Sponsored by
Allergan Global Medical Affairs








DED affects millions of our patients, and can be both detrimental to refractive surgical outcomes and debilitating for patients. As understanding of the underlying pathology of DED has evolved, so has the way in which we treat it. Our experience with and perspective on older therapies interplays into our current and future practice patterns. We look at past evolution to predict the future in many disciplines, and that is just as true with ophthalmology and DED. A group of exceptional physicians recently gathered together to review the therapeutic evolution and make predictions about the future treatment of a disease that is impacting a growing number of our patients.
DIAGNOSIS
John Sheppard, MD: The definition of DED is evolving in part due to the increased ability to diagnose it, the result of so many new technologies on the market. Patient histories now frequently include dry eye questionnaires, and much more attention is paid to meibomian glands at the slit lamp. Let us discuss the utility of the wide variety of diagnostic technologies available including tear osmolarity, meibography, matrix metalloproteinase 9 (MMP-9) testing, lissamine staining, Schirmer tests, and tear break-up time.
Karl Stonecipher, MD: The first step in treating dry eye is to diagnose it. If you are not picking up some form of DED in one out of every five patients, you are missing the boat because, in my experience, at least 20% of patients will have some form of DED.
Eric Donnenfeld, MD: There is no definitive test for dry eye. All of the tests are helpful and a combination of clinical history, diagnostic testing, and point-of-service testing are all used to make the diagnosis of dry eye. We should resist any attempt to narrow down DED to a single diagnostic that qualifies a patient for treatment or not. Just as one glaucoma test does not diagnose or evaluate glaucoma, we need to do a better job of understanding and managing the complexity of DED. There are companies working on software that will integrate all of these test results and show progression of disease, which will allow us to make more intelligent decisions about management.
Dr. Sheppard: The glaucoma analogy is very important. A patient with high pressure and no optic nerve damage is very fortunate to be in your care, just like a patient with dry eye and punctate keratopathy with normal osmolarity is fortunate to be getting care. We have seen over and over again that catching any disease early leads to better outcomes. With so many tests out there, it is tempting to try and narrow it down to just one or two that are the most useful. It seems overwhelming to invest in and understand so many new technologies for one disease state. However, I believe all of them are useful. Each patient experiences dry eye differently, and each test increases your understanding of the pathophysiology of that patient.
Elizabeth Yeu, MD: We used to just drop fluorescein in the eye and, if they did not stain, then they were not dry. Our approach is very different now. What we know about the meibomian glands has really changed the way I perform a slit-lamp examination. I compress the gland, looking at the egress and the quality of the meibum. I never looked at telangiectasis or lid margin pitting as much before to connect to the severity of posterior lid margin disease, but these are important examination considerations. It is also important to understand mechanical causes such as conjunctival chalasis, corneal epithelial dystrophy and degenerations, as well as the level of the tear film meniscus.
Preeya Gupta, MD: Certainly, if someone has a positive MMP-9 test, I am much more apt to put them on an antiinflammatory regimen. However, the ocular surface is very complex and MMP-9 is farther downstream in the inflammatory pathway. To only treat patients who have a positive MMP-9 test would be restricting treatment to more severe patients, and there is a huge subset of patients that potentially will never become moderate or severe DED sufferers if we treat them earlier.
Dr. Donnenfeld: Osmolarity is not a static number, and the loss of stability or homeostasis is a cardinal sign of DED. A significant difference between two eyes is just as important as a high osmolarity score for diagnosing DED.
Dr. Yeu: A normal tear osmolarity score does not necessarily mean that a patient does not have DED. It can, oftentimes, mean they have a good compensatory mechanism that allows them to have a lower basal osmolarity score. That, in itself, is actually very telling in someone you know is dry by their symptoms.
Jodi Luchs, MD: A patient with a tear osmolarity score below 300 may still have DED and may still respond very well to therapy. We have to be more thorough than a single test.
Dr. Gupta: There are so many excellent tests, but some of them have more utility for screening and others have greater utility for tracking disease. Sometimes eye care providers see that list and get overwhelmed at the thought of integrating so many testing modalities into practice. The important message is to pick one or two options for screening disease, and then pick one or two for following the disease state, and start there. It is essential that as a profession we raise the bar for diagnosing and managing dry eye.
