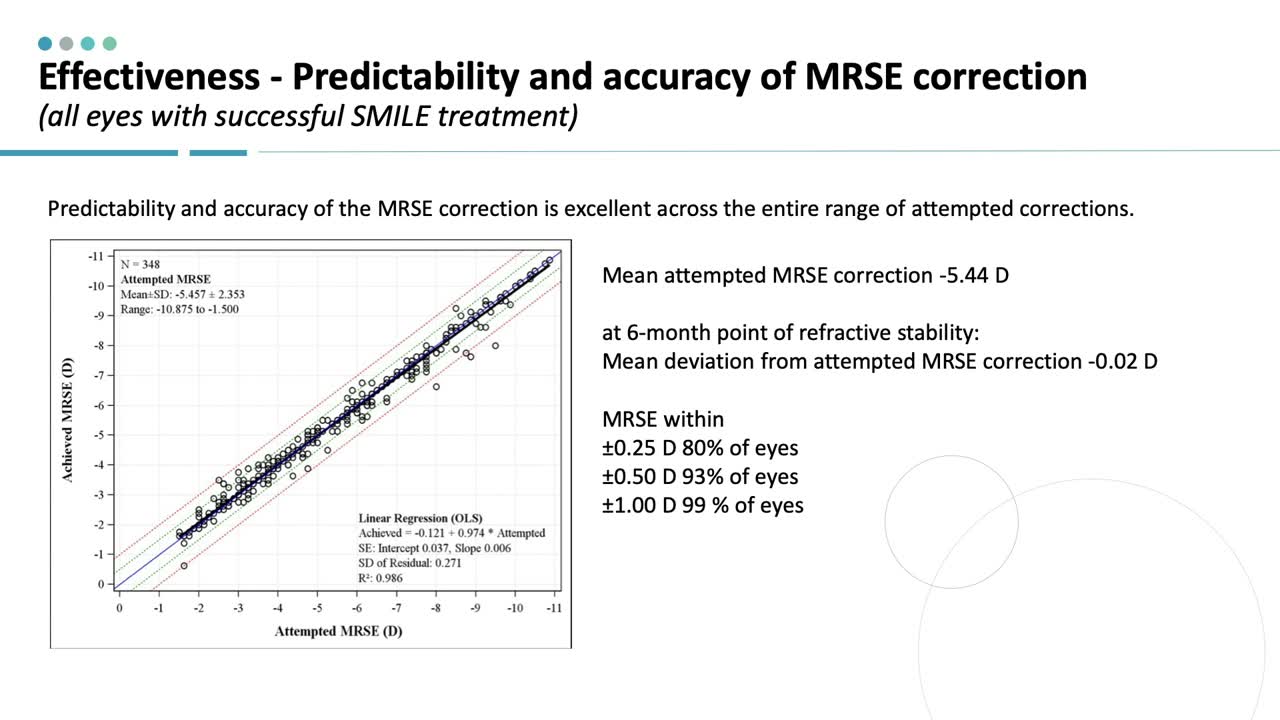
The ReLEx SMILE procedure first gained US Food and Drug Administration (FDA) approval for the reduction or elimination of myopia ≥ -1.00 D to ≤ -8.00 D, with ≤ -0.50 D cylinder and Manifest Refraction Spherical Equivalent (MRSE) ≤ -8.25 D in September 2016. This indication was later expanded to include eyes with myopia and astigmatism with sphere: -1.00 D to -10.00 D, cylinder between -0.75 D and -3.00 D, and MRSE: -10.00 D. The latter approval was based on an analysis of 307 spherocylindrical procedures; baseline mean MRSE was -5.41 D (range, -10.88 to -1.5), which may be a higher average than clinicians typically treat with LASIK, and baseline cylinder was a mean -1.52 D (range, -0.75 to -3.00). Average age was 33 years, and 60% of patients were male (Figure 1).
Figure 1.
