6) creates significant coma for a patient that appears to be practically neutralized via accommodation and/or the patient’s choosing on subjective refraction cylinder of a different axis. When I perform wavefront-optimized treatments, I leave the subjective refraction unchanged—a time-proven principle for all of us. Nevertheless, we have noticed that, years later, cylinder “changes” may manifest in the subjective refraction of many patients as “regression.” Our findings support that topography-guided LASIK with topographic modified refraction may be the most accurate refractive correction by optimizing the cornea for the functional visual pathway of each patient’s eye, but its success may depend heavily on the patient’s positioning and the accuracy of measurement, registration, and delivery. Small biases may affect the result. We have found and reported that the benefits in straightforward cases can be 1 to 2 lines better distance UCVA!
These findings seem to have ignited a strong interest in topography-guided corrections of irregular and now even regular eyes, as demonstrated by the large number of prominent refractive surgeons participating in our all-day workshops on topography-guided treatments (Topography University) held during the past year 1 day prior to all major ophthalmic meetings. More information may be found at www.topo-guided.com. Both Drs. Tan and Stonecipher have graciously offered expert tutoring at these events.

WHAT I DID: KARL G. STONECIPHER, MD
In light of the increased root mean square higher order, which was mainly coma and minimal residual refractive error, I lifted the flap and performed an Advanced CustomVue LASIK enhancement (Figure 2). I made no physician adjustments to the treatment. The patient’s postoperative refraction at present is +0.25 +0.25 × 025, with a BCVA of 20/20 and a UCVA of 20/25. The results are early, but the patient was doing well postoperatively at her last visit.
1. Kanellopoulos AJ. Topography-guided custom retreatments in 27 symptomatic eyes. J Refract Surg. 2005;21(5):S513-518.
2. Kanellopoulos AJ, Pe LH. Wavefront-guided enhancements using the WaveLight excimer laser in symptomatic eyes previously treated with LASIK. J Refract Surg. 2006;22(4):345-349.
3. Kanellopoulos AJ, Asimellis G. Novel Placido-derived topography-guided excimer corneal normalization with cyclorotation adjustment: enhanced Athens protocol for keratoconus. J Refract Surg. 2015;31(11):768-773.
4. Kanellopoulos AJ. Topography-modified refraction (TMR): adjustment of treated cylinder amount and axis to the topography versus standard clinical refraction in myopic topography-guided LASIK. Clin Ophthalmol. 2016;10:2213-2221.
5. Kanellopoulos AJ. Topography-guided LASIK versus small incision lenticule extraction (SMILE) for myopia and myopic astigmatism: a randomized, prospective, contralateral eye study. J Refract Surg. 2017;33(5):306-312.
6. Basmak H, Sahin A, Yildirim N, et al. Measurement of angle kappa with synoptophore and Orbscan II in a normal population. J Refract Surg. 2007;23(5):456-460.