The Homeostatic Tear Film
The tear film is made up of three primary layers. The mucin layer is produced by goblet cells in the conjunctiva and is essential for tear stability. The aqueous layer is produced by the lacrimal glands and contains a complex mixture of proteins, including T-cells. The lipid layer is produced by the meibomian gland and helps to lubricate the eye by restricting evaporation. Understanding the tear film is essential to treating it. While T-cells are frequently considered negative, an indicator of inflammation, regulatory T-cells actually help inhibit a variety of autoimmune and inflammatory diseases (Figure 1).2 When the tear film’s delicate homeostatic balance is disturbed, a stress response is triggered that activates a perpetuating cycle of inflammation. It is now well understood that treating inflammation is at the root of treating DED.3,4
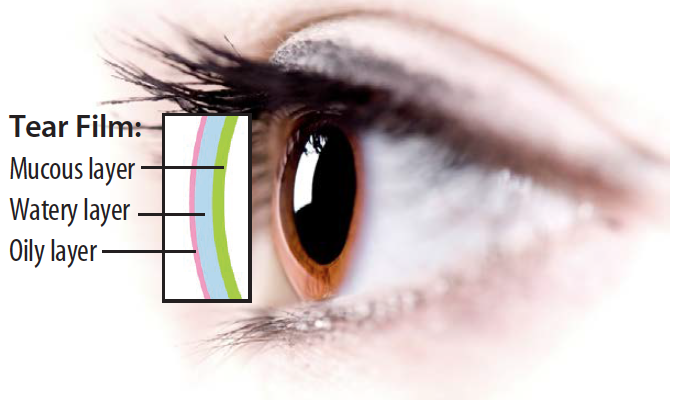
Figure 1. The aqueous layer of the tear film contains important regulatory T-cells that inhibit autoimmune and inflammatory diseases.1
Treating the Cause of Disease
Dr. Sheppard: As the most widely used antiinflammatory agent, corticosteroids are frequently prescribed for induction and pulse therapy for DED.5 While not approved by the FDA specifically for the treatment of DED, Pred Forte (prednisolone acetate; Allergan), FML Forte (fluorometholone; Allergan), and then Lotemax (loteprednol; Bausch & Lomb) changed the way eye care providers treated anterior segment disease. Steroids decrease inflammation by inhibiting T-cells on the ocular surface, inhibiting ICAM-mediated vascular diapedesis, and by inducing proteins that control the biosynthesis of inflammatory mediators. An effective, broad-spectrum therapy, corticosteroids continue to be an important tool in fighting DED. However, the known drawbacks to steroids left a need to search for other antiinflammatory treatments. Thomas Starzl, MD, revolutionized transplant surgery in the 1980s with cyclosporine, a peptide that suppresses the immune system (Sandimmune; Sandoz Pharmaceuticals).6 It was later found to be useful systemically for uveitis as well as severe corneal and scleral inflammatory diseases.7-9 Topical cyclosporine was also compounded by specialty pharmacies, usually in 1% to 4% concentrations, for numerous challenging ocular surface disorders.10 In 2002, the ophthalmic emulsion Restasis (cyclosporine A 0.05% ophthalmic emulsion; Allergan) was approved by the FDA to become the foundation of dry eye therapy (Figure 2).