Similar to findings in the study of sphere-only correction, the predictability and accuracy of MRSE correction was consistent across the range of myopia, with 93% of patients overall achieving ±0.5 D of the targeted MRSE correction. Correspondingly, uncorrected visual acuity results were very good, with 84% of patients achieving 20/20 or better 6 months after surgery.
SMILE vs LASIK: The Best Apples-to-Apples Comparison of Data We Have to Date
Since its approval, one of the questions regarding SMILE is how postoperative outcomes compare with LASIK, particularly spherocylindrical SMILE versus traditional laser vision correction. One way we may begin to address this question is to compare premarket approval (PMA) data from SMILE procedures with those from recent FDA applications for advanced forms of LASIK to correct myopia and astigmatism. It could be argued that such an analysis provides the best apples-to-apples comparison we have at this point, as the studies were carefully conducted, controlled, and monitored. In addition, the procedures were all performed before aftermarket refinements in technique and thus represents “out of the box” outcomes.
We recently reviewed the PMA data for SMILE and compared it with data for the STAR S4 IR Excimer Laser System iDesign Advanced WaveScan Studio System (Johnson & Johnson Vision), Allegretto Wave Eye-Q Excimer Laser (Alcon), and the EC-5000 Excimer Laser System (Nidek).1-4 Not unexpectedly, we found that the reported rates for patients achieving 20/40 or better or 20/20 or better VA at 6 or 12 months were comparable (Figure 2). Of note, the outcomes in sphere-only SMILE and spherocylindrical SMILE procedures were virtually the same.
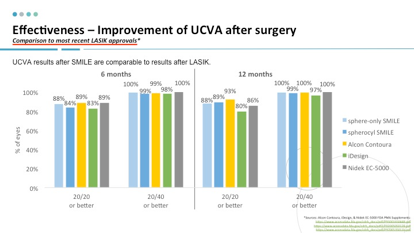
Figure 2.
We also compared the predictability of the MRSE and astigmatic correction. The percentage of patients within ±0.50 D of the intended MRSE correction 6 months postoperatively was 93%, 94%, 93%, 91%, and 69% for sphere-only SMILE, spherocylindrical SMILE, Alcon Contoura, EC-5000, and iDesign procedures, respectively. The percentage of patients within ±0.50 D of the intended MRSE correction 6-months postoperatively was 88%, 90%, 91%, and 85% for spherocylindrical SMILE, Alcon Contoura, EC-5000, and iDesign procedures, respectively.
Conclusion
With the FDA approval of SMILE for myopia and astigmatism in eyes with -1.00 D to -10.00 D sphere and cylinder up to -3.00 D, the range of approved treatments with this technology will encompass over 80% of the population seeking vision correction surgery. Data showing that postoperative outcomes with SMILE are comparable with those with LASIK should provide extra confidence in the predictability and accuracy of astigmatic correction with this procedure. In the future, it is expected that technique optimization, including optimizing energy levels, will help to further refine the ability to accurately, predictably, and reproducibly to correct astigmatism and myopia.
1. US Food and Drug Administration. Summary Of Safety And Effectiveness Data (SSED): VisuMax Femtosecond Laser – P150040/S003. Available at: https://www.accessdata.fda.gov/cdrh_docs/pdf15/p150040s003b.pdf. Accessed January 9, 2019.
2. US Food and Drug Administration. Summary Of Safety And Effectiveness Data (SSED): STAR S4 IR Excimer Laser System iDesign Advanced WaveScan Studio System. Available at: https://www.accessdata.fda.gov/cdrh_docs/pdf/P930016S044B.pdf. Accessed January 9, 2019.
3. US Food and Drug Administration. Summary Of Safety And Effectiveness Data (SSED): ALLEGRETTO WAVE Eye-Q Excimer Laser Available at: https://www.accessdata.fda.gov/cdrh_docs/pdf2/P020050S012B.pdf. Accessed January 9, 2019.
4. US Food and Drug Administration. Summary of Safety And Effectiveness Data (SSED): Nidek EC-5000 Excimer Laser System. Available at: https://www.accessdata.fda.gov/cdrh_docs/pdf/P970053S011b.pdf. Accessed January 9, 2019.