Key Takeaways
- Surgeons consider how to proceed after the posterior capsule ruptures during phacoemulsification.
- Key to complication management in this situation is not to withdraw instruments from the eye immediately.
- Possible IOL fixation strategies include revised positioning in the bag, forms of optic and haptic capture, scleral fixation, and sulcus fixation.
CASE PRESENTATION
A 58-year-old man seeking improved vision presents for a consultation. The patient’s BSCVA is -2.00 -0.25 x 95º = 0.2 OD and -1.50 -1.50 x 83º = 0.2 OS. He is experiencing diplopia in the right eye.
An examination of the anterior and posterior segments finds a clear cornea, advanced corticonuclear lens opacification, and posterior capsular opacification in each eye. Both optic nerves and retinas appear to be healthy. He has no known history of ocular surgery or disease.
The patient is scheduled for cataract surgery with a monofocal aspheric IOL (Akreos Adapt AO, Bausch + Lomb). Preoperative measurements with the IOLMaster 700 (Carl Zeiss Meditec) find total corneal astigmatism of -0.59 D OD and -0.46 D OS. The target refraction is -2.00 D in each eye.
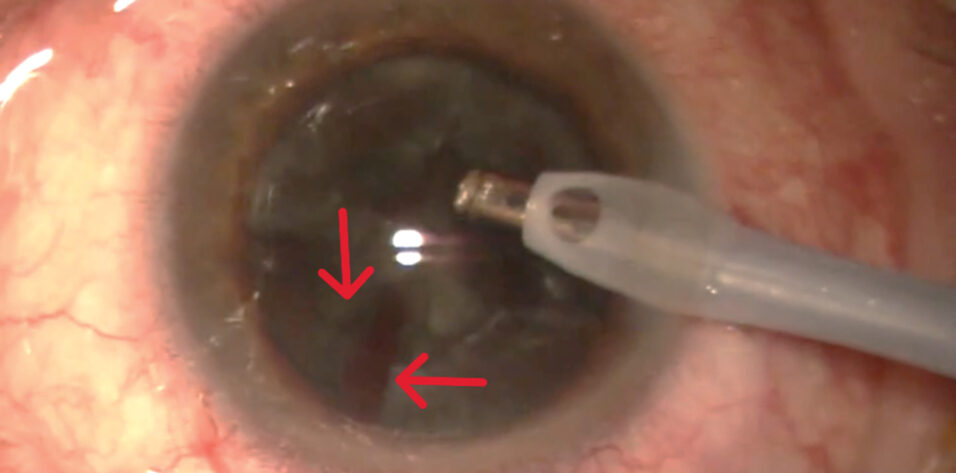
During phacoemulsification in the right eye, a posterior capsular rupture (PCR) with vitreous prolapse occurs (Figure 1). How would you proceed?

Figure 1. The posterior capsule ruptures during phacoemulsification of the right eye.
Courtesy of Suphi Taneri, MD, FEBOS-CR
— Case prepared by Suphi Taneri, MD, FEBOS-CR

FRANCESCO CARONES, MD, FWCRS
Before the phaco instrument is retracted from the anterior chamber, a small amount of an OVD would be injected to prevent further vitreous prolapse. Next, an anterior vitrectomy (with or without the aid of a visualization agent such as triamcinolone acetonide) would be performed.
Once the anterior segment stabilizes, the questions would be which IOL to implant and where. The PCR seems to be limited enough to allow an IOL to be implanted in the capsular bag, as originally planned. My preference would be a lens with a modified C-loop haptic design, its haptics oriented 90º away from the PCR. If extension of the PCR raises safety concerns related to positioning the IOL in the capsular bag, I would opt for the same IOL design but perform reverse optic capture, with the haptics in the capsular bag and the optic located in front of the anterior capsule and captured by the capsulorhexis (requiring it to be smaller than the optic diameter). If neither of these implantation strategies is viable, I would perform scleral fixation of a Carlevale IOL (Soleko).

KENDALL E. DONALDSON, MD, MS
Phacoemulsification would be halted immediately upon the detection of vitreous loss. The second instrument would be withdrawn, and a dispersive OVD would be injected through the sideport incision before removal of the phaco handpiece. The bottle height would be lowered. Ideally, an OVD would be instilled in the region of the PCR and used to elevate the nuclear remnants and form a cushion between the vitreous and residual lens material.
It appears that a large amount of nuclear and cortical material remains. A limited pars plana vitrectomy would be performed at the highest possible cut rate, with the cutter placed 3.5 mm posterior to the limbus and infusion delivered through the paracentesis. Once the vitreous prolapse has largely resolved, the vitrector would be passed through a second sideport incision to allow removal of the remaining lens material. A dilute mixture of preservative-free triamcinolone acetonide would then be injected to identify any residual vitreous in the anterior chamber.
After the removal of all vitreous and lens material, a three-piece IOL would be implanted in the sulcus, and optic capture within the anterior capsulotomy would be attempted. If the capsulotomy is larger than 6 mm, however, optic capture would not be possible. In this situation, the lens would be placed entirely within the sulcus without capture, and the IOL power would be reduced by 1.00 D to maintain the original refractive target. Finally, carbachol intraocular solution (Miostat, Alcon) would be injected to ensure uniform pupillary constriction, and the wound would be closed with a single 10-0 nylon suture.