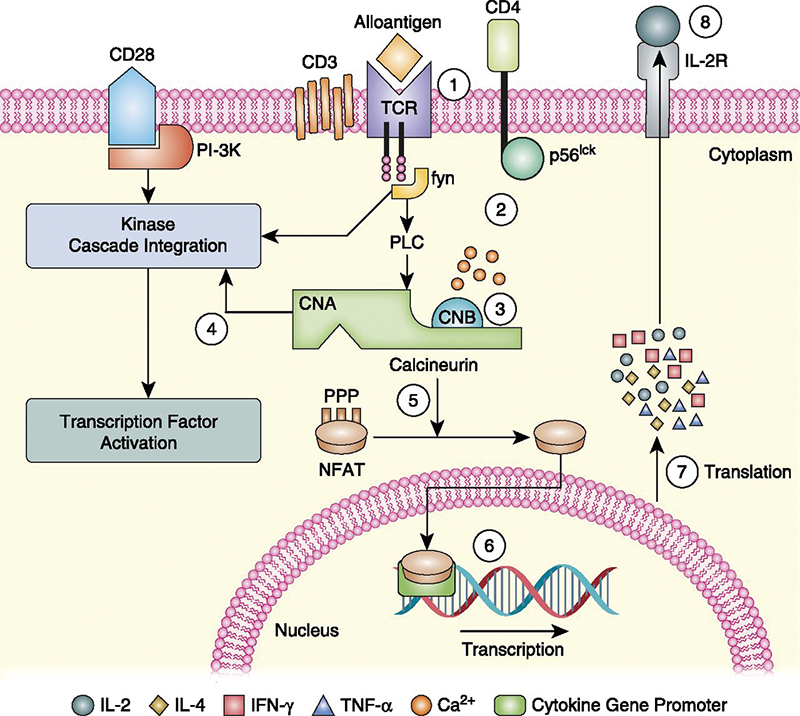
(Courtesy of Michael Cooper, MD)
Figure 2. The calcineurin inhibitor, T-cell, and cyclosporine A tacrolimus pathway.Those of us who have been around a long time remember the process of educating the public about ocular surface disease and dry eye being an inflammatory process. The academic cornea specialists were the first to understand the role of inflammation and the value of the new therapeutic. Because the vast majority of these physicians were treating advanced stages of DED, Restasis was initially used almost exclusively for moderate to severe DED. After 13 years of proven safety and increased understanding of DED, the indication for Restasis has changed considerably.
Dr. Donnenfeld: The 13 years of clinical studies and the safety record of Restasis have really transformed its indication.11 An early fear among practitioners had to do with the potential systemic absorption and ensuing negative side effects. However, with my colleagues we figured out how long a patient would need to use Restasis twice a day to reach the level of systemic absorption of a single oral therapy. The answer is 1,996 years. Confidence in the safety of the drug has really enabled the evolution of its use.
Dr. Luchs: Ten years ago, I was hesitant to prescribe Restasis to young contact lens-wearing patients. I was unsure of the long-term consequences. Now I do not hesitate. In my experience, it will help and it is safe.
William Trattler, MD: In the very early days after Restasis approval, we were told that it would not work in patients with punctal plugs. But based on work done by Cal Roberts, MD, and others, we now know that there is a synergistic affinity between plugs and Restasis.12 I feel very comfortable prescribing Restasis in patients with plugs despite what it says on the label.
Dr. Donnenfeld: Once we gained confidence in the safety of Restasis, the doors really opened to maximizing its therapeutic value. One of the really unmet needs that Restasis answered was the use of dry eye therapy before cataract and corneal surgery. We never considered using it before LASIK or cataract surgery 15 years ago, and when I started to do that, everyone thought I was crazy. Now, use of Restasis preventatively to counteract the negative effect of surgery and improve quality of vision is a mainstay in therapy (Figure 3).
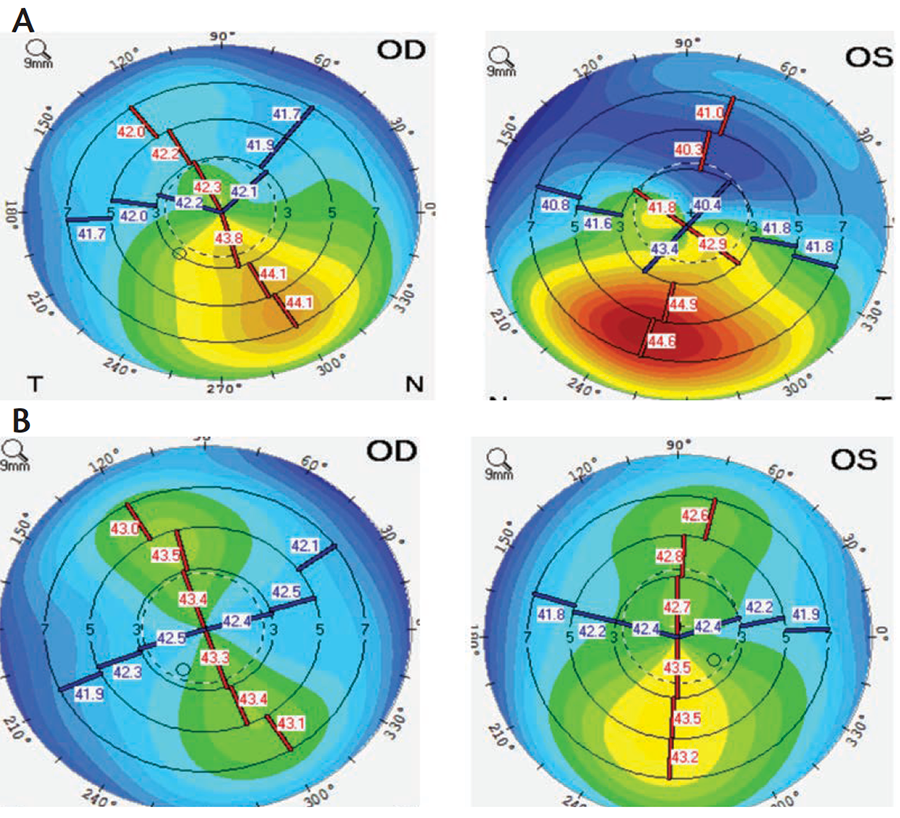
(Courtesy of William Trattler, MD)
Figure 3. Tomography performed during refractive surgery consultation at baseline (A) versus after 2.5 months of treatment with antiinflammatory drops and plugs (B).Dr. Yeu: Practice patterns in regards to DED have certainly changed. The switch from using Restasis reactively to where we are using it proactively, such as with perioperative patients, has dramatically empowered surgeons to improve their results and their patients’ satisfaction.
Dr. Donnenfeld: Another major change has been the adoption of Restasis in the optometric community. Optometrists have been very successful in using Restasis to make their patients more tolerant of contact lenses.
Dr. Sheppard: Do you think steroids and cyclosporine are synergistic and, if so, do you use them simultaneously?
Dr. Yeu: Yes. Similar to how steroids and nonsteroidal antiinflammatory drugs are synergistic postoperatively for inflammation.
Dr. Trattler: Definitely. And if I am going to use both, I am going to start them at the same time.
Dr. Luchs: Most often, I will use them together. However, if an eye is extremely uncomfortable, anything you put on that eye is going to burn. My goal is to make the eye feel better first so that the patient is more likely to tolerate and continue therapy with cyclosporine down the line.
Dr. Yeu: I think compliance with cyclosporine is significantly improved when you have an eye that is quieter than when you have an eye that is riddled with marks from epitheliopathy, which exposes the subbasal corneal nerve plexus, and can magnify the discomfort that can be associated with various topical drop therapies.
Dr. Sheppard: In 2016, the topical LFA inhibitor Xiidra (lifitegrast; Shire Pharmaceuticals) was approved for the treatment of the signs and symptoms of DED, bringing forward a totally different mechanism of action for addressing inflammation. Lifitegrast effectively blocks the interaction between LFA-1 and ICAM-1 and the subsequent cycle of T-cell-mediated inflammation. Clinically effective LFA-1 inhibition also has systemic origins. Efalizumab (Raptiva; Genentech) is a humanized monoclonal antibody that prevented LFA-1–expressing lymphocytes from interacting with ICAM ligands, and was approved systemically in 2003 for the treatment of plaque psoriasis. However, in 2009 efalizumab was voluntarily removed from the market due to an association with progressive multifocal leukocephalopathy, a rare brain infection caused by the JC polyomavirus. The virus is carried by more than 50% of the population, but is kept in check by T-cells.13
Lifitegrast was designed to be used only on the ocular surface. It has a similar pH to the ocular surface and is suspended in an aqueous solution with a stable shelf life. Although lifitegrast and efalizumab are both LFA-1 inhibitors, lifitegrast is a unique molecule and not the same molecule as efalizumab (Raptiva). The safety and efficacy of Xiidra has been evaluated in over 1,000 patients in 4 randomized studies, including a 12-month safety study.14 After 12 weeks, Xiidra demonstrated significant improvement in signs and symptoms of DED compared to controls, and seems to be achieving rapid success in the market. As our diagnosis of dry eye becomes more differential, so should our choice of therapy. We now must ask ourselves questions such as: How important are T-cells? When do you need a steroid to wipe the slate clean in terms of inflammatory markers? If you are using a T-cell inhibitor, does that restore the tear film back to its natural state, or will those cytokines return?