ARTEMIS MATSOU, MD, MRCP(UK), FEBOS-CR, FEBO, PGDIP CRS
In Figure 1, it appears that the PCR occurred with a substantial amount of nuclear material still present. My priorities would therefore be to prevent a dropped nucleus and remove the remaining lens material in a controlled fashion.
The moment a PCR happens, the basic principles for the surgeon are to stay calm, continue irrigation, keep the instruments in the eye to avoid chamber collapse, inject a dispersive OVD to plug the rent, and shift to slow-motion fluidics (low bottle height, IOP, flow, and vacuum).
With a sizable, reasonably dense nucleus still in the bag and a PCR with vitreous prolapse in the anterior chamber, continuing standard phacoemulsification would be risky. I would therefore convert to a manual approach.
A sub-Tenon block would be administered, and a 5.5- to 6-mm scleral tunnel with a good posterior lip would be created. Viscoexpression of the nucleus would be performed with an irrigating vectis and a large amount of an OVD. Triamcinolone acetonide would be injected to guide a bimanual anterior vitrectomy and ensure the clearance of all vitreous and lens material. A high cut rate and low vacuum would be used, with the cutter positioned posterior to the iris.
Because the anterior capsulorhexis appears to be intact, my preference would be to implant a three-piece monofocal IOL in the sulcus. The backup three-piece IOL in my OR is a spherical Sensar AR40e (Johnson & Johnson Vision). If optic capture can be executed successfully, the original IOL power would be maintained; the small theoretical contrast sensitivity trade-off would be outweighed by the stable effective lens position and centration that optic capture offers. If optic capture is not possible but sulcus support is adequate, the three-piece IOL would be placed in the sulcus, but its power would be reduced by approximately 0.50 D to account for the more anterior effective lens position.
After constricting the pupil, I would inject diluted triamcinolone acetonide, perform a final check to confirm that no vitreous remains in the anterior chamber, and then suture the wounds. Aggressive treatment of inflammation would be initiated postoperatively. Once the eye quiets and the refraction stabilizes, I would confirm that the patient’s preoperative diplopia was purely monocular and has resolved.
During surgery on the left eye, I would be on high alert for complications, and a backup three-piece IOL would be readily available in the OR.

WHAT I DID: SUPHI TANERI, MD, FEBOS-CR
As the panelists emphasize, when the PCR with vitreous prolapse occurred early during phacoemulsification in the right eye, my priorities were to (1) keep the chamber formed, (2) eliminate vitreous traction, and (3) protect the anterior capsulorhexis because that structure determines the safest fixation options.
First, I stabilized the anterior chamber before withdrawing the instruments. I immediately paused phacoemulsification and injected a dispersive OVD through the sideport incision to tamponade the rent and reduce further vitreous migration. I removed the phaco handpiece only once the chamber was stable. Next, I cleared the vitreous in a controlled, low-traction way. I performed a bimanual anterior vitrectomy using high cut rates and low aspiration to minimize vitreoretinal traction. I injected dilute triamcinolone (1:5) to identify and confirm complete removal of the vitreous from the anterior chamber and incisions. I then performed careful cortical cleanup without chasing any material that might have migrated posteriorly to avoid enlarging the defect and creating additional traction.
IOL Selection and Fixation
Although the posterior capsule had been compromised, the anterior capsulorhexis was intact, continuous, and well centered. Rather than switch to sulcus placement or scleral/iris fixation, I performed diagonal haptic capture (DHC) of the originally planned Akreos Adapt AO IOL because its four-loop haptics are well suited to this rescue approach when the capsulorhexis is preserved. My goal was to implant the aspheric IOL to which the patient had consented and achieve stable centration without sulcus contact or suturing while keeping the effective lens position predictable.
The DHC technique was originally described by Jin and Zhang.1 I intentionally positioned two diagonally opposed haptics posterior to the anterior capsular rim and captured the other two haptics anterior to the rim in a diagonal configuration, creating a stable cross-locking effect across the capsulorhexis. I confirmed centration and stability before removing the OVD (Figure 2).

Figure 2. Schematic illustration of the DHC of an IOL. Two opposing loop haptics are placed in front of and two haptics behind the well-centered capsulorhexis to provide long-term, biocompatible, stable IOL fixation.
Outcome
In the first days after surgery, the patient’s visual acuity was low, as expected, due to corneal edema and Descemet folds. The IOP was within a normal range, the IOL was well centered, and no vitreous was visible in the anterior chamber. At his final follow-up visit 3 years after IOL implantation, his manifest refraction was close to the intended -2.00 D at -1.75 -0.25 x 2° = 1.0 (20/20) OD. The lens sat firmly and exhibited no tilt.
Final Thoughts
During the past 10 years, I have performed DHC in 20 eyes and have seen no evidence of pigment dispersion glaucoma, chronic inflammation, or late dislocation thus far. Care should be taken, however, not to generalize these encouraging results to other IOL models.
Dr. Taneri used an AI language model (ChatGPT 5.2, OpenAI) to assist with language editing. All content was reviewed, verified, and revised by the article’s authors, who assume full responsibility for the accuracy and integrity of the manuscript. The AI tool was not used for data analysis, interpretation, or drawing scientific conclusions.
1. Jin H, Zhang H. Diagonal haptic capture of a plate intraocular lens with 4 haptics. J Cataract Refract Surg. 2020;46(4):503-506.