Dr. Donnenfeld: T-cells are everywhere in the body as part of the immunologic landscape. T-cell helpers have to be in balance with T-cell suppressors, and when you lose that balance is when you develop pathology. That is what is happening with dry eye. There is an imbalance in the T-cell regulation process.
Dr. Yeu: The aqueous concentrations of Xiidra are very low and do not appear to confer to systemic levels of absorption. The 1-year safety studies demonstrated detected lifitegrast at the Day 365 serology check, but the amount was not cumulative and was too low for quantifiable detection.15 However, cyclosporine has been more widely used in young adults and children so I would feel much more comfortable prescribing Restasis to this population.16 Xiidra is still untested here.
Dr. Sheppard: Looking at activation markers, Restasis restores the normal ocular surface. Other agents remove inflammatory targets nonspecifically. We know that steroids have a plethora of mechanisms of action, so should this knowledge affect our approach to dry eye?
Dr. Luchs: It is always better to have a more targeted bullet to combat the diseased pathway without collateral damage. It is much better if we can use a targeted therapy for the mechanism that is at the heart of the pathophysiology rather than a broad spectrum antiinflammatory, but patients must be educated that, even though they are being successfully treated, they may have flare-up symptomatology that will have to be addressed with another form of therapy. This does not mean that their current therapy is not working.
Dr. Yeu: Acuity and severity are two key components of figuring out what kind of antiinflammatory to start with. For an acute, severe dry eye patient, I like to start with steroids and cyclosporine. The steroids calm the eye quickly, while the cyclosporine works to restore immunologic balance in the eye.
Dr. Gupta: We should be empowering the larger audience to use steroids for DED as needed, but I think you cannot overstate the need to be cautious when using them long term. I prefer to choose a nonsteroid-based antiinflammatory medication as a sustainable base therapy and pair it with a limited course of steroids or use them as pulsed therapy only as necessary.
Dr. Luchs: Clinical presentation should guide you to institute the therapy that you feel is required. In many cases, I will start a steroid because of acute symptomatology, and then I will also put Restasis on board at the same time. A red, inflamed eye is not going to calm down quickly with cyclosporine, so try a more broad-spectrum antiinflammatory to quickly calm down the clinical presentation. But you also need the cyclosporine on board to target the T-cell inflammatory process of dry eye that is the root cause here.
Dr. Donnenfeld: Anytime you use medications, the primary concern should always be the risk/reward ratio. A medication that is broad spectrum tends to have more side effects, versus a more narrow mechanism of action that will tend to have fewer side effects. So you balance this depending on the patient. If the patient is extremely symptomatic, I use a broad-spectrum antiinflammatory and, once the disease is controlled, I move to a more targeted treatment that minimizes the side effects and controls the pathology.
Neurostimulation
Dr. Sheppard: We now have a novel alternative to pharmacotherapy for the treatment of DED. The first neurostimulation device for dry eye, TrueTear (Allergan). It offers a completely new mechanism of action and is available to patients nationwide. This handheld, intranasal device stimulates the normal afferent and efferent trigeminal pathways that produce all three key constituents of natural tears: aqueous, lipid, and mucin, in physiological concentrations.17,18 While neurostimulation is an established therapy in other fields, it is completely new to eye care (Figure 4). How do you predict this will integrate with current therapeutics for DED?
(Courtesy of Allergan)
Figure 4. Neurostimulation is a new mechanism of action for treating DED.Dr. Donnenfeld: The history of neurostimulation is full of success stories. Almost every disease you can think of as chronic and irritating has a neurostimulation device that has been helpful in treating it. Introducing this device for the treatment of DED, I believe, will be as disruptive as the introduction of Restasis more than a decade ago.
Dr. Gupta: I think there is still so much to learn about neurostimulation in terms of ophthalmic use. This is going to be a very novel technology and may even have the ability to stimulate multiple layers of the tear (aqueous, mucin, and lipid). Potential applications are great, and I believe there are many patients out there that want to have a device that they can use at home to treat their DED.
Dr. Yeu: Meibography has shown us what happens to the architecture of each gland. Inflammation (such as in rosacea blepharitis) or a poor, incomplete blink that commonly occurs with contact lens wear and computer use, lead to congestion of the glands, which leads to truncation and finally atrophy (Figure 5). But a device like TrueTear constantly promotes egress of fluid and meibum, and I feel will be restorative to the gland architecture. We have seen some change over time in patients who do not have complete atrophy, and I am excited to see what else we will learn to improve science and health in the future.
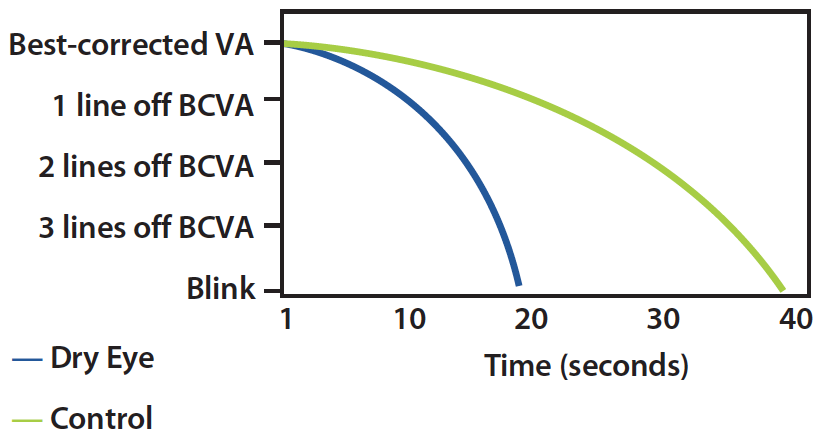
Figure 5. Rate of visual acuity decay between blinks.19
Dr. Donnenfeld: What is exciting about neurostimulation is that it allows the patients to take control of their ocular surface disease. All the therapies that we have used in the past have really been predicated on the idea of maximum dosage: instilling cyclosporine six times per day is not better than two times per day. This is a therapeutic that allows the patient to control the disease process. I believe patients will overwhelmingly accept a therapy that allows them to be the arbiters of their own disease.
Dr. Luchs: Having seen the time-lapse photography of the meibomian sphincter activated by TrueTear, I believe it will be very useful in reducing gland atrophy that leads to gland drop out over time. I postulate that it will be more effective than thermal pulsation therapy simply because it can be repeated throughout the day, whereas pulsation therapy is only one time.
Dr. Donnenfeld: I believe that neurostimulation and thermal pulsation therapy both have real value and will be additive to each other. While DED has received a lot of attention for the past 15 years, there has been very little movement in the market for meibomian gland dysfunction. This new therapy will provide us a great tool to address this unmet need in disease management.
Dr. Yeu: I am very excited to see how a neurostimulation pathway will work with thermal pulsation and even lid exercises to allow us to diagnose and treat meibomian gland dysfunction much earlier. My guess—a very nice synergy will occur between TrueTear and thermal pulsation therapy.
Applying Science to the Clinic
Dr. Sheppard: Now that we have discussed our tried-and-true and upcoming therapies, let us figure out how we would apply them in these six common patient profiles.
Patient Profile 1
A postoperative LASIK patient with terrible ocular surface disease.
Dr. Donnenfeld: It is important to look at the mechanism of action of dry eye. In most cases, it is T-cell mediated. However, after LASIK, it is overwhelmingly anesthetic, with the loss of the feedback loop to the lacrimal gland. TrueTear directly stimulates the feedback loop that is lost, so that might be my first line therapy because you are basically pairing the mechanism of disease precisely to the therapy.
Dr. Yeu: Comparable studies demonstrate that patients on cyclosporine demonstrate better subbasal nerve regeneration, so this is an example of a great patient to start cyclosporine earlier rather than later.20
Dr. Sheppard: When you have a viable, measurable, quantitative, anatomical biomarker through confocal microscopy, you can very clearly measure the length and quality of corneal nocioceptors nerves and compare regeneration rates. In my mind, there may be a huge pre-emptive use for neurostimulation in virtually all LASIK, penetrating keratoplasty, and even LRI patients.
Patient Profile 2
A 65-year-old woman with no prior history of DED is diagnosed with DED for the first time after cataract surgery. (Note: Only 13% of patients report dry eye symptoms half of the time or more, but >60% have a very abnormal tear break-up time [<5 seconds], >50% have central corneal staining and >20% have a very low Schirmer score [<5mm].21)
Dr. Luchs: While this patient did not have a diagnosis of dry eye, she most likely did have it preoperatively and just became symptomatic postoperatively. Thus, I believe she should be started with immunomodulatory treatment, and then she may also benefit from TrueTear.
Dr. Donnenfeld: In my clinical experience, the most unhappy patient is the dry eye patient who was marginally compensated so felt normal before surgery, and you missed the diagnosis, and they develop more severe dry eye after surgery. This is the prototypical patient in which preoperative point-of-service testing could have helped diagnose dry eye prior to surgery, and the reason why many of us advocate for routine testing of every patient.
Dr. Trattler: In this patient who developed dry eye after surgery, her main symptom is typically not pain or discomfort, but blurry or fluctuating vision. A patient dissatisfied with their quality of vision following surgery, no matter the reason, will directly relate it to the skills of the surgeon in an unfavorable manner. That makes it important to diagnose dry eye and treat it preoperatively.
Patient Profile 3
A postoperative LASIK patient who has developed DED. The patient is currently using the TrueTear stimulator successfully, but he or she needs additional therapy. What is your next choice? Do you throw the book at the patient to get him or her better as fast as possible, or do you go scientific?
Dr. Trattler: The first step here is to find out how they are using the neurostimulator. If they are using it only once per day, they may need to be coached that it is okay to use more often, or at different times of day. As we get more experience, we will be able to help our patients be more successful with this treatment modality.
Dr. Luchs: The second step is to know exactly what is going on. Do they have untreated meibomian gland dysfunction? Does their tear film need more support? Do they need an immunomodulator? Whether you choose an antiinflammatory, thermal pulsation, or punctal plugs depends on the individual patient.
Patient Profile 4
A rosacea patient with DED.
Dr. Stonecipher: Rosacea in a female patient wearing makeup can be difficult to recognize, so the diagnosis is the first step. My first therapy would be doxycycline or metronidazole gel, or another tested and proven rosacea therapy. I have had great success treating the lid margins and face with intense pulsed light therapy. It is also necessary to reduce the bacterial load on the lid margins with hypochlorous acid or another, similar product. Immunomodulators can also play a role in reducing inflammation.
Dr. Gupta: I am much more worried about chronic undertreated lid margin disease related to rosacea, as well as the related potential keratitis and corneal neovascularization. A meibomian gland-focused therapy would be front line.
Dr. Sheppard: There are excellent patients for a very intensive nutraceutical therapy. In addition, I would manage the ocular surface with appropriate therapeutics. I also recommend topical azithromycin pulse therapy for these cases.
Patient Profile 5
A symptomatic dry eye patient who uses toric soft contact lenses.
Dr. Luchs: A toric lens is a tight-fitting lens, and many users are dry. This would be a great patient for neurostimulation because it provides an immediate increase in tear film volume.
Dr. Donnenfeld: I like neurostimulation for this patient, but I would start them on cyclosporine. Many patients have concomitant ocular allergy and inflammatory disease and, with Restasis, you are treating both the DED and the inflammatory component of their allergic reaction. TrueTear does not address the inflammatory component, so this is a perfect patient to use both therapies.
Dr. Gupta: I agree with those treatments, and would also be okay prescribing a pulsed dose of steroid if I was able to dictate the safety measures: that they come back for a pressure check and also understood that abuse of steroids can lead to negative consequences, and as such would benefit from Restasis or Xiidra for chronic long-term therapy.
Patient Profile 6
A keratoconic patient using rigid gas permeable lenses with symptomatic dry eye.
Dr. Sheppard: Therapy here is the same as for the soft contact lens wearer: an immunomodulator to address inflammation and a neurostimulator. However, there is an added imperative that this patient get out of rigid gas permeable lenses.
Dr. Trattler: I agree with getting this patient out of rigid gas permeable lenses. One recent advance is the scleral lens for keratoconus. Scleral lenses rest on the sclera and avoid touching the cornea. In my experience, keratoconus patients report excellent vision with scleral lenses and less dry eye.
Dr. Stonecipher: Another benefit of scleral lenses is that they not only allow the patient to see, but also allow a nice big tear lake to be on the eye. Punctal occlusion would also be an excellent option for this patient.
This educational supplement is sponsored by Allergan Global Medical Affairs.
1. Wohler J, Bullard D, Schoeb T, Carnum S. LFA-1 is critical for regulatory T cell homeostasis and function. Mol Immunol. 2009;46(11-12):2424-2428.
2. Siemasko KF, Gao J, Calder VL, et al. In vitro expanded CD4+ CD25+ Foxp3+ regulatory T cells maintain a normal phenotype and suppress immune-mediated ocular surface inflammation. IOVS. 2008;49:5434-5440.
3. Calonge M, Enríquez-de-Salamanca A, Diebold Y, et al. Dry eye disease as an inflammatory disorder. Ocul Immunol Inflamm. 2010;18:244–253.
4. Stevenson W, Chauhan SK, Dana R. Dry eye disease: an immune-mediated ocular surface disorder. Arch Ophthalmol. 2012;130:90–100.
5. Marsh P, Pflugfelder SC. Topical non-preserved methylprednisolone therapy of keratoconjunctivitis sicca in Sjogren’s syndrome. Ophthalmology. 1999;106:811–816.
6. AP. Drug that reduces risk in transplants gets early approval. http://www.nytimes.com/1983/09/03/us/drug-that-reduces-risk-in-transplants-gets-early-approval.html. Published September 3, 1983. Accessed May 26, 2017.
7. Nussenblatt RB, Palestine AG. Cyclosporine: immunology, pharmacology and therapeutic uses. Surv Ophthalmol. 1986;31(3):159-169.
8. Lee SH, Chung H, Yu HG. Clinical outcomes of cyclosporine treatment for noninfectious uveitis. Korean J Ophthalmol. 2012;6(1):21–25.
9. Mathews D, Mathews J, Jones NP. Low-dose cyclosporine treatment for sight-threatening uveitis: efficacy, toxicity, and tolerance. Indian J Ophthalmol. 2010; 58(1):55–58.
10. Tatlipinar S, Akpek EK. Topical ciclosporin in the treatment of ocular surface disorders. Br J Ophthalmol. 2005;89:1363–1367.
11. Restasis Package Insert. Allergan, Inc. 2016.
12. Roberts C, Carniglia P, Brazzo B. Comparison of topical cyclosporine, punctal occlusion, and a combination for the treatment of dry eye. Cornea. 2007;26(7):805-809.
13. Brew BJ, Davies NW, Cinque P, et al. Progressive multifocal leukoencephalopathy and other forms of JC virus disease. Nat Rev Neurol. 2010;6(12):667-679.
14. XIIDRA Package Insert. Lexington, MA: Shire US Inc.; 2016.
15. Donnenfeld ED, Karpecki PM, Majmudar PA, et al. Safety of lifitegrast ophthalmic solution 5.0% in patients with dry eye disease: a 1-year, multicenter, randomized, placebo-controlled study. Cornea. 2016;35(6):741-748.
16. Wu MM, Yau GS, Lee JW, et al. Retrospective review on the use of topical cyclosporin a 0.05% for paediatric allergic conjunctivitis in Hong Kong Chinese. Scientific World Journal. 2014;2014:396987.
17. Pondelis NJ, Dieckmann G, Kataguiri P, et al. Intranasal Neurostimulator induces morphological changes in meibomian glands in patients with dry eye disease. Presented at: ARVO; May 7-11, 2017; Baltimore, Maryland.
18. Woodward AM, Senchyna M, Franke M, et al. Effect of intranasal neurostimulation on tear protein content in patients with dry eye. Presented at: ARVO; May 7-11, 2017; Baltimore, Maryland.
19. Walker P, Ousler III GW, Workman DA, et al. Visual function in normals compared to patients diagnosed with dry eye as measured by the inter-blink interval visual acuity decay (IVAD) test. IOVS. 2007;48:ARVO e-Abstract 422.
20. Iaccheri B, Torroni G, Cagini C, et al. Corneal confocal scanning laser microscopy in patients with dry eye disease treated with topical cyclosporine. Eye (Lond). 2017;31(5):788-794.
21. Trattler WB, Reilly CD, Goldberg DF, et al. Cataract and dry eye: prospective health assessment of cataract patient ocular surface (PHACO) study. Paper presented at: ASCRS Symposium and Congress; May 25-29, 2011; San Diego, CA